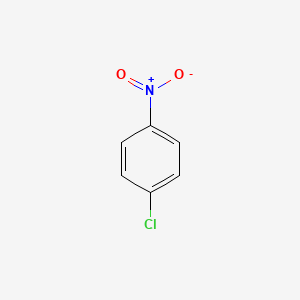
Para Nitro Aniline. Sulphuric Acid Group Product.
1 Chloro 4 Nitrobenzene No2c6h4cl Pubchem
This category is used most commonly for agents mixtures and exposure circumstances for which the evidence of carcinogenicity is inadequate in.
Para chloro nitro benzene. Tetrabutyltins which have the molecular formula C 4 H 9 4 Sn. Stock Journal of the American Chemical Society 1957 79 19 5175-5179 DOI. The main target users are workers and those responsible for occupational safety and health.
P-Nitroanilina Polish PNA VAN Azoamine Red 2H. Rates and Isomer Distributions in the Chlorination of Benzene Toluene and t-Butylbenzene in Aqueous Acetic Acid Solvents. J high resolut chromatogr chromatogr commun 5 6.
So a given substituted phenyl compound has three isomers ortho 12-disubstitution meta 13-disubstitution and para 14-disubstitution. With or without substitution of the benzene ring of the anilide moiety for an aromatic heterocycle. A disubstituted phenyl compound trisubstituted benzene may be for example 135-trisubstituted or 123-trisubstituted.
Presence of a nitro group at ortho or para position increases the reactivity of haloarenes towards nucleophilic substitution. The Influence of Solvent on. Cette étape est létape cinétiquement déterminante de la réaction.
24 Di Nitro Chloro Benzene. To explain this a third mechanism for nucleophilic substitution has been proposed. 2-Hydroxy-5-Nitro Benzaldehyde 2 lacs ADD TO CART.
Higher degrees of substitution of which the pentafluorophenyl group is an example exist and are named according to IUPAC. Brown and Leon M. Rates of Chlorination of Benzene Toluene and the Xylenes.
The primary aim of the cards is to promote the safe use of chemicals in the workplace. Overall evaluation upgraded to Group 1 based on mechanistic and other relevant data 1 99 100F 2012 000214-17-5 Benzobchrysene 3 92 2010 000196-78-1 Benzog. 13- and 14- respectively.
13- separated by one carbon in a benzene ring para- p. The cards are data sheets intended to provide essential safety and health information on chemicals in a clear and concise way. II With or without substitution at the 3-propanone position with an alkyl.
Samples incl vent emissions from a fungicide manufacturing process and contaminated soil and water samples. Before a vowel par-. IC50 of 23 nM.
Nitro group being an electron withdrawing group decreases the electron density over the benzene ring. The California Office of Environmental Health Hazard Assessment is establishing this website to provide the public with information on chemicals products and locations often associated with Proposition 65 warnings. Raisonnons sur le 1-chloro-2-nitrobenzène en tant que substrat.
This two-step mechanism is characterized by initial addition of the nucleophile hydroxide ion or water to the. When chloro and nitro groups are attached to the benzene ring we first locate the chloro group then nitro groups. Finally the benzoic ester gave predominantly the meta-nitro product 73 accompanied by the ortho 22 and para 5 isomers as shown by the relative rates.
If only inhalation and oral exposure occurs the benzene MADL is exceeded when. In the case of multiple substituted aromatic compounds sometimes terms like ortho o meta m and para p are also used as prefixes to indicate the relative positions 12-. Ethanol 2-chloro- phosphate 31 which has the molecular formula C 6 H 12 Cl 3 O 4 P.
Mass spectrometry to analysis of 28 cmpd including a number of chloro- and nitro-substituted anilines in environmental samples are presented. 25 Di Chloro Nitro Benzene. Hunt gt hoyt mp.
The ICSC project is a common undertaking between the World Health Organization WHO and. 34 Di Chloro Nitro Benzene. Nitro group donates electrons to stabilize the intermediate ion formed by the.
000092-87-5 Benzidine 1 29 Sup 7 99 100F 2012 Benzidine dyes metabolized to NB. Soluble in ethanol. Last time we covered a comparatively simple reaction.
The agent mixture or exposure circumstance is not classifiable as to its carcinogenicity to humans. 14- across from each other in a benzene ring Using the same example above in figure 9a 13-dichlorobenzene we can use the ortho- meta- para- nomenclature to transform the chemical name into m-dichlorobenzene as shown in the figure below. Welcome to the Proposition 65 Warnings Website.
Para Nitro Chloro Benzene. La relative stabilité de cet intermédiaire est due à la présence du groupe nitro attracteur -I -M. Chlorobenzene has a longer C-Cl bond due to resonance.
Although chlorobenzene is much less reactive than benzene the rate of ortho and para-substitution greatly exceeds that of meta-substitution giving a product mixture of 30 ortho and 70 para-nitrochlorobenzene. Oral dose 24 μgday inhalation dose 49 μgday 10. Benzene chloromethyl- which has the molecular formula C 7 H 7 Cl.
