5-Hydroxyindole-2467-d 4-3-acetic-22-d 2 Acid. Chemically rosmarinic acid is a caffeic acid ester with tyrosine providing another phenolic ring via dihydroxyphenyl-lactic acid.

Cyclohexaneacetic Acid 99 5292 21 7
45 An aqueous solution of hydrogen peroxide acetic acid peroxyacetic acid octanoic acid peroxyoctanoic acid sodium 1-octanesulfonate and 1-hydroxyethylidene-11-diphosphonic acid.
Cyclohexyl acetic acid. It is currently marketed in capsule tablet and oral solution formulations. The primary aim of the cards is to promote the safe use of chemicals in the workplace. 1581694-94-1 Isotopic Enrichment.
The cards are data sheets intended to provide essential safety and health information on chemicals in a clear and concise way. CI Fluorescent Brightner 71 244 250 or260. It is rare with unknown prevalence.
NA Isotopic Enrichment. The positively charged structures are referred to as carbocations whereas the negatively charged hydrocarbons are called carbanions. QMA 0005 mg6 dm² s.
005 13- Phenylenedioxydiacetic acid. Haloalkanes or alkyl halides are the functional groups which contain a bond between a carbon atom and a halogen. Only those codes applicable to the University of Maryland are listed Hazardous waste is any solid waste that either exhibits any of the characteristics of hazardous waste or is a listed EPA waste.
Phosphoric acid Acetic acid Conversion products of phosphorus nitrile chloride max. For medical fire or law enforcement assistance call 911 For EHS emergencies including after hours call 650 725-9999. COASY is a bifunctional enzyme that catalyzes the 2 last steps in CoA synthesis.
Download the SDS sheet for the Kirkland Signature Heavy Duty Laundry Detergent - 119903. The presence of. NA Isotopic Enrichment.
2-Propenoic acid telomer with sodiumsulfite 11 sodium salt. In addition EPA Hazardous Waste Codes are also classified as acute and non-acute. Methacrylic acid cyclohexyl ester.
Catalogue D-7955 CAS Number. These activities are performed by 2 separate enzymes phosphopantetheine adenylyltransferase PPAT. The main target users are workers and those responsible for occupational safety and health.
Acetic acid Butyric acid n- Formic acid Propionic acid Rosin Oil Tall oil Group 3. I For any combination consisting solely of those unlawful substances listed in subdivisions a through h of this paragraph an amount equal to or in excess of the threshold amount as determined by the application of section 13-3420. Examples include names such as bornyl and cyclohexyl.
The level of free fatty acid FFA in the fat of cocoa beans measures the rancidity of the cocoa. For example the simplest nitrate ester methyl nitrate is formed by reaction of methanol and nitric acid in the presence of sulfuric acid. It is reported that the symmetrically substituted isomeric diol drawn in the shaded box rearranges exclusively by a methyl shift but.
And still bottoms from the recovery of these spent solvents and spent solvent mixtures. Only a few cases have been described. In this event the acetate becomes the favored leaving group presumably coordinated with acid followed by a 12-phenyl shift.
In recent years however gabapentin has. H Two pounds of marijuana. In addition to use on food-processing equipment and utensils this solution may be used on food-contact surfaces in public eating places subject to the limitations in paragraph c39 of this section.
Nitrate esters are typically prepared by condensation of nitric acid and the alcohol. Abbreviations the shortened form of a word or phrase and acronyms words formed from the initial letters of a phrase are commonly used in technical writing. Ethylene glycol monomethyl ether acetate 136 90-02-8 salicylaldehyde 137.
Paris Nomina Anatomica partial nail avulsion percutaneous needle aspiration Personal Needs Allowance Pharmaceutical Needs Assessment purine nucleoside analogue. The ICSC project is a common undertaking between the World Health Organization WHO and. This condensation is sometimes called nitroxylation.
CH 3 OH HNO 3 CH 3 ONO 2 H 2 O. 886 Free fatty acid FFA content. Hawkinsin 4-dihydroxy-cyclohexyl acetic acid is formed from tyrosine metabolite precursors with only minimal to no elevation of tyrosine.
