This reaction requires 6 electrons and 14 hydrogen ions. Here are the rules for using this free website.

What Is The Molecular Formula And Molar Mass Of Potassium Dichromate Youtube
Ammonium sulfate NH 4 2 SO 4.

Mass of dichromate. Potassium dichromate K2Cr2O7 dissoved slightly acid solution excess oxalic acid H2C2O4 is added and the following reactiion take place. Sodium dichromate is highly corrosive and is a strong oxidizing agent. Im glad you found your way here for chemistry help because youre in the right spot to be enlightened.
115994 g mol 1 and 215988 g mol 1 Conjugate acid. 266 describe the oxidation of alcohols using acidified potassium dichromateVI with reference to formation of aldehydes and carboxylic acids from primary alcohols formation of ketones from secondary alcohols and resistance to oxidation of Unit A2 1. Compound A molecular formula C 3 H 8 O is treated with acidified potassium dichromate to form a product B molecular formula C 3 H 6 O.
Sign In For Use With. Showing 1 of 1. Mole - mass calculations Conversion Factor.
NH 4 NO 3. Reduction half equation. Find the percent composition by mass of potassium dichromate K 2 Cr 2 O 7 Step 1.
Concentrations are also often reported on a mass-to-mass mm basis or on a mass-to-volume mv basis particularly in clinical laboratories and engineering applications. Potassium dichromate K 2 Cr 2 O 7 is a common inorganic chemical reagent most commonly used as an oxidizing agent in various laboratory and industrial applications. II solution then allows you to calculate the number of moles of iron as Fe 2 in your unknown sample and the mass of Fe in g.
Finding the percent by mass means finding the mass of the elements in the compound and adding the. Material Properties - Material properties for gases fluids and solids - densities specific heats viscosities and more. If heated in a closed container the container may rupture due to the decomposition of the material.
And we use the method of half-equations. The molar mass of water is 18015 gmol and the molar mass of sulfuric acid is 98078 gmol. And ferrous ion is the species that is oxidized.
Except where otherwise noted data are given for materials in their standard state at 25 C 77 F 100 kPa. Dichromate is the oxidizing agent. The way it is handled packed and stored prior to the installation and use of the.
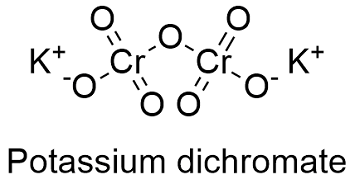
It is a crystalline ionic solid with a very bright red-orange color. As with all hexavalent chromium compounds it is acutely and chronically harmful to health. Potassium Dichromate Formula and Molecular Structure.
Molar Mass of Frequently Calculated Chemicals. Two different starting assumptions are shown Solution assuming a certain volume of solution is present. Chemical Elements Periodic Table Compound Name Formula Search Moles to Grams Calculator Common Compounds List Chemical Equation Balancer Complete List of Acids Complete List of Bases Molar to Mass Concentration Converter Molar Mass Calculator Cations Anions List Dilution.
Dichromate ion reduces to two chromiumIII ions. B forms a shining silver mirror on warming with ammoniacal silver nitrate. Dark green solution was formed and.
Further Physical and Organic Chemistry. White rust is an occurrence that happens following galvanization. Shri Ram Murti Smarak College of Engineering Technology Bareilly is a self financed institute managed and run by SRMS Trust BareillyIt was founded in 1996 and the college has demonstrated perennial evidence of merit and quality by achieving a respectable position among all parallel private and govt.
NanoDrop CF-1 Calibration Check Fluid. A concentration on an mv basis is the number of grams of solute per milliliter of solution. It can be calculated by adding the invididual molar mass of every atom that are composing the molecule CH4.
Aqueous Potassium Dichromate K2Cr207 Solution. By the way if its not actually the holidays and I havent updated this recently sorry about that. Ammonium oxalate NH 4 2 C 2 O 4.