Slightly soluble in benzene. Presence of nitro group at ortho or para position makes nucleophilic substitution in chlorobenzene easier. How Many Monochlorination Isomers Are Formed From Free-Radical Chlorination Of Alkanes.
B Levels for male children and adolescents were calculated by application of the default bodyweights specified in Section 25703a8 to the procedure specified in Sections 25801 and 25803 Title 27 California Code of Regulations. 3-Amino-1-Adamantanol or Vildagliptin Intermediate 45 lacs ADD TO CART. Chloro-NNEI N-Naphthalen-1-yl 1.
Stearoyl-CoA Desaturase 1 Inhibitor MF-438 CAS 921605-87-0 is a cell-permeable inhibitor of Stearoyl-CoA Desaturase 1 SCD1. These warnings inform Californians about their exposures to chemicals that cause cancer birth defects or other reproductive harm. 34 Di Chloro Aniline.
000071-43-2 Benzene 1 29 Sup 7. A combining form used in the names of benzene derivatives in which the substituting group occupies the para position in the benzene ring. Propane 2-nitro- which has the molecular formula C 3 H 7 NO 2.
Fused alkylenedioxy alkoxy haloalkyl hydroxyl nitro fused furan fused benzofuran fused dihydrofuran fused tetrahydropyran fused alkyl ring or halide substituents. CI Azoic Diazo Component 37. 291 1982 hazardous.
The presence of electron-withdrawing groups such as nitro ortho and para to the chlorine substantially enhance the rate of substitution as shown in the set of equations presented on the left below. Chlorobenzene is less polar than chloro methane. OEHHA chemical database meta data Export database as CSV file If you are having trouble with the download and would like a copy of the database just drop me LaurieMonserratoehhacagov a note and I will provide you a csv file.
Partial Rate Factors for the Chlorination Reaction Herbert C. 000060-09-3 para-Aminoazobenzene 2B 8 Sup 7 1987 000097-56-3 ortho-Aminoazotoluene 2B 8 Sup 7. Free-radical chlorination of methane to CH 4 to give chloromethane CH 3 Cl and saw that the reaction proceeds through three stages initiation where free radicals are created propagation the main product-forming step of the chain.
100F 120 In prep. Vildagliptin Intermediate 128 lacs ADD TO CART. Oleum 65.
Tributyltins which contain the grouping C 4 H 9 3 Sn. Addition du nucléophile et formation dun complexe intermédiaire de type Meisenheimer. Toxicity criteria on chemicals evaluated by OEHHA.
Para Amino Phenol 31 lacs ADD TO CART. Substances mixtures and exposure circumstances in this list have been classified by the International Agency for Research on Cancer IARC as group 3. We are a leading supplier to the global Life Science industry with solutions and services for research biotechnology development and production and pharmaceutical drug therapy development and production.
A Assertion and reason both are correct and reason is correct explanation of.
Question from Student Questionschemistry. Vapour pressures of benzene hexafluorobenzene and naphthalene The Journal of Chemical Thermodynamics 1990 22 6.
Vapor Pressure Of Benzene From Dortmund Data Bank
Temperature K A B C Reference Comment.

Vapour pressure benzene. The solubility of naphthalene is closest to ideal for solvents that are similar to it eg. Benzol Phenyl hydride Benzene and mixtures having 10 benzene or more IBC code Name of the supplier. Solution A has vapour pressure of 7 5 4.
Phenylformic acid E210 Sodium benzoate E211 3113. 5 mm of Hg at the normal boiling point of water and solution B has the same vapour pressure at the normal boiling point of benzene. 65-85-0 200-618-2 Sodium benzoate.
However it has threetarget organs central nervous system kidney and hematopoietic system. Vapour Pressure of a Solute above a Solution Henrys Law. Heating the molecules of the liquid can help change them to the vapour phase and thus increase the vapour pressure of the liquid.
Vapour cartridge or canister may be permissible under certain circumstances where airborne concentrations are expected to exceed exposure limits. Temperature and Pressure. If the liquid is heated a little over 100 C the transition from liquid to gas will occur not only at the surface but.
So theres 566 mols of molecules for every 1L of solution one comes from glucose and 556 from water as calculated. The apparent enthalpy of fusion is different and the plot may become curved. The ebulliometric method of vapour-pressure measurement.
It has 6 carbon atoms joined in a ring and has 1 hydrogen atom attached to each of the carbon atoms. Q-Suggest two materials other than hydrogen that can be used as fuels in fuel cells. Lead is also a specific organ toxin.
Vapour may accumulate in hazardous amounts in low-lying areas especially inside confined spaces resulting in a toxicity hazard. Ammonia - Vapour Pressure at gas-liquid equilibrium. P A P A 0 x A and P B P 0 B x B as the total vapour pressure P A 0 x A P 0 B x B is greater than what it should be according to Raoults Law.