QMA 005 mg6 dm². Annex 1 list III11. Catalogue D-7978 CAS Number.
G One-half milliliter of lysergic acid diethylamide or in the case of blotter dosage units fifty dosage units. 98 atom D Read More. P-listed codes and certain dioxin codes F020-F023 and F026-F028 are considered to be acute.
High levels of FFA in cocoa are not acceptable and in Europe manufacturers may not legally sell cocoa butter containing over 175 FFAThe presence of large amounts of FFAs in the fat of. Amines and Alkanolamines Aminoethylethanolamine Aniline Diethanolamine Diethylenetriamine Diisopropanolamine Dimethylamine Ethylenediamine Hexamethylenediamine 2-Methyl-5-ethylpyridine Monoethanolamine Monoisopropanolamine. QM T 1 mgkg in FP ea.
NCO Resorcinol diglycidyl ether. In the first years of life affected patients can present with failure to thrive and metabolic acidosis but this disorder appears to be asymptomatic later in life. Antiredeposition agent Viscosity modifier.
We are a leading supplier to the global Life Science industry with solutions and services for research biotechnology development and production and pharmaceutical drug therapy development and production. Caustics Caustic potash solution Caustic soda solution Group 4. Ethylene glycol monoethyl ether acetate 134 108-05-4 vinyl acetate 135 110-49-6 2-methoxyethyl acetate.
Acetic acid icsc 2-ethoxyethyl acetate icsc acetyl bromide icsc 111-trimethylolpropane icsc ethyl acetate icsc n-decyl acrylate icsc adipic acid icsc alachlor icsc allyl isothiocyanate icsc aluminum hydroxide icsc n-butylamine icsc m-anisidine icsc antipyrine icsc. 98 atom D Read More. All spent solvent mixturesblends containing before use a total of ten percent or more by volume of one or more of the above non-halogenated solvents or those solvents listed in F001 F002 and F005.
130 94-74-6 4-chloro-2-methylphenoxyacetic acid. MCPA 131 563-47-3 3-chloro-2-methyl-1-propene 132-cobalt and its compounds 133 111-15-9 2-ethoxyethyl acetate. Methiocarb-d 3 N-methyl-d 3 Catalogue D-7966 CAS Number.
This regulation designates under section 102a of the Comprehensive Environmental Response Compensation and Liability Act of 1980 the Act those substances in the statutes referred to in section 10114 of the Act identifies reportable quantities for these substances and sets forth the notification requirements for releases of these substances. The action of a Lewis acid in acetic anhydride on the other hand may selectively acetylate the less hindered dimethyl carbinol. 0001 Sodium hydrogen carbonate max.
Robin Dand in The International Cocoa Trade Third Edition 2011. EC 27124 in prokaryotes. Gabapentin is a prescription medication approved by the Food and Drug Administration FDA for the treatment of neuropathic pain and epileptic disorders.
EC 2773 and dephospho-CoA kinase DPCK. The following spent non-halogenated solvents. Rosmarinic acid was first isolated and characterized in 1958 by the Italian chemists Scarpatti and Oriente from rosemary Rosmarinus officinalis after which the acid is named.
02 Methyl-tris-sec-butylaminosilane 12 Methyl-tris-acetoxysilane Ethyl-tris-acetoxysilane Methyl-tris-butanoximosilane 12 Di-4-methyl-benzoylperoxide Esters of alkyl-C1-C8-silicic acid or orthosilicic acid with. Biosynthesis of coenzyme A CoA from pantothenic acid vitamin B5 is an essential universal pathway in prokaryotes and eukaryotes. 99 atom D Read More.
05 7. Cresols and cresylic acid and nitrobenzene. 2-1-aminomethyl cyclohexyl acetic acid.
The hydrocarbon functional groups may have an ionic charge on them.
2-Aminoethanol chlorosulfonic acid ethylene diamine ethyleneimine NFPA 1991. Store highly volatile flammable liquids in an explosion-proof refrigerator.
180 Heartland Blvd Edgewood.

Is acetic acid flammable. Irritating vapors are generated when heated. Emergency eye wash fountains and safety showers should be available in the immediate vicinity of use or handling. Acids bases oxidizers and poisons.