Ammonium dichromate is a bright orange red crystalline solid. The molar mass of a substance also often called molecular mass or molecular weight although the definitions are not strictly identical but it is only sensitive in very defined areas is the weight of a defined amount of molecules of the substance a mole and is expressed in gmol. NanoDrop 8000 NanoDrop Lite NanoDrop.
The chemical formula for potassium dichromate is K 2 Cr 2 O 7 and the molar mass is calculated to be 294185 gmol. Catalog Number Specifications Unit Size Description Price USD Availability Quantity. Dichromate salts contain the dichromate anion Cr 2 O 2 7.
This compound is. The salt is popular in the laboratory because it is not. MassFe moles Fe 5585 g Femole Fe mFe5585 x total volume x molarityFeII Take into account that your total volume is 100 mL 5.
Molecular Weight Molar Mass. B when treated with an aqueous solution of N H 2 C O N H N H 2 H C l and sodium acetate gives a. Potassium oxalate K2C2O4H2O was added.
Re-passivation of a surface can be achieved by treating the surface with a solution of 01 sulfuric acid and 5 sodium dichromate then brushing with a solid wire brush for 30 seconds and finally rinsing the surface thoroughly. It is readily ignited and burns producing a voluminous green residue. I promise that I wont take your.
1 Assume that a volume of 1000 L of the solution is present. 3 mol H 2 2 mol Al 3 mol H 2 2 mol Al 60 mol Al 18 g H 2 2016 g 1 mol H 2 9 Mass - mole calculations A balanced chemical equation A known mass of one of the reactantsproduct in grams Given. How find the percent of an element in a compound.
At a temperature of 0 o C the solubility of this compound in water corresponds to 49 grams per litre. C2H52O Ether NH42C2O4 Ammonium Oxalate NH42CO3 Ammonium Carbonate NH42CrO4 Ammonium Chromate NH42HPO4 Di-Ammonium Phosphate NH42S Ammonium Sulfide NH42SO4 Ammonium Sulfate NH43PO3 Ammonium Phosphite NH43PO4 Ammonium Phosphate Ag2O SilverI Oxide Ag2S Silver Sulfide Ag2SO4 Silver. The number of atoms of each element on both sides of the equation is the same and therefore mass is conserved.
It is soluble in. SRMS College of Engineering Technology Bareilly. The sum of the positive and negative charges is the same on both sides of the equation and therefore charge is conserved.
42 Enthalpy entropy and free energy. Mole ratio between unknown substance hydrogen and known substance aluminum. It may also act as a strong oxidizing agent if mixed with or contaminated with combustible material.
Sodium Dichromate is an orange to red colored crystalline inorganic compound that emits toxic chromium fumes upon heating. NanoDrop CF-1 Calibration Check Fluid. 1 Saturated aqueous solutions of inorganic salts are convenient for calibration of instruments for measurement of relative humidity.
424 calculate the standard entropy change ΔSӨ in a. Some other important properties of potassium dichromate are listed below. A concentration expressed on an mm basis is equal to the number of grams of solute per gram of solution.
Find the molar mass of the compound. Divide the total mass of each element by the molar mass and multiply by 100 to find the mass. They are oxyanions of chromium in the 6 oxidation state and.
Charge is conserved because electrons are neither created nor destroyed in a chemical reaction. Ammonium dichromate NH 4 2 Cr 2 O 7. Given a density of 1836 gmL and a mass percent of H 2 SO 4 of 9600 find the molarity molality and mole fraction.
Sodium dichromate - Na 2 Cr 2 O 7. Infobox references Chromate salts contain the chromate anion CrO 2 4. 8H3H2C2O46K2C2O4H2OK2Cr2O7 6CO29H2O8K2K3CrC2O433H2O Vigorouly orange solution was formed and filtered then leaves for crystallization.
K 2 Cr 2 O 7 has a crystalline reddish-orange appearance. It is an ionic compound with two potassium ions K and the negatively charged dichromate ion Cr2O7- in which two hexavalent chromium atoms with oxidation state 6 are each attached to three oxygen atoms as well.
