Modern iNO delivery systems include monitoring for NO and NO2 levels. In an experiment 0625 mole of N2O4was introduced into a 500 L vessel and was allowed to decompose until equilibrium was reached.
Answered Nitrogen Monoxide And Ozone React To Bartleby
Heat the mixture at 60 o C using a constant temperature water bath set to 60-65 o C for about 15 hour.

No2 oxygen mixture. When it comes to Calibration Gas you can count on Grainger. Any less and the creature starts to suffocate. These primary emissions lead to the generation of secondary pollutants such as ozone nitrates and organic aerosol.
It includes a mixture of solid particles and liquid droplets found in the air or simply aerosols. Nitrous acid is also highly volatile in the gas phase it exists predominantly as a trans-planar moleculeIn solution it is unstable with respect to the disproportionation reaction. Calculate the volume of 36 mol nitrogen at STP.
This reaction is slow at 0 C. Sodium and potassium are stored in kerosene oil because when they come in contact with oxygen they form respective oxides and the reaction is explosive. Sodium nitrite is an inorganic sodium salt having nitrite as the counterion.
Breathing air with a high concentration of NO2 can affect the respiratory system. In the first of two steps in the industrial synthesis of sulfuric acid elemental sulfur reacts with oxygen to produce sulfur dioxide. They vary in size.
It is a nitrite salt and an inorganic sodium salt. It can be fatal if inhaled in large quantity. All humans pets and lizard-people need more than 16 kPa of oxygen in the air or internals to breathe.
Other gases belonging to this group are nitrogen monoxide or nitrous oxide N2O and nitrogen. NO 2 is an intermediate in the industrial synthesis of nitric acid millions of tons of which are produced each year for use primarily in the production of fertilizersAt higher temperatures it is a reddish-brown gas. A fuel-water emulsion creates a leaner fuel environment in the engine lowering PM emissions.
Calculate the volume of 245 mg CO2 at STP125 L. For the purposes of this calculation well assume a 2-liter flask holds 3 gases. The concentration of N2O4 at equilibrium was 00750 M.
Calculate the mass of 178 mL CO2 at STP350. Acid mixture flows downward countercurrent to ascending vapors. Our sunrise and sunset calculator displays the Sun rise and set times for the US.
However an easier and more straightforward procedure is to heat the sample at 60 o C for 15 minutes after reaching this temperature allow about 5 min for warming the sample before starting to record the time then incubate for the 15 minutes. Nitrogen dioxide NO2 when fuel burns nitrogen and oxygen react with each other and form nitrogen oxides NOx. It is required to oxidize fires.
Oxygen canisters are marked in. 3HNO 2 aq H 3 O NO 3 2NO. The concentrated acid vapor leaves the column and goes to.
What is the partial pressure of oxygen when this mixture is delivered at a total pressure of 85 atm34 atm. To detect it use your PDA or a wall mounted Air Alarm. Our first base gas is Oxygen.
Calculate the volume of 225 mol Cl2 at STP. Some particles such as dirt smoke fly ash mists condensing vapors mist soot or dust are conspicuous enough to be visible with the naked eye. Nitrogen dioxide is a chemical compound with the formula NO 2It is one of several nitrogen oxides.
TRAP is a complex mixture of PM derived from combustion including elemental or black carbon and non-combustion sources eg road dust tyre wear and brake wear and primary gaseous emissions including nitrogen oxides. Supplies and solutions for every industry plus easy ordering fast delivery and 247 customer support. Overview Health effects Environmental effects Sources of emissions References Description Oxides of nitrogen are a mixture of gases that are composed of nitrogen and oxygen.
Others are so tiny that they can only be detected with the help of an electron microscope things into perspective PM25 is. In the second step sulfur dioxide reacts with additional oxygen to form sulfur trioxide. Emulsified diesel can be used in any diesel engine but there is a decrease in.
PK a 33 at 18 C. Servomex pioneered the use of Paramagnetic sensing technology and it remains a proven trusted cornerstone of our oxygen O 2 analysis productsUsed by many of our industrial portable and multi-gas analyzers it delivers fast and reliable measurements of percentage O 2 concentrations. Calculate the equilibrium constant for the overall reaction at.
Oxygen is an invisible gas. Non metals react with oxygen to form non metal oxide Non Metal Oxygen Non Metal Oxide C O2CO2 N2O2NO2. Nitrite is the nitrogen oxoanion formed by loss of a proton from nitrous acidIt has a role as a human metabolite.
The tool will also show you the length of day when dawn will break and when darkness will descend. Nitrogen Dioxide NO2 forms from emissions from cars trucks buses power plants and off-road equipment. Calculate the volume of 148 g NO2 at STP.
It is a conjugate base of a nitrous acid. A For every mole of N2O4 reacted there will be 2 moles. Emulsified diesel fuel is a blended mixture of diesel fuel water and other additives.