It is possible to buy fizzy paracetamol tablets. 64-19-7 Acetic acid ACGIH TLV. 64-19-7 Product code.
Freezing and thawing does not affect product quality. 35071253 35071202 Part of Kits. Hydrochloric acid Sulfuric acid Phosphoric acid Hydrofluoric Acid etc Poisons Toxic chemicals Many are in aqueous.
How to check that ethanoic acid and ethanol reaction has reached equilibrium. 25mgm3 64-19-7 Acetic acid OSHA PEL. Acetic acid chromic acid nitric acid ethylene glycol perchloric acid peroxides and permanganates Acetone concentrated sulfuric and nitric acid mixtures Acetylene copper tubing fluorine bromine chlorine iodine silver mercury Ammonia anhydrous mercury halogens calcium hypochlorite hydrogen fluoride HF Ammonium Nitrate acids metal powders flammable liquids chlorates nitrates.
Dangerous when exposed to heat or fire. Glacial Acetic Acid Catalog Number. Section 8 - Exposure Controls Personal Protection.
Acetic Acid Safety Data Sheet according to Federal Register Vol. 14 09282020 EN English US Page 1 SECTION 1. Because it is highly volatile and flammable it is generally not used on automatic tissue processors.
Glacial acetic acid solidifies below 62F 17C. Work in a well-ventilated area or fume cupboard Wear eye protection safety glasses or goggles. Ensure that dust-handling systems exhaust ducts dust collectors vessels and processing equipment are designed to prevent the escape of dust.
Store in flammable storage cabinet and away from sources of ignition. Flammable vapour and liquid. It will contract slightly on freezing.
29 ferric chloride 200 ml Distilled water 4750 ml Hydrochloric acid 50 ml Mix well label with initial and date. Class III Combustible Liquid Liquids having flashpoints at or above 140F 60C 1910106a18ii. After mixing reactants give some time to.
Wear appropriate personal protective equipment as specified in Section 8. Ethanoic acid acetic acid Pure liquid glacial Also methanoic formic acid. Propanone acetone is volatile and highly flammable.
Accidental Release Measures Ventilate area of leak or spill. Public health information CDC Research information NIH SARS-CoV-2 data NCBI Prevention and treatment information HHS Español. Acetic acid should be kept above its freezing point of 62F17C to allow it to be handled as a liquid.
Flammable liquids oxidizers poisons and acids. Class III liquids are subdivided into two subclasses. Substance Substance name.
Ethanol reacts with acetic acid to give ethyl ethanoate which is an ester compound. Remove all sources of ignition. Ethanol is a volatile flammable colorless liquid with a slight characteristic odor and carboxylic acid is a colourless liquid and has a strong odor very much like vinegar.
Nitroparaffins Inorganic bases amines. 58 Monday March 26 2012 Rules and Regulations Issue date. Class IIIA Flash point at or above 140 F and below 200 F.
Class IIIA Combustible Liquid Liquids having flashpoints at or above 140F 60C and below 200F except any. Flammable solids self-reactive substances solid desensitised explosives and polymerizing substances. Acetone should not be used on some tissue processors because it will adversely affect seals and other components of the equipment.
DO NOT taste the paracetamol. Flammable liquid and vapor. Butyric acid Pentanoic acid etc.
Oxalic acid Silver mercury and their salts. Acetic acid solution with more than 10 percent and less than 50 percent acid by mass 8 UN2790 III 8 IB3 T4 TP1 154 203 242 5L 60 L A Acetic anhydride 8 UN1715 II 8 3 A3 A6 A7 A10 B2 IB2 T7 TP2 154 202 243 1L 30 L A 40 Acetone 3 UN1090 II 3 IB2 T4 TP1 150 202 242 5L 60 L B Acetone cyanohydrin stabilized 61 UN1541 I 61 2 A3 B9 B14 B32 B76 B77 N34 T20 TP2 TP13 TP38. Substances which on contact with water emit flammable gases.
Laboratory chemical Identification of Manufacturer. Product and Company Identification Product Identifier. Acetic acid glacial fuel oil no.