Sodium thiosulfate ____Na2S2O3_____ 3. Potassium permanganate is commercially prepared by mixing solution of potassium hydroxide and powdered manganese oxide with oxidizing agents like potassium chlorate.
Sulfur dioxide ____SO2_____ 2.
Potassium dichromate formula. Whether it is primary secondary or tertiary and on the conditions. There is a formula for calculating COD. S 2 O 3 2-thiosulfate.
Use the stock form for the transition metals. The predominance diagram shows that the position of the equilibrium depends on both pH and the analytical concentration of chromium. Crude form of sodium carbonate.
When the formula unit contains two or more of the same polyatomic ion that ion is written within parentheses and a subscript is written outside the parentheses to indicate the number of polyatomic ions. In aqueous solution chromate and dichromate anions exist in a chemical equilibrium. There are several general rules however that can bring some order out of this apparent chaos.
Many potassium salts are of utmost importance including the hydroxide nitrate carbonate chloride chlorate. Potassium dichromate primarily affects the respiratory tract causing ulcerations shortness of breath bronchitis pneumonia and asthma but can also affect the gastrointestinal tract liver kidneys and immune system. Copper sulfate crystals blue lead.
Potassium Dichromate Formula and Molecular Structure The chemical formula for potassium dichromate is K 2 Cr 2 O 7 and the molar mass is calculated to be 294185 gmol. 2 CrO 2 4 2 H Cr 2 O 2 7 H 2 O. Im glad you found your way here for chemistry help because youre in the right spot to be enlightened.
Potassium dichromate K2Cr2O7 is an oxidising agent that causes alcohols to oxidise. ZnHCO32 zinc bicarbonate 15. SnCN2 tin II cyanide.
Compounds composed of molecules are thus called molecular compounds. Acid Alkali Salt Water. This substance is a known human carcinogen and is associated with an increased risk of developing lung cancer and cancer of the.
The COD test involves a blank which is a sample made by adding the reagents of acid and an oxidizing agent to distilled water. KHCO3 potassium bicarbonate 7. 4 MOLECULAR COMPOUNDS When non-metal atoms share electrons with other non-metal atoms they often form basic units called molecules.
Potassium dichromateVI solution acidified with dilute sulphuric acid is commonly used as an oxidising agent in organic chemistry. By the way if its not actually the holidays and I havent updated this recently sorry about that. NH42CrO4 ammonium chromate 17.
Potassium dichromate VI solution and dilute. AgNO3 silver nitrate 10. Salt of Potassium Sodium and Ammonium.
It is an ionic compound with two potassium ions K and the negatively charged dichromate ion Cr2O7- in which two hexavalent chromium atoms with oxidation state 6 are each attached to three oxygen atoms as well. Name Formula Systematic Name Common Name Formula Name Formula Methane CH 4 Methanoic acid Formic acid HCO 2H 12-Dichloroethane C 2H 4Cl 2 Ethane C 2H 6 Ethanoic acid Acetic acid CH 3CO 2H Methylamine CH 3NH 2 Propane C 3H 8 Propanoic acid Propionic acid C 2H 5CO 2H Methylammonium ion CH 3NH 3 Butane C 4H 10 Butanoic acid Butyric acid C 3H 7CO. The exact reaction however depends on the type of alcohol ie.
How find the percent of. If the COD is high the amount of pollution in the test sample is high. Parentheses and a subscript are not used unless more than one of a polyatomic ion is present in the formula unit eg calcium sulfate CaSO 4 not CaSO 4.
K2Cr2O7 potassium dichromate 6. Primary alcohol aldehyde Reagent. CdOH2 cadmium hydroxide 13.
Potassium dichromate or anhydrochromate is prepared by adding to the neutral yellow chromate of potassium in solution a moderate quantity of one of the stronger acids. Hg2NO22 mercury I nitrite 16. More specifically the test is a process of decomposing pollutants in water after two hours of boiling the water in a solution of potassium dichromate.