Identification SDS Record Number. Protection provided by air-purifying respirators is limited. Trade names and abbreviations 3114.
The pressure lowering of the water is PX as P stands for the pressure of pure solvent and X is the molar fraction of the solute. 1L of water has 1000g of water so there are 100018 mols of water 556 mols. Of solutions say vapour pressure with the concentration of the solution and quite useful in describing the calculations involving gas mixtures.
PCS 95004 Date of SDS. The concentration of a vapor in contact with its liquid especially at equilibrium is often expressed in terms of vapor pressure which will be a partial pressure a part of the total gas pressure if any other gases. At 100 C and atmospheric pressure equilibrium is not reached until the air is 100 water.
Online calculator with figures and tables showing density and specific weight of ammonia for temperatures ranging -50 to 425 C -50 to 800 F at atmospheric and higher pressure - Imperial. There are two effects when the difference between solute and solvent increases. It may be chemical physical or biological in.
CAS EINECS ELINECS number CAS EINECS Benzoic acid. The vapour pressure of the liquid increases with an increase in its temperature. Its quite an experience hearing the sound of your voice carrying out to a over 100 first year.
While naming the substituted benzene compounds we prefix the name of the substituent to the word benzene. 50 g of urea NH 2 CONH 2 is dissolved in 850 g of water. Structural formula Benzoic acid Sodium benzoate.
Benzene Product Description. Benzoic acid Benzene carboxylic acid. Glucose or sucrose are soluble in water but cyclohexane or benzene simple six membered ring compounds are insoluble in water.
The former results from. New Movies to Watch with Your Family this Thanksgiving. Temperature and Pressure - Online calculator figures and table showing density and specific weight of benzene C 6 H 6 at temperatures ranging from 5 to 325 C 42 to 620 F at atmospheric and higher pressure - Imperial and SI Units.
Figures and table showing ammonia saturation pressure at boiling point SI and Imperial units. 5 Toxicology Toxic agent or substance Toxic agent is anything that can produce an adverse biological effect. Common VOCs include ethanol formaldehyde benzene toluene and xylene.
Q-Vapour pressure of pure water at 298 K is 238 mm Hg. This percentage increases as the temperature goes up. What is the mole ratio of benzenepB150 torr and toluenepT50 torrin vapour phase if the given solution has a vapour pressure of 120 torr.
A solution of liquid toluene dissolved in liquid benzene has a benzene mole fraction of 850. Assume that we have 100 g of solution one can start with any amount of solution because the results obtained will be the same. Outdoor VOCs are usually emitted from industrial facilities and vehicles and.
Benzene is a hydrocarbon with the chemical formula C 6 H 6. Calculate the vapor pressure of the solution given that the vapor pressures of pure benzene and toulene are 183 mmHg and 592 mmHg respectively. 11 February 2015 Identity of the substance.
For example Acetone and benzene have higher vapour pressure than water at a particular temperature. Coefficents calculated by NIST from authors data. Benzene is a specific organ toxin in that it is primarily toxic to the blood-forming tissues.
In thermodynamics and chemical engineering the vaporliquid equilibrium VLE describes the distribution of a chemical species between the vapor phase and a liquid phase. VOCs are of concern to both indoor air quality and outdoor quality. Ammonia Gas - Density vs.
Chemistry 38 Calculate the mole fraction of ethylene glycol C 2 H 6 O 2 in a solution containing 20 of C 2H6O2 by mass. In this article we. Assuming X undergoes partial dimerization in benzene what is the percentage of X dimerized in benzene solution.
These liquids will show positive deviation when Raoults Law when. Petrochemical Corporation of Singapore. Use a positive-pressure air-supplied respirator if there is any potential for uncontrolled.
By replacing one or more of the hydrogen atoms with some functional group we get several benzene compounds. Liquid can accumulate static charge by flow splashing or agitation. The slope of the plot changes ie.
Take A Sneak Peak At The Movies Coming Out This Week 812 New Movie Releases This Weekend. Benzene SDS Page 1 of 11 SAFETY DATA SHEET SDS BENZENE 1. Besley and Bottomley 1974.
Closed containers may rupture. Benzene - Density and Specific Weight vs. So the solute molar fraction is 1566 176810-2.
Vapour may travel a considerable distance to a source of ignition and flash back to a leak or open container. Let the vapour pressure pure vapour pressure and mole fraction of component A be P A P A 0 and x A respectively and that of component B be P B P B 0 and x B respectively. At room temperature and pressure the water jar reaches equilibrium when the air over the water has a humidity of about 3.
Indoor VOCs are usually emitted from consumer products and building materials such as paints and carpets and may adversely impact the health of people that are exposed.
Alkyl hypochlorites are violently explosive. The Km of CYP2E1 for alcohol is 10 mM 10-fold higher than the Km of ADH for ethanol but still within the range of alcohol concentrations seen in.

