Small spills of bromoform should be taken up. Kr for bromoform is 144Cmolal.
Bromoform should be stored in cool dark areas away from chemically active metals and strong caustics.
Decomposition of bromoform in light. P-listed codes and certain dioxin codes F020-F023 and F026-F028 are considered to be acute. Sep 21 2009 Cyclohexane C6H12 Bromine Br2 -- 1-Bromocyclohexane HBr. 287 289 For example Mediterranean grassland experiments where UV 289 291 or total solar radiation has been selectively.
Exposure to UV-A is associated with premature aging of the skin and some skin cancers. 2 M KCl and 10 drops. Bromine is the third halogen being a nonmetal in group 17 of the periodic table.
Methane is an odorless nontoxic flammable gas which has a boiling point of -1640C. The following is the List of Hazardous Substances prepared by the Director pursuant to Labor Code Section 6380. Condensed light ends spent filters and filter aids and spent desiccant wastes from the production of certain chlorinated aliphatic hydrocarbons by free radical catalyzed processes.
Chlorine dioxide gas can be absorbed by the skin where it damages tissue and blood cells. UV-A 315 to 400 nm visible light and other solar radiation are only weakly absorbed by the ozone layer. Bromoform See Benzene Bromoform See 12-Dichlorobenzene See Benzene See Bromoform Sources.
Acute exposure of the skin to chlorine that originates from the decomposition of chlorine dioxide causes irritations and burns. 950 n-Butyl. Gap-filled data are available for 53 sites.
It is not an official legal edition of the CFR. Natural gas is about 70-95 methane depending on the location in which it is obtained. 710 sec-Butyl acetate.
These chlorinated aliphatic hydrocarbons are those having carbon chain lengths ranging from one to and including five with varying amounts and positions of chlorine substitution T F026. Dec 07 2009 Ultrafast photolysis of bromoform CHBr3 with a 267 nm pulse of light followed by broadband transient electronic absorption identifies the photoproducts and follows their evolution in both neat bromoform and cyclohexane solutions. See 29 CFR 19101051.
STEL 1 ppm5 ppm Butanethiol. A short summary of this paper. In most instances the symptoms of primary irritation are observed shortly after exposure.
What is the empirical weight of the compound. The efficiency of ultraviolet UV light for primary disinfection of water depends on the intensity of the light and the length of time the. A laboratory preparation involves removing a bromine from bromoform using sodium arsenite.
INTRODUCTION 1-1 11 OBJECTIVE OF THIS MANUAL 1-1 12 BACKGROUND 1-2 13 REGULATORY CONTEXT 1-3 131 Disinfection Profiling and Benchmarking 1-7 14 USE OF DISINFECTANTS AS CHEMICAL OXIDANTS 1-8 15 How CHLORINE is ADDRESSED IN THIS GUIDANCE MANUAL 1-8 16 A SUMMARY OF ALTERNATIVE DISINFECTANT PROPERTIES 1-9 17 SELECTING A. Dec 07 2009 Ultrafast photolysis of bromoform CHBr3 with a 267 nm pulse of light followed by broadband transient electronic absorption identifies the photoproducts and follows their evolution in both neat bromoform and cyclohexane solutions. Only those codes applicable to the University of Maryland are listed Hazardous waste is any solid waste that either exhibits any of the characteristics of hazardous waste or is a listed EPA waste.
Cyclohexane has no pi-unsaturation and is therefore not nucleophilic. Note that this substance will attack some forms of plastics rubber and coatings. However some chemicals produce a delayed irritant effect because the chemicals are absorbed through the skin and then undergo decomposition within aqueous portions of the skin to produce primary irritants.
As pH increases the rate of decomposition of ozone is accelerated. The Code of Federal Regulations CFR is the official legal print publication containing the codification of the general and permanent rules published in the Federal Register by the departments and agencies of the Federal Government. Reckhow and Singer 2011.