Paramagnetic an innovative solution for percentage oxygen. WHATS NEW IN GRI-Mech 30 GRI-Mech 30 is an optimized mechanism designed to model natural gas combustion including NO formation and reburn chemistry. The water is suspended in droplets within the fuel creating a cooling effect in the combustion chamber that decreases NOx emissions.
Two of the most toxicologically significant compounds are nitric oxide NO and nitrogen dioxide NO2. Interestingly the efficacy of nebulized NTG given with a gas mixture that was 50 oxygen was not superior to inhalation of 100 oxygen while inhaled milrinone was slightly better than 100 oxygen. Used as a food preservative and antidote to cyanide poisoning.
Our partial pressure equation. Calculate Kc for the reaction A 75 B 0125 C 00750 D 010 E 0050. Nitrite is the conjugate base of the weak acid nitrous acid.
Going long is not bad Caution. Concentrated nitric acid leaves the top of the column as 99 percent vapor containing a small amount of NO 2 and oxygen O 2 resulting from dissociation of nitric acid. Intense experimental and epidemiological.
So in order to avoid contact with air it is stored in kerosene oil. HNO 2 H NO 2. It has a role as an antimicrobial food preservative an antihypertensive agent a food antioxidant a poison and an antidote to cyanide poisoning.
Calculate the mass of 155 mL O2. The brown gas NO2 and the colorless gas N2O4 exist in equilibrium 2NO2 N2O4. It is a nitrogen oxoanion a member of reactive nitrogen species and a monovalent inorganic anion.
Simply type in your ZIP or Postal code below. We need to find the partial pressure for each gas and the total pressure the gas mixture exerts in the container. There are 10g of each gas and the temperature of each gas in the flask is 37 degrees C 986 degrees F.
Nitrogen N 2 oxygen O 2 and carbon dioxide CO 2. The reaction for each step is shown as is the value of the corresponding equilibrium constant at 25C. It is the successor to version 211 and another step in the continuing updating evolution of the mechanism.
The specifics of each fire reaction will be detailed down below.
Sodium and potassium are stored in kerosene oil because when they come in contact with oxygen they form respective oxides and the reaction is explosive. Non metals react with oxygen to form non metal oxide Non Metal Oxygen Non Metal Oxide C O2CO2 N2O2NO2.
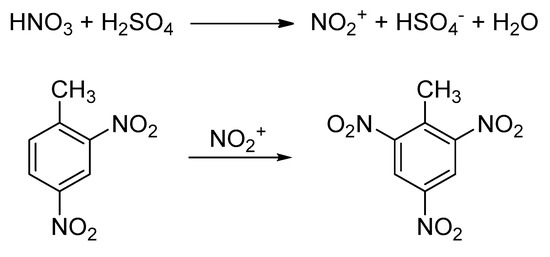
Molecules Free Full Text Synthesis Of 2 4 6 Trinitrotoluene Tnt Using Flow Chemistry Html
4C3H5N3O9l 12CO2g 10H2Og 6N2g O2g a A sample of nitroglycerine was detonated and produced 0350 g.
Is a mixture of o2 and no2 explosive. The broad scope of topics presented in this Forum illustrates NOs rich chemistry in which inorganic and bioinorganic chemists are taking the field to new and exciting levels. Academiaedu is a platform for academics to share research papers. Heating Cu2O and Cu2 S will give.
A bond angle is the angle between any two bonds that include a common atom usually measured in degrees. Mixtures are either homogeneous or. Reacts with reducing agents to generate heat and products that may be gaseous causing pressurization of closed containers.
The density of a gas mixture of O2 and N2 is 11705 gL at 27 C and 750 mmHg. It also aids with understanding the bonds formed in the molecule and the electrons not participating in any bond formation. The of N2 in the mixture is.
Oxides of nitrogen NOx for example nitrogen dioxide NO2 are also produced when air is heated in furnaces. A Cu SO2 b Cu SO3 c CuO CuS d Cu2 SO3 20. Use data from Example 13-5 to determine the partial pressure of O2 above an aqueous solution at 0 C known to contain 500 mg O2 per 1000 ml of solution.
The exact composition of the mixture depends on the temperature with higher temperature favoring conversion to NO2. Nitric oxide NO should not be confused with nitrous oxide N2O a general anaesthetic or with nitrogen dioxide NO2 which is another poisonous air pollutant. Complete Solutions Manual General Chemistry Ninth Edition.
ΔG vs T plot in the Ellinghams diagram sloped downward for the reaction. Vaporizes readily to give NO2 also an oxidizing agent. Alternatively the ending -ate is used with the oxidation number of the element given in brackets.
The nitric oxide molecule is a free radical which is relevant to understanding its high reactivity. So in order to avoid contact with air it is stored in kerosene oil. O2 occupying 100 ml underwent reaction so as to form H2O2 l and N2H2 g as the only products causing the volume to contract by 60 ml.