New Thiosemicarbazide And Dithiocarbazate Based Oxidovanadium Iv And Dioxidovanadium V Complexes Reactivity And Catalytic Potential New Journal Of Chemistry Rsc Publishing Doi 10 1039 C9nj01486a
Ethanol intended for industrial use is usually denatured rendered unfit to drink typically with methanol benzene or kerosene.
Ethyl benzene alcohol. Most benzene exposure comes from the air from a number of sources including forest fires auto exhaust and gasoline from fueling stations. Ethanol also called ethyl alcohol grain alcohol or alcohol. Prediction of safe industrial exposure levels and correlation of their thermodynamic properties.
Sensory irritation pulmonary irritation and respiratory stimulation by airborne benzene and alkylbenzenes. The main target users are workers and those responsible for occupational safety and health. Ethanol is toxic affecting the central nervous system.
Stearoyl-CoA Desaturase 1 Inhibitor MF-438 CAS 921605-87-0 is a cell-permeable inhibitor of Stearoyl-CoA Desaturase 1 SCD1. The electrophilic carbonyl carbon atom bonds to the nucleophilic oxygen atom of ethyl alcohol to give an intermediate in square brackets that eliminates HCl yielding the ester ethyl acetate. A acetamide b formyl acetamide c dimethyl acetate d NN-dimethylformamide e dimethylamine 28.
Ethanol is a clear colourless liquid with a characteristic pleasant odour and burning taste. Ethanol is a primary alcohol that is ethane in which one of the hydrogens is substituted by a hydroxy group. PEA Phenyl Ethyl Alcohol.
Mixtures of ethyl alcohol with concentrated hydrogen peroxide form powerful explosives. Its main use as a solvent is in. Pure ethanol is a colourless flammable liquid boiling point 785 C 1733 F with an agreeable ethereal odour and a burning taste.
Ethyl acetate is the most common ester in wine being the product of the most common volatile organic acid acetic acid and the ethyl alcohol generated during the fermentation. The primary aim of the cards is to promote the safe use of chemicals in the workplace. R U peak area of methanol from Sample solution A IDENTIFICATION r S peak area of methanol from.
May 1994 Immediately Dangerous to Life or Health Concentrations IDLH CAS number. Sensitivity varies with most people having a perception threshold around 120 mg. The compound illustrated below is called _____.
As mentioned previously the phenyl group Ph-R C 6 H 5-R can be formed by removing a hydrogen from benzene and attaching a substituent to where the hydrogen was removedTo this phenomenon we can name compounds formed this way by applying this rule. The compound given below is called _____. We are a leading supplier to the global Life Science industry with solutions and services for research biotechnology development and production and pharmaceutical drug therapy development and production.
The cards are data sheets intended to provide essential safety and health information on chemicals in a clear and concise way. Dard solution A Standard solution B Standard solution C and Standard solution D DEFINITION Methanol calculation Alcohol contains NL T 923 and NMT 938 by weight corresponding to NLT 949 and NMT 960 by vol- Result r Ur S ume at 1556 of C 2H 5OH. The racemic product is prepared by boiling for 24 hours optically inactive lactic acid with ethyl alcohol in carbon tetrachloride.
Benzene is also used as a solvent in the chemical and pharmaceutical industries. Ethanol is considered a volatile organic compound by the National. Ethanol is a versatile solvent miscible with water and with many organic solvents including acetic acid acetone benzene carbon tetrachloride chloroform diethyl ether ethylene glycol glycerol nitromethane pyridine and toluene.
Benzene however is an extraordinary. They are readily obtained by reacting hypochlorous acid and alcohols either in. Toxicol Appl Pharmacol 26853.
Methyl Ethyl Ketone removed from the list of hazardous air pollutants in December 2005 - Federal Register - December 19 2005 70 FR 75047. Note that ethyl alcohol may be used instead of isopropyl alcohol but no other types of alcohol including methanol and 1-propanol are acceptable in hand sanitizer because they can. 12-Ethanediol HOCH 2 CH 2 OH.
Its low cost versatility and general acceptability on odor its excellent stability are factors speaking strongly in favor of this otherwise relatively weak odorant. Phenyl substituentFor example a chlorine attached in this manner would be named phenyl chloride and a. Some of the significant properties of CYP2E1 are listed in LIST 4 5558.
A clear physiological function for CYP2E1 has not been identified. The l-form is prepared in similar fashion starting from l-lactic acid. In all of these examples the reactivity of the electrophiles and nucleophiles is sufficient to allow reaction to proceed spontaneously at room temperature.
Mixtures of hydrogen peroxide and 1-phenyl-2-methyl propyl alcohol tend to explode if acidified with 70 sulfuric acid Chem. Besides ethanol CYP2E1 can oxidize many other compounds including acetone benzene and other alcohols. Benzene is primarily used as a feedstock or raw material to make other industrial chemicals such as ethylbenzene cumene and cyclohexane.