This reaction is heightened by the presence of mineral acids nitric perchloric sulfuric acid etc Chem. It is incorporated in compound fixatives. May be harmful if absorbed through the skin.
VMA-Skreen Glacial Acetic Acid Chemical Name. 25mgm3 Appropriate Engineering controls. Nitrites Potassium or sodium cyanide.
Polymers of allene or its equilibrium mixture methylacetylene - allene are reactive and refractory impurities which if exposed to air slowly autoxidize to dangerous peroxidic compounds. Keep away from flames. Acidic Flammable Liquids.
Mixing acetic acid in equal molar portions with any of the following substances in a closed container caused the temperature and pressure to increase. Water diluted acid can re act with metals to form hydrogen gas. Glacial acetic acid 100 Acetic acid 80 Acetic anhydride Formic acid 85 Propanoic acid 100 also called Propionic acid Mixtures of acids and flammable liquids Organic Acids.
Special Hazards of Combustion Products. Acetic acid-D C2H4O2 CID 2723902 - structure chemical names physical and chemical properties classification patents literature biological activities safetyhazardstoxicity information supplier lists and more. Reacts violently with water to generate acetic acid.
Classification of Flammable and. Formic acid is a colorless liquid having a pungent penetrating odor at room temperature comparable to the related acetic acidIt is miscible with water and most polar organic solvents and is somewhat soluble in hydrocarbonsIn hydrocarbons and in the vapor phase it consists of hydrogen-bonded dimers rather than individual molecules. Owing to its tendency to hydrogen-bond gaseous formic.
Use water spray to dilu te spill to a. Both acids cause severe skin burns and eye damage. Glacial Acetic Acid 01.
Substances liable to spontaneous combustion. Acetic acid or acetic anhydride can explode with nitric acid if not kept cold. Dilute spills to non-flammable mixtures.
35070303 35070001 35070401 Use of chemical. For a 15-minute exposure the concentration of methanoic acid vapour in. USCG 1999 Health Hazard.
Acetic acid solution with more than 10 percent and less than 50 percent acid by mass 8 UN2790 III CORROSIVE YES CORROSIVE 1 L 5 L 30 L 154 203 Acetic anhydride 8 3 UN1715 II CORROSIVE FLAMMABLE LIQUID YES CORROSIVE FLAMMABLE LIQUID 05 L 1 L 30 L A3 A7 A10 154 202 Acetone 3 UN1090 II FLAMMABLE LIQUID YES FLAMMABLE LIQUID 1 L 5 L 30 L 150 202 Acetone. Usually supplied as 90 solution about 20 M Some descalers for kettles irons etc use methanoic acid. Potassium hydroxide residue in a catalyst pot reacted violently when acetic acid was.
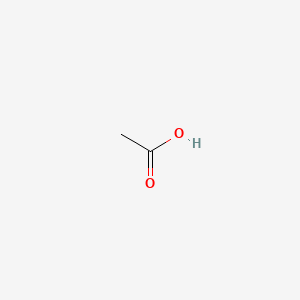
Acetic acid CH 3 COOH is coagulant in action with nucleic acids but generally does not fix proteins. Stable for 1 year. Acetic acid ə ˈ s iː t ɪ k systematically named ethanoic acid ˌ ɛ θ ə ˈ n oʊ ɪ k is an acidic colourless liquid and organic compound with the chemical formula CH 3 COOH also written as CH 3 CO 2 H C 2 H 4 O 2 or HC 2 H 3 O 2.
Acetic acid acetone alcohol aniline chromic acid flammable gases and liquids hydrocyanic acid hydrogen sulfide and nitratable substances. Acetic acid naphtha and stoddard solvent. Vinegar is no less than 4 acetic acid by volume making acetic acid the main component of vinegar apart from water and other trace elements.
In these tablets the paracetamol has been mixed with citric acid 2-hydroxypropane-123-tricarboxylic acid and sodium hydrogencarbonate. Approved flammable storage cabinet. Weigerts Hematoxylin Working Solution Solution A 250 ml Solution B 250 ml Mix well solution will remain stable for 3 - 4 days.
Oxygen liquid or enriched air Flammable gases liquids or solids such as acetone acetylene grease hydrogen oils. Corrosive avoid contact and inhalation. Kee p unnecessary and unprotected personnel from entering.