An alloy of sodium and potassium NaK is used as a heat-transfer medium. I promise that I wont take your. The relative formula mass of a substance is the sum of the relative atomic masses of the elements present in a formula unit.
Chemical Formula Nomenclature Practice. BO 3 3-borate. This activity includes every compound formula and name that can be formed from the list 44 Ions provided in Chemistry A at Pickerington High School Central.
The name of the ion usually ends in either -ite or. Potassium is an essential constituent for plant growth and is found in most soils. 3 chromium III dichromate BaC ℓO 2 barium hypochlorite PbC ℓO 2 4 lead IV chlorite PbSO 3 2 lead IV sulfite 2.
As with all hexavalent chromium compounds it is acutely and chronically harmful to health. The salt is popular in the laboratory because it is not. We shall learn the nomenclature of only binary molecular compounds.
Preparation of Sodium Chloride NaCl HCl NaOH NaCl H2O. Acid Metal Carbonate Salt Water Carbon. Total compositions by your criteria.
Find the molar mass of the compound. Using potassium dichromateVI as an oxidising agent in organic chemistry. Potassium dichromate K 2 Cr 2 O 7 is a common inorganic chemical reagent most commonly used as an oxidizing agent in various laboratory and industrial applications.
Partial Oxidation of Primary Alcohols Reaction. FeNO22 iron II nitrite 9. The chromate ion is the predominant species in alkaline solutions but dichromate can become the predominant ion in acidic solutions.
Dichromate Cr 2 O 7 2-dihydrogen phosphate H 2 PO 4 - or H 2 O 4 P-formate CHO 2-or HCOO-or CHOO-hydrogen sulfate or bisulfate HSO 4-hydrogen sulfite or bisulfite HSO 3-hydrogen phosphate HPO 4 2-hydroxide OH-hypochlorite ClO-nitrate NO 3-nitrite NO 2-oxalate C 2 O 4 2-perchlorate ClO 4-permanganate MnO 4-peroxide O 2 2-phosphate PO 4 3. It is a crystalline ionic solid with a very bright red-orange color. At first glance the nomenclature of the polyatomic negative ions in the table above seems hopeless.
This activity includes every compound formula and name that can be formed from the list 44 Ions provided in Chemistry A at Pickerington High School Central. Copper sulfate crystals blue vitriol. BaClO42 barium perchlorate 11.
Students enrolled in Dr. Potassium dichromate K2Cr2O7 aluminum hydrogen sulfate AlHSO43 or bisulfate. You should complete this by Sunday.
Here are the rules for using this free website. HPO 4 2-hydrogen phosphate-3 ions. Divide the total mass of each element by the molar mass and multiply by 100 to find the mass.
Acid Reactive metal Salt Hydrogen Gas. Salt of non-Potassium Sodium and Ammonium. Potassium manganateVII solution has some tendency to do.
Acid Metal Oxide Salt Water. Give the formula for the following. It is a reasonably strong oxidising agent without being so powerful that it takes the whole of the organic molecule to pieces.
Write the chemical formula for the following ionic compounds. SnSO4 tin II sulfate 14. Complete these in lab and on your own time for practice.
PbCO 32 lead II carbonate 12. Draganjacs Introduction to Chemistry CHEM1003 General Chemistry I CHEM1013 and General Chemistry II CHEM1023 classes are responsible for learning the names and formulae for the common acids and common reagents and for learning the names formulae and the charges for the common cations and anions listed below. Find the percent composition by mass of potassium dichromate K 2 Cr 2 O 7 Step 1.
Pyrotechnics data for your hobby. Cu2CO3 copper I carbonate 8. Zinc carbonate ZnCO 3 aluminum hypochlorite Aℓ C ℓO 3 calcium phosphate Ca 3PO 4 2 cadmium phosphate Cd 3PO 4 2 iron III sulfate Fe 2SO 4 3 mercury II chlorite HgC ℓO 2 2 potassium.