It is highly flammable. Ethylene glycol monobutyl ether EGBE 2-Butoxyethanol - removed from the list of hazardous air pollutants glycol ethers category in November 2004 - Federal Register - November 29 2004 69 FR 69320. Ethanol also called ethyl alcohol grain alcohol drinking alcohol or simply alcohol is an organic chemical compound.
IC50 of 23 nM. This material enters perfume compositions at the rate of 5-10-20 or sometimes much more. Denatured alcohol is ethyl alcohol with toxic or bad tasting.
D-Ethyl lactate is obtained from d-lactic acid by azeotropic distillation with ethyl alcohol or benzene in the presence of concentrated H2SO4. A butyl acetate b ethyl pentanoate c propyl pentanoate d ethyl butanoate e butyl ethanoate 27. Ethyl acetate Acetic acid ethyl ester CH 3 COOCH 2 CH 3 Calss 3.
Ethy el ther. The aroma of ethyl acetate is most vivid in younger wines and contributes towards the general perception of fruitiness in the wine. Chemical Compatibility of the Tubing Materials Name Tygon LFL Tygon ST R-3603 PharMed Tygon HC F-4040-A Tygon MH 2075 Tygon SI Silicone Platinum Silicone.
Ethanol is used to dissolve other chemical substances and mixes readily with water and many organic liquids. Benzene in cigarette smoke is a. Floral-rose fresh rosewater honey Arctander writes fairly extensively about it.
The ICSC project is a common undertaking between the World Health Organization WHO and. It has a role as an antiseptic drug a polar solvent a neurotoxin a central nervous system depressant a teratogenic agent a NMDA receptor antagonist a protein kinase C agonist a disinfectant a human metabolite a Saccharomyces cerevisiae metabolite an Escherichia coli metabolite. Denatured alcohol and isopropyl alcohol have similar uses but they differ chemically.
We would like to show you a description here but the site wont allow us. Because it contains only carbon and hydrogen atoms benzene is classed as a hydrocarbon.
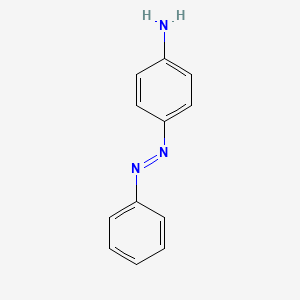
4 Aminoazobenzene C12h11n3 Pubchem
1 Aquatic Chronic 2 H318 H317 H411 016-073-00-7.
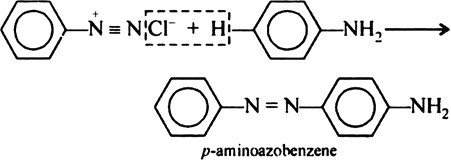
Para amino azo benzene. The Km of CYP2E1 for alcohol is 10 mM 10-fold higher than the Km of ADH for ethanol but still within the range of alcohol concentrations seen in. It is a conjugate acid of a 4-aminobenzoate. There are also no generic terms eg carbohydrate or mixtures of no fixed composition eg naphtha gasoline.
Arene diazonium salts such as benzene diazonium salts react with phenol or aromatic amines to form coloured azo compounds. A clear physiological function for CYP2E1 has not been identified. The MATERIALS database contains chemical physical visual and analytical information on over 10000 historic and contemporary materials used in the production and conservation of artistic architectural archaeological and anthropological materials.
16 03 05 RMB21. The reaction of joining two aromatic rings through the N N bond is known as coupling reaction. When exposed to light aminobenzoic acid.
It consists of a benzene ring substituted with amino. It has a role as an Escherichia coli metabolite a plant metabolite and an allergen. This makes ammonia a base and.
Benzene is a natural constituent of crude oil and is one of the elementary petrochemicals. Enter the email address you signed up with and well email you a reset link. Mixed Low Hazard Products.
You will recall that the nitrogen atom of ammonia is sp 3 hybridized and there is an unshared pair of electrons in the fourth tetrahedral orbital. Acta 1994 77 2035-2050. It derives from a benzoic acid.
Academiaedu is a platform for academics to share research papers. Due to the cyclic. Sulfamethoxazole is an isoxazole 12-oxazole compound having a methyl substituent at the 5-position and a 4-aminobenzenesulfonamido group at the 3-positionIt has a role as an antibacterial agent an antiinfective agent an epitope an EC 25115 dihydropteroate synthase inhibitor an antimicrobial agent a P450 inhibitor an EC 111153 sepiapterin reductase L-erythro-78.
The benzene molecule is composed of six carbon atoms joined in a planar ring with one hydrogen atom attached to each. IC50 of 23 nM. Non Hazardous Products including Dyes Cosmetic Bulking Powders Silicone Waste Vitamin E Allatoin and Resins.