It is produced by the bacterial decomposition of organisms in the absence of oxygen and is found in natural gas marsh gas bovine flatulence etc. The numeric value of 2445 in both formulae is the molar volume of air in litres at normal temperature and pressure NTP which is considered to be 25ºC and 1 atmosphere 101325 kPa or 760 mm Hg or 760 torr. 35 Full PDFs related to this paper.
Summary of Sampling Requirements for Physical and Chemical Parameters for Acceptability Aspects Parameter l. The Electronic Code of Federal Regulations eCFR is a continuously updated online version of the CFR. Academiaedu is a platform for academics to share research papers.
APHA 22nd ed2011 ADWG 34 Table D-3. Depletion of the ozone layer increases primarily the amount of UV-B radiation that reaches the surface Q16. 950 tert-Butyl acetate.
The substances on this list are subject to the provisions of Labor Code Sections 6360 through 63997 and Section 5194 in Title 8 of the California Code of Regulations. Full PDF Package Download Full PDF Package. A 1263 g sample of this compound was dissolved in 25 g of bromoform producing a solution that freezes at 44c the solvent has a freezing point of 78C.
Decomposition of plant litter via UV-induced processes has been shown to occur in arid-land ecosystems. Ethylene oxide epichlorohydrin hydroxylamines and the chemical mustard agents such as. It has been postulated that this decomposition process is responsible for a decrease in the classical formation of ozonation by-products eg aldehydes.
11 Consequently the results are best the bromine reagent is used in excess. Taste o Container Minimum Volume of Material Sample Glass-stoppered 500 mL Mode of Preservation Holding Time. 285 286 However new studies suggest that exposure to solar radiation can stimulate the breakdown of plant litter in a range of ecosystem and plant litter types.
Avoiding ozone depletion that would increase human exposure to UV-B radiation is a. What is the molality of the bromoform solution. Inhalation of chlorine dioxide gas causes coughing a sore throat severe headaches.
Eye exposure eyes to chlorine dioxide causes irritations watering eyes and a blurry sight. Molecular weights can be found in the NIOSH Pocket Guide to Chemical Hazards chemical supplier lists the NIST Chemistry WebBook or other online databases. Its properties are thus similar to those of fluorine chlorine and iodine and tend to be intermediate between those of the two neighbouring halogens chlorine and iodineBromine has the electron configuration Ar4s 2 3d 10 4p 5 with the seven electrons in the fourth and outermost shell acting as its valence.
2-Butanone Methyl ethyl ketone 78-93-3. 29 CFR 191019l 106-99-0. Kirk-Othmer Encyclopedia of Chemical Technology.
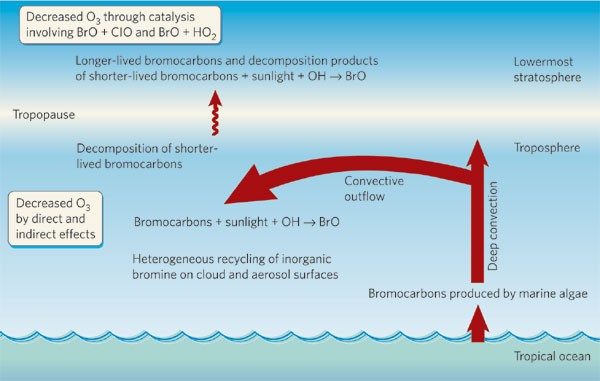
Mc Graw Hill-Water and Wastewater Engineering 2010 RETAi L EBook-Di Gi Book. Bromoform may be shipped via air rail road and water in containers bearing the label Keep Away From Food. Halomethane compounds are derivatives of methane CH 4 with one or more of the hydrogen atoms replaced with halogen atoms F Cl Br or IHalomethanes are both naturally occurring especially in marine environments and human-made most notably as.
See Butyl mercaptan. In addition EPA Hazardous Waste Codes are also classified as acute and non-acute.