The goal of the system is to give each structure a unique and unambiguous name and to correlate each name with a unique and unambiguous structure. Ideally every possible organic compound should have a name from which an unambiguous structural.
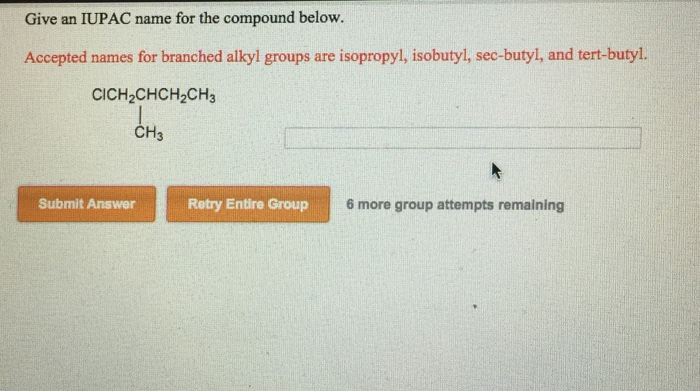
Solved Give An Iupac Name For The Compound Below Accepted Chegg Com
ESCKH In order to give compounds a name certain rules must be followed.
Give an iupac name for the compound below. OR submit details below for a call back clear. I did need to dig in the IUPAC Blue Book for quite a while to clarify the priority of functional groups but also wanted to thank James at MOC for a helping post. The formula for a substituted cycloalkane is shown below.
In chemical nomenclature the IUPAC nomenclature of organic chemistry is a method of naming organic chemical compounds as recommended by the International Union of Pure and Applied Chemistry IUPAC. In general the base part of the name reflects the number of carbons in what you have assigned to be the parent chain. The Trivial nomenclature system is limited to only a few compounds in each specific group.
In numbering the ring which carbon a-f is 1. It is published in the Nomenclature of Organic Chemistry informally called the Blue Book. These names are listed within the discussion of naming alkanes.
Systematic IUPAC name of 246-trinitrotoluene common name or TNT. Fundamental Principle IUPAC. For ketones with more than four carbon atoms it is important specify the position of ketone group.
The suffix of the name reflects the types of functional groups present on or. A single chemical compound can have several acceptable systematic names and the systematic method of naming organic chemical compounds is called IUPAC Nomenclature but no two compounds can have the same. Computed by XLogP3 30 PubChem release 20210507 Hydrogen Bond Donor Count.
How many alkyl substituents are attached to the ring. How to name organic compounds using the IUPAC rules. Property Name Property Value Reference.
Knowing these rules and given a structural formula one should be able to write a unique name for every distinct compound. Give the IUPAC name of this compound. Give the IUPAC name for this compound.
Likewise given a IUPAC name one should be able to write a structural. An example of this can be observed in the alternate names of Phenol for which names such as hydroxybenzene and carbolic acid also exist. There are however guidelines as to which naming method should be adopted.
Select the correct IUPAC name for the organic reactant. So its name in according to IUPAC is Butanone. The systematic name for the compound in Problem 21 is _____.
In numbering the ring which carbon a-f is 2. Anyway its IUPAC name is 255-trimethylhexan-3-ol. CH 3 OCH 2 CH 3.
This table should be very self-explanatory and following the strategies will give you the knowledge to name alcohols aldehydes nitriles ketones esters carboxylic acids present with different combinations in complex molecules. It has a role as a serotonergic antagonist a dopaminergic antagonist an adrenergic antagonist a histamine antagonist a. Recommend 1 Comment 0 person.
Recommend 0 Comment 0 ASK A QUESTION. 43 IUPAC naming and formulae ESCKG What is IUPAC naming. Short Summary of IUPAC Nomenclature of Organic Compounds Introduction The purpose of the IUPAC system of nomenclature is to establish an international standard of naming compounds to facilitate communication.
For this reason the common chemical name 246-trinitrotoluene or TNT as shown in figure 17 would not be advisable under the IUPAC systematic nomenclature. A few shortcomings of the trivial system for naming organic compounds are listed below. When naming organic compounds the IUPAC International Union of Pure and Applied Chemistry nomenclature naming scheme is used.
A primary amine b secondary amine c tertiary amine d primary amide e secondary amide 22. To correctly name TNT under the IUPAC system the simple benzene naming system should be used. An example of this.
Answer 1 of 2. In the IUPAC system what is the root or base name of this compound. Rules underlying IUPAC nomenclature of alkanes alkenes and alkynes are discussed below.
Several trivial names can exist for one specific compound. The compound below is classified as a _____. The concept of a preferred IUPAC name PIN a part of the revised blue book for organic compound naming has not yet been adopted for inorganic compounds.
The longest hydrocarbon chain is selected and is termed as parent chain in case of alkanes. For the following reaction draw the major organic products and select the correct IUPAC name for the organic reactant. You mean the IUPAC name of this.
By using this system it is possible to give a systematic IUPAC name to an organic compound just by looking at its structure and it is also possible to write the structure of organic compound by following the IUPAC name for that compound. Give the IUPAC name for this compound. The recommendations describe a number of different ways in which compounds can be named.
In order to name organic compounds you must first memorize a few basic names. It is a second generation antipsychotic used in the treatment of psychiatric disorders like schizophrenia. A pentyl amine b methyl-n.
Computed by PubChem 21 PubChem release 20210507 XLogP3-11. Stereoisomers are expected so draw both. The IUPAC nomenclature system is a set of logical rules devised and used by organic chemists to circumvent problems caused by arbitrary nomenclature.
When drawing hydrogen atoms on a carbon atom either include all hydrogen atoms or none on that carbon atom or your structure may. Clozapine is a benzodiazepine that is 5H-dibenzobe14diazepine substituted by a chloro group at position 8 and a 4-methylpiperazin-1-yl group at position 11. Computed by Cactvs 34818 PubChem release 20210507 Rotatable Bond.
Computed by Cactvs 34818 PubChem release 20210507 Hydrogen Bond Acceptor Count. The IUPAC system of nomenclature is a set of logical rules framed which are mainly aimed at giving an unambiguous name to an organic compound. A dimethyl ether b methoxyethane c methylethyloxide d propyl ether e none of the above 21.