La ECHA organiza consultas para conocer las reacciones de todas las partes interesadas y recopilar la más amplia gama posible de informaciones científicas de cara a los procesos de regulación. Besides ethanol CYP2E1 can oxidize many other compounds including acetone benzene and other alcohols. Trisodium 3-amino-613-dichloro-10-3-4-chloro-6-2-sulfophenylamino-135-triazin-2-ylaminopropyl amino-411-triphenoxydioxazinedisulfonate 410-130-3 136248-03-8 016-072-00-1 3-amino-4-hydroxy-N-2-methoxyethyl-benzenesulfonamide 411-520-6 112195-27-4 Eye Dam.
For Australia the EJ20G engine powered the GCGM Impreza WRX from 1994 to 1996. Mixed Non Hazardous Products. Functional ingredients are substances that are not used to provide odor or malodor coverage but which are essential for the functionality or durability of a fragrance compound such as an antioxidant preservative.
H 2 N-CH 2 CH 2 CH 2 OH is therefore 3-amino1-butanol. These properties may have significant implications for their use in homogeneous catalysis. The CH 3 2 N- substituent is eg NN-dimethylamino.
A necessity for chemical engg studs. PABA is a white solid although commercial samples can appear gray. This page aims to list well-known organic compounds including organometallic compounds to stimulate the creation of Wikipedia articlesNote that purely inorganic compounds minerals and chemical elements are not included on this list.
It can be observed that the para positions of phenol aniline are coupled with the diazonium salt. It is slightly soluble in water. The cards are data sheets intended to provide essential safety and health information on chemicals in a clear and concise way.
Iv Coupling reaction. Mixtures of phenols. 4-aminobenzoic acid is an aminobenzoic acid in which the amino group is para to the carboxy group.
Azocarboxamides a special class of azo ligands display intriguing electronic properties due to their versatile binding modes and coordination flexibility. What types of ingredients are on the List. Proyecto de recomendación de inclusión en la Lista de Autorización y consulta.
The main target users are workers and those responsible for occupational safety and health. Benzene is an organic chemical compound with the molecular formula C 6 H 6. Synthesis of Tri- Penta- and Heptapeptides Containing an R-2-Alkyl-2-amino- 3-methyl aminopropionic Acid Residue in the Central Position D.
Key features of the EJ20G engine included its. The ICSC project is a common undertaking between the World Health Organization WHO and. Therefore the hydroxyl group of a phenol is considered to be activating ie its presence causes the aromatic ring to be more reactive than benzene and ortho-or para-directing.
We are a leading supplier to the global Life Science industry with solutions and services for research biotechnology development and production and pharmaceutical drug therapy development and production. Different coordination motifs of. Fragrance ingredients are basic substances used for odor or malodor coverage.
Subarus EJ20G was a turbocharged 20-litre horizontally-opposed or boxer four-cylinder petrol engine. This stabilization is most effective for attack at the ortho or para position of the ring. The substituent name of the NH 2 is amino.
In this reaction arene diazonium salt reacts with aromatic amino compound in acidic medium or a phenol in alkaline medium to form brightly coloured azo compounds. 4-Aminobenzoic acid also known as para-aminobenzoic acid or PABA because the two functional groups are attached to the benzene ring across from one another in the para position is an organic compound with the formula H 2 NC 6 H 4 CO 2 H. Aminobenzoic Acid is an organic acid with UV absorption and antifibrotic properties.
Other substituted phenols are used in the dye industry to make intensely coloured azo dyes. Some of the significant properties of CYP2E1 are listed in LIST 4 5558. 宝塚の広告企画会社クルーズが年に4回発行している地域コミュニティ情報誌ComiPaコミパ 宝塚市のグルメやお稽古街の素敵な情報を発信 情報提供してくださる方バナー広告主様も募集中です.
If para position is blocked it occurs at ortho position and if both ortho and para positions are occupied than no. The reaction generally takes place at para position to the hydroxy or amino group. Die-cast aluminium block and cylinder head.
The name of the compound. The primary aim of the cards is to promote the safe use of chemicals in the workplace. There are two main types of ingredient on the List.
Stearoyl-CoA Desaturase 1 Inhibitor MF-438 CAS 921605-87-0 is a cell-permeable inhibitor of Stearoyl-CoA Desaturase 1 SCD1. In the present report half-sandwich IrCp complexes of two different azocarboxamide ligands are presented. REACTIONS OF AMINES.
Identificación de sustancias extremadamente preocupantes.
Panoli Intermediates - Manufacturers and exporters of dye intermediate chemicals manufacturer industrial chemicals manufacturers chemicals supplier industrial dye intermediate supplier of chemicals chemical exporter chemicals suppliers industrial chemicals dye intermediate manufacturer indian industrial chemicals industrial chemicals industrial chemicals and dye integrated. To learn about other processes for the commercial.