Anhydride manufactured by acetic acid pyrolysis sometimes contains ketene polymers eg acetylacetone diketene dehydroacetic acid and particulate carbon or soot is occasionally encountered. Flammableavoid contact and inhalation.
Potassium hydroxide residue in a catalyst pot reacted violently when acetic acid was added MCA Case History 920. It is abundant multivalent and nonmetallicUnder normal conditions sulfur atoms form cyclic octatomic molecules with a chemical formula S 8Elemental sulfur is a bright yellow crystalline solid at room temperature.
Peracetic acid is a colorless liquid with a strong pungent acrid odor.
Molecular weight of glacial acetic acid. Ethyl acetate ethyl ethanoate is an ester. Glacial acetate acidaniline 731 at room temperature at dark for 2 hours to induce the site-specific cleavage. Mobile phase acetonitrile and buffer solution.
2 Qualitative and Quantitative Composition. Results show a higher recovery for pure glacial acetic acid samples 1143 than its corresponding 50 glacial acetic acid samples 9601. In simple words 1 mole is equal to the atomic weight of the substance.
Apart from the number of double bonds present in the molecule the IV is also influenced by the molecular weight thus IV oleic acid 2 127282 100 901 IV glyceryl 6 12741 3 281 100 862 triolein similarly IV linoleic acid 181 trilinolein 1732 IV linolenic acid 2735 trilinolenin 2616. Sulfur is the tenth most common element by mass in the universe. 1 filter to remove any particulate matter and store in appropriate bottles Destaining solution.
This structure is also available as a 2d Mol file or as a computed 3d SD file The 3d structure may be viewed using Java or Javascript. Acetic Acid CH3COOH- Acetic Acid is an organic compound with formula CH3COOHVinegar is a water solution of acetic acid containing 5-8 of acetic acid by volume. During these processes detonating mixtures may be produced.
The recovery of the method was higher than other spectrophotometric methods reported previously 15 18. And as a polymerization catalyst for. 4 wv Paraformaldehyde-1 vv glutaraldehyde.
After precipitation the NaBH 4-treated RNAs were subsequently treated with aniline-acetate solution H 2 O. APO-Tranexamic Acid tablets contain the active ingredient tranexamic acid. The standard composition is 01 of dye and 5 of acetic acid Biotium Novus biologicals and Sigma-Aldrich.
For example 1 mole of acetic acid is equivalent to 6005 g of acetic acid molecular weight. Molecular weight 1X molarity Add for 500ml Add for 1L. Molecular weight of H2SO4.
LIQUID AND MIST CAUSE SEVERE BURNS TO ALL BODY TISSUE. 0874 M 5 25 ml. Hazards Identification Emergency Overview----- POISON.
Tranexamic acid is a white crystalline powder that is odourless or almost odourless. Concentration of Sulfuric acid. Addition of a small amount of water.
Pure compound is a solid below 62F. Add glacial acetic acid to pH 55. When properly mixed filter the solution through a Whatman No.
Molarity refers to the number of moles of the solute present in 1 liter of solution. 95 by mass wtwt Step 1. The difference between them is the melting point of their.
For example 1 mole of H2SO4 is equal to 9808 grams of H2SO4 molecular weight 9808. Visit BYJUS for more content. Density weight volume or Volume weight density or The.
As an oxidant for preparing epoxy compounds. It has a pungent smell and a sour taste. Calculate the volume of 100 grams of Sulfuric acid.
Where several fatty acids are present the IV. More information on the manner in which spectra in this collection were collected can be found here. Other examples of esters include ethyl propanoate propyl methanoate propyl ethanoate and methyl butanoate.
Methacrylic acid appears as a clear colorless liquid or low-melting solid with a pungent odor. Acetic acid glacial 6005. Glacial acetic acid reagent grade.
Often used in an aqueous solution. Add 90 ml methanolwater 11 vv and 10ml of Glacial acetic acid mix properly using a magnetic stirrer. QTBSBXVTEAMEQO-UHFFFAOYSA-N Copy CAS Registry Number.