For example NO2 is the nitrite ion and NO3 is the nitrate ion. The volume of. Where k 34 10 5 s 1 at 25 C.
The reaction 2NO O2 2NO2 obeys the. Mixtures A mixture consists of two or more substances. Amylene C5H10 and acetic acid react to give the.
1 1 a Mg O2 MgO b 2Ag O2 Ag2 O 2 2 1 1 d CO O2 CO2 c C O2 CO 2 2 Chapter 7. Therefore samples of air collected in different cities would probably differ in composition because of differences in altitude pollution and so on. If the reaction 2HI H2 I2 is second order what graph will yield a linear plot.
If more than than one chemical is dispersed in the air as is normally the case then LeChateliers mixing rule can be applied to get the cumulative LEL for the mixture. A mixture is a combination of two or more substances in which the substances retain their distinct identities. 802 E-2 atm C.
Noncombustible but can accelerate the burning of combustible materials. O2g 2CaOs With water. DISADVANTAGES Difficult to transport as it is a gas at room temperature Forms explosive mixture with air very dangerous 66 Simple Cells A cell is a device which converts chemical energy into electrical energy and is composed of two metals of different reactivity connected by an external circuit and an electrolyte The process works due to the different reactivity of metals Consists of a.
A sample of hydrogen gas was collected over water at 21C and 685 mmHg. Some examples are air soft drinks milk and cement. It reacts with the ozone in air to form nitrogen dioxide signalled by the appearance of the reddish-brown color.
For example NO2 is the nitrateIII ion and NO3 is the nitrateV ion see p. The mixture is analyzed and is found to contain 127 mol CO2 304 mol CO and 150 mol Ar. 218 10-3 M O2 02095 atm 457 10 -4 M O2 100 atm Assess When working problems involving gaseous solutes in a solution in which the solute is at very low concentration use Henry s law.
It is observed. The equation for this decomposition is given below. Give the name of the electronic arrangement and the name for the molecular geometry for 1.
Complete Solutions Manual GENERAL CHEMISTRY NINTH EDITION EbbingGammon. Free essays homework help flashcards research papers book reports term papers history science politics. The geometry of XeF6 molecule and.
Nitroglycerine C3H5N3O9 is an explosive which on detonation decomposes rapidly to form a large number of gaseous molecules. The Forum on Renaissance in NO Chemistry presents 14 contributions of which a few focus on NO chemistry itself and the remaining papers focus on the interaction of metal complexes with NO. A mixture of three gases has a total pressure of 1380 mmHg at 298 K.
An equilibrium mixture of the three gases has a partial pressure of 0095 atm and 0171 atm for NO2 and Cl2 respectively. Calculate the heat of reaction for the combustion of a mole of CH6N2. In a separate experiment the heat capacity of the calorimeter is measure to be 7794 kJC.
When writing formulae of ionic. These gases dissolve in rainwater to produce acid rain Figure 18 overleaf. What is the partial pressure of NOCl in the mixture.
2 CH6N2 l 5 O2 g -- 2 N2 g 2 CO2 g 6 H2O l When 400 g of methylhydrazine is combusted in a bomb calorimeter the temperature of the calorimeter increases from 2500 to 3950 C. The lower explosive limit LEL is the minimal amount of concentration needed of a particular chemical to cause an explosion. Hydrogen Chloride HCl Hydrogen Cyanide HCN Nitric Oxide NO Nitrogen Dioxide NO2 Phosphine PH3 Sulfur Dioxide SO2 or Photo Ionization Detectors PIDs used to detect volatile organic compounds VOCs including Benzene C6H6.
The decomposition of N2O5g to NO2g and O2g obeys first-order kinetics. 1995 - 4 Marks which is 55. Mixture may be explosive if combustible material is very finely divided.
Academiaedu is a platform for academics to share research papers. Contact your service representative immediately if you suspect that the MX6 monitor is working abnormally. What is the initial rate of reaction at 25 C where N2O50 50 10 2 M.
Mg 24 S 32 Q23 A mixture of H2 N2. The composition of the equilibrium mixture attained at 1200C is determined by measuring the rate of effusion through a pinhole. The LEL is determined empirically for each pure chemical and air mixture at a given temperature.
The mixture reacts completely with 130 g of O2 to produce CO2 and H2O. Assuming ideal gas behaviour calculate the mole fractions of C2H4 and C2H6 in the mixture. Mixtures do not have constant composition.
Ultimate BOD BODu by BOD Bottle Method Concentrations of NO3-N and NO2-N of the incubated sample are measured on day 0 and on the last day of incubation if interested in nitrification rates then measuring at regular intervals is needed - oxygen equivalency of nitrification is computed and subtracted from the exerted oxygen demand Oxygen equivalency of nitrification of NH3N to NO3. Assuming the form of the rate law is. What is the partial pressure of Ar.