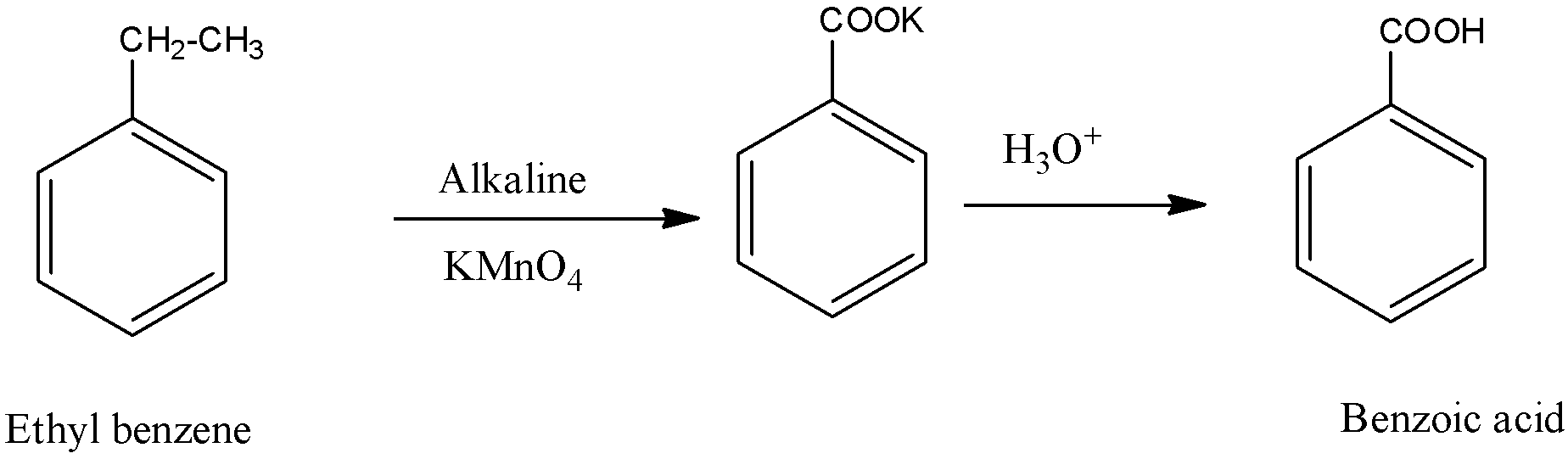
How Is Benzene Prepared From Benzenesulfonic Acid Quora
Palladium 5 On Activated Charcoal LR 10GM.

From benzene sulphonic acid. Acid - definition of acid by The Free Dictionary. High quality detergent and shampoo use Linear Alkyl Benzene Sulphonic Acid labsa 96 General Information Name. PM 10500 1 October 2020 Sodium Thiosulphate01N Hydrochloric Acid Ammonium Chloride Starch indicator ii.
The absorption of weakly acidic drugs was significantly decreased while that of bases and strong sulphonic acid increased. O-ring compatibility chart offers guidance on chemical resistance and swell for Viton o-rings FFKM o-rings Silicone o-rings Teflon o-rings and more. R-C6H4-SO3HRC12 Molecular weight323 UN NO.
Poly Aluminium Chloride PAC 28 30 30. O-ring compatibility plays a critical role in o-ring performance â find the right material for your application and avoid o-ring failures. Weakly acidic cation exchanger---carboxylic acid groups attached to acrylic and divinyl benzene co-polymer Strongly basic anion exchanger.
Toluene-4-sulfonic acid is an arenesulfonic acid that is benzenesulfonic acid in which the hydrogen at position 4 is replaced by a methyl group. Sodium Dichloroisocyanurate SDIC 60. Respiratory sensitising properties Article 57f - human health ED612018.
Paragraph beyond past by paradox. 1 Octane Sulphonic Acid Sodium Salt Monohydrate HPLC 25GM. Aluminium Sulphate 158 17.
Classification of ion exchange resins Strongly acidic cation exchanger ---sulphonic acid groups attached to styrene and di vinyl benzene copolymer. 9 Characteristics brown adhesive liquid organic weak acid soluble in water produce heat when diluted with water Usage. By extension designating objects or activities auxiliary to or derivative of that denoted by the base word parody.
R-C6H4-SO3HRC12 Molecular weight323 UN NO. Benzene can be prepared from sulphonic acids through their hydrolysis. It is a conjugate acid of a toluene-4-sulfonate.
The hydroxyl groups in alcohols can form H. Linear alkylbenzenes sometimes also known as LABs are a family of organic compounds with the formula C 6 H 5 C n H 2n1Typically n lies between 10 and 16 although generally supplied as a tighter cut such as C 12-C 15 C 12-C 13 and C 10-C 13 for detergent use. Benzene groups are modified to produce cation exchange resin and anion exchange resin 10.
Benzene-124-tricarboxylic acid 12 anhydride. At Union Chemicals Factory we are proud of many things to state a few Our superior products and top-notch. The major decarboxylation stage in the conversion of glucose to carbon dioxide takes place in the citric acid cycle and the pentose phosphate pathway.