MAY BE FATAL IF SWALLOWED. 571 ml Glacial Acetic Acid 1861g Na2EDTA2H2O QS to 10 liter with water - do not adjust pH but check. The recovery studies were carried out in both pure glacial acetic acid and 50 glacial acetic acid.
Sampling medium 450 µgmL a For sampling in ambient temperatures 60 F. May polymerize exothermically if heated or contaminated. As a reagent in making caprolactam and glycerol.
To Learn about the structure of Acetic acid its preparations chemical physical properties uses and FAQs. 76057 4 Na salt 13 mM 01 05 g. Acetic acid ə ˈ s iː t ɪ k systematically named ethanoic acid ˌ ɛ θ ə ˈ n oʊ ɪ k is an acidic colourless liquid and organic compound with the chemical formula CH 3 COOH also written as CH 3 CO 2 H C 2 H 4 O 2 or HC 2 H 3 O 2.
Chat now for more business. Sulfur in nontechnical British English. The density of glacial acetic acid is 1049 gml at 25C which means that the weight of the 1 ml of glacial acetic acid is 1049 grams at 25C.
Sulphur is a chemical element with the symbol S and atomic number 16. If the polymerization takes place inside a container the container may rupture violently. Contact China Manufactory Zouping TaiZi Welfare Vinegar Company for the product Food grade glacial acid acetic 997 200KG Shunxin brand.
There are several formulations available with a difference in the percentage of acetic acid and dye. Small molecules such as amino acids. Except where noted spectra from this collection were measured on dispersive instruments often in carefully selected solvents and hence may differ in detail from measurements on FTIR instruments or in other chemical environments.
Corrosive to metals and tissue. CH 3 COOH Glacial 100 175 Molar Strength 100 Density 105 Molecular Weight 6005 1 liter 1050 gm CH 3 COOH 1748 moles 175M. During the production of terephthalic acid n-xylene is oxidized in the presence of acetic acid.
Acetic acid aqueous Ethanoic acid Glacial acetic acid Methanecarboxylic acid Note. The hydrogen on the carboxyl group of acetic acid is replaced with an ethyl group. Most proteins peptides and enzymes of low molecular weight.
Vinegar is no less than 4 acetic acid by volume making acetic acid the main component of vinegar apart from water and other trace elements. 4 wv Paraformaldehyde 4 wv Paraformaldehyde-1 vv glutaraldehyde 10 Neutral-buffered formalin NBF Delicate tissue. Zenkers solution Helly.
Acetic acid or acetic anhydride can explode with nitric acid if not kept cold. Concentration information is not available for. Glycerides are fatty acid esters of glycerol.
After cleavage the RNA samples were purified by ethanol precipitation and used for cDNA library construction using NEBNext Small RNA Library Prep Set New England Biolabs. Can be found in concentrations of 5-8 in vinegar Colorless liquid or crystals with a sour vinegar-like odor. Can use stock EDTA to make buffer will need to check and adjust pH depending on stock 100 ml of 05 M EDTA at ph 80 1X40 mM Tris pH 76-80 20 mM acetic acid 1 mM EDTA 10X TBE Stock 1080 g Tris Base.
It is freely soluble in water. GAA - Glacial acetic acid GFP - Green fluorescent protein GUS - -Glucuronidase HEPA - High Efficiency Particulate Air HEPES - N-2-hydroxyethylpiperazine-N-2-ethanesulphonic acid H2O2 - Hydrogen peroxide HCl - Hydrochloric acid HPLC - High performance liquid chromatography h - Hours IR - Infra red IPTG - Isopropyl-thiogalactoside kDa - kilodaltons kbp - kilobase pairs Km - Michaelis-Menton. As a bleaching agent.
Each APO-Tranexamic Acid tablet contained 500 mg of tranexamic acid. Acetic Acid 64-19-7 995 - 100 Yes 3. Dissolve 204 g sodium acetate trihydrate in 2 L distilled-deionized water.
Weigh 025g of Coomassie Brilliant Blue R250 in a beaker. Used as a bactericide and fungicide especially in food processing. Liver spleen bone marrow.
Density of Sulfuric acid. Fats and oils are examples of esters.