Lower alcohols are colourless liquids members from C 5 C 11 are oily liquids and higher members are waxy solids. Paronomasia and hence abnormal or defective paranoiaAs an English prefix para-1 is also productive in. Benzene Sulphonic acid.
Many carboxylic acids do not lose CO 2 in this manner because it forms an unstable carbanion. As anaesthesia solvent coolant in the production of alcohol etc. Ittehad Chemicals Limited ICL is the pioneer and one of the largest manufacturers of industrial chemicals in Pakistan.
Linear Alkyl Benzene Sulphonic Acid LABSA 96. In this process Benzene sulphonic acid is exposed to superheated steam leading to the formation of benzene. Linear Alkyl Benzene Sulphonic Acid Sodium Lauryl Ether Sulphate and Sodium Lauryl Sulphate.
1 Octane Sulphonic Acid Sodium Salt Anhydrous HPLC 25GM. 375-73-5 magnesium perfluorobutanesulfonate EC No. A sulfonic acid or sulphonic acid refers to a member of the class of organosulfur compounds with the general formula RSO 2 OH where R is an organic alkyl or aryl group and the SO 2 OH group a sulfonyl hydroxide.
1 Octane Sulphonic Acid Sodium Salt Monohydrate. Trichloroisocyanuric Acid TCCA 90 PowderGranularTablet. But when a carbonyl exists.
No iv of Table 2 i Annex F of IS 13428 Filter paper and filtration assembly Hot plategas burner Ammonium Dichromate Ammonium Acetate Ammonium Hydroxide Potassium Iodide. Polyfluor Plastics bv Minervum 7006 NL-4817 ZL BREDA T 31 076 7920000 E infoapolyfluornl. Union Chemicals Factory UCF is a subsidiary of Ittihad International Investment LLC a large-scale private holding group governing several prestigious companies in the region.
9 Characteristics brown adhesive liquid organic weak acid soluble in water produce heat when diluted with water Usage. At 96 hr a differential effect of MMC on the absorption of each drug was seen. To Make Pakistan self reliant in basic chemicals with State of the art technologyTo capitalize and optimize all available domestic resourcesTo develop quality human resources at par to the international standards and make them.
206-793-1 CAS No. Linear Alkyl Benzene Sulphonic Acid Soft 90. Our Mission Our Mission is to take the best that exists and make it better.
The decreased absorption of salicylic acid and aspirin correlated with the reduced gastric mucosal blood flow. 1 Octane Sulphonic Acid Sodium Salt Anhydrous. It produces Caustic Soda Liquid Chlorine Hydrochloric Acid Sodium Hypochlorite Zinc Sulphate Calcium Chloride Linear Alkyl Benzene Sulphonic Acid LABSA and sodium Lauryl ether sulfate SLES.
As a substituent it is known as a sulfo groupA sulfonic acid can be thought of as sulfuric acid with one hydroxyl group replaced by an organic substituent. The C n H 2n1 chain is unbranched. In the production of saccharin in the production of solute colour in making sulpha drugs etc.
Ii From benzene sulphonic acid iii From diazonium salts iv From cumene. ICL has strategically. Carboxylic acids that consist of a carbonyl group two carbon atoms can lose carbon dioxide readily by heating above 150 o C.
1 in hair shampoo bath shampoo dish detergents and complex. At 96 hr there were severe. Reaction products of 134-thiadiazolidine-25.
Acid synonyms acid pronunciation acid translation English dictionary definition of acid. A prefix appearing in loanwords from Greek with the meanings at or to one side of beside side by side parabola. Any of a class of substances whose aqueous solutions are characterized by a sour taste the ability to turn blue litmus red and the.
C 6 H 5-SO 3 H H 2 O C 6 H 6 H 2 SO 4. It is a member of toluenes and an arenesulfonic acid. Sodium Lauryl Ether Sulfate SLES 70.
Palladium 10 On Activated Charcoal LR 10GM. High quality detergent and shampoo use Linear Alkyl Benzene Sulphonic Acid labsa 96 General Information Name. These were some basic methods for the laboratory preparation of benzene.
Preparation of benzene from sulphonic acids. Linear Alkyl Benzene Sulphonic Acid Soft 96. They are mainly produced as intermediate in the production of surfactants for use in detergent.
Benzoic acid 3-Aminoiminomethyl amino-4-methyl- methyl ester nitrate 1025716-99-7 Benzy l chloroformate 501-53-1 Benzyl S- glycidyl ether 16495-13-9. 1 in hair shampoo bath shampoo dish detergents and complex. 112233444-nonafluorobutane-1-sulphonic acid EC No.
Dodecyl Benzene Sulphonic Acid Methyl Ester Tetrapropylene Type Stock Standard Solution iv. Finar is now a part of ACETO a global manufacturer providing specialty chemicals intermediates excipients APIs to the life science.