UOP672-84 Aromatic Hydrocarbon Types in Detergent Alkylates Using Low Voltage Mass Spectrometry. 3 F Boiling Point.
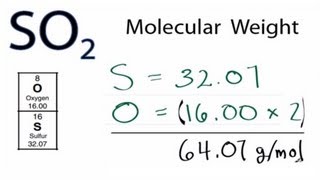
Molar Mass Molecular Weight Of So2 Sulfur Dioxide Youtube
15 Full PDFs related to this paper.

Relative molecular mass of sulfur dichloride. Oxides of sulfur Air contaminants classified as oxides of sulfur consist essentially of only two compounds sulfur dioxide SO2 and sulfur trioxide 803. Bakelite Structure Properties Application. A short summary of this paper.
A three-membered ring consisting of one oxygen atom and two carbon atomsEthylene oxide is a colorless and flammable gas with a faintly sweet odor. Ethylene oxide is an organic compound with the formula C 2 H 4 OIt is a cyclic ether and the simplest epoxide. Using the strict IUPAC naming.
The ratio of these is. As a result they will be pushed down giving the C2H4 molecule a trigonal planar molecular geometry or shape with respect to the Carbon on the left. Hence Beryllium Chloride will have a linear shape or.
This structure is also available as a 2d Mol file. Use section 28 of the data booklet. Handbook of Chemistry and Physics 84th - David R Lide.
Bromine number range 210-320 For use only as an adjuvant in vinylidene chloride copolymer coatings. If 84 g of the. This technique determines the ratio of biomass and fossil derived carbon dioxide in the carbon dioxide from.
This is in accordance with the VSEPR theory. The Shape Of Scl2 Molecule Is Sulfur dichloride SCl2 has a bent molecular geometry with approximate bond angles of 103 and a bond length of 201 pm. Contact or ingestion causes irritation or chemical burns to skin eyes and mucous membranes.
2 100 120 80 60 40 20 0 15 20 25 30 35 40 45 50 55 58 44 57 42 41 40 39 38 37 31 36 28 29 27 26 16 25 15 14 43 40 Relative intensity mz mz 58. Determine the empirical formula of a compound that contains 529 aluminum and 471 oxygen A. HEA have showed unusual physicochemical properties relative to traditional.
A compound has the empirical formula CH and a molecular mass of 78. C8H10N4O2 Using only whole numbers with no decimal point 4 mol of caffeine has ____mol of. 561 gmole 200 28049 gmole So the molecular formula is two times the empirical formula or Hg2Br2.
5 For purpose of determining if an emission threshold has been exceeded include in the emissions calculation any CO 2 that is captured for transfer off site. We would like to show you a description here but the site wont allow us. Because it is a strained ring ethylene oxide easily participates in a number of addition reactions that result in ring-opening.
Caesium The shape and position of the voltammetric wave is seen to depend on the relative proportions of Cs and Na in solution. Lead chloride PbCl2 Lead2 chloride. From the information provided the mass of sulfur is the difference between the.
Which of the following is a molecular compound. Sulfur monochloride appears as a yellow-red oily fuming liquid with a sharp odor. The Tetrahedral shape is a type of shape which a molecule takes form of when there are four faces or sides to the molecule forming a regular.
Its not like we have to reinvent the wheel. Brady The Molecular Nature of Matter 6th Edition Copia. UOP700-70 Free Sulfur In Petroleum Distillates and LPG by the AC Polarograph.
Atomic Radius In Periodic Table In Basic Chemistry. The molecular mass as reported in the problem is 561 gmole. Atomic Mass Of Elements.
A 15 mol of K atoms b 64 g of S c 20 g of Be d 25 g of Na. A 6 x 1023 S atoms b 075 mol of Cu atoms c 15 mol of N atoms d 3 x 1023 Na atoms. GHG i Mass emissions of each greenhouse gas metric tonsyear.
GWP i Global warming potential for each greenhouse gas from Table A-1 of this subpart. LeadII chloride ultra dry. The pri- mary source of both is the combination of atmo- spheric oxygen with the sulfur in certain fuels during their combustion.
An ion and a neutral atom Two ions of opposite charge Two neutral atoms Any combination above. Brady The Molecular Nature of Matter 6th Edition Copia. UOP704-70 Equivalent Weights of Heavy Organic Amines By Non-Aqueous Potentiometric Titration.
Polycarbonate resins complying with 1771580. Brady The Molecular Nature of Matter 6th Edition Copia. Fluorine on the other hand is toward the far right of the table just.
Atomic Mass And Molecular Mass. Polybutadiene resin molecular weight range 2000-10200. Ferric nitrate hexahydrate FeNO 3 2 6H 2 O 999 cobalt nitrate hexahydrate CoNO 3 2.
An element X is found to combine with oxygen to form a compound with the molecular formula X4O6. Analysis is by accelerator mass spectrometry beta-ionization or liquid scintillation. Given that the atomic mass is 2299 for sodium Na 15999 for oxygen O and 1008 for hydrogen H what mass of sodium hydroxide NaOH is needed to prepare 100 ml of 0125 M sodium hydroxide s.
Use this demonstration to determine the relative molecular masses of different gases using the ideal gas equation. C To calculate GHG emissions for. LeadII Chloride purified by sublimation for Perovskite precursor Cl2Pb.
Also poisonous by inhalation of. Chloride anhydrous RuCl 3 97 Chloro15-cyclooctadiene iridiumI dimer Bis15-cyclooctadienediiridiumI and dichloride C 16 H 24 Cl 2 Ir 2 98 was provided by Shanghai Civi Chemical Technology Co Ltd. N The number of greenhouse gases emitted.
Includes kit list and safety instructions. Download Full PDF Package. In association with Nuffield Foundation.
UOP676-84 Molecular Weight by Vapor Pressure Osmometry. Polyester resin formed by the reaction of the methyl ester of rosin phthalic anhydride maleic anhydride and ethylene glycol such that the polyester resin has an acid number of 4 to 11. Atomic Number Mass Number.
In general covalent bonding can occur between which of the following. Atomic Mass Molecular Mass. However molecular structure is actually three-dimensional and it is important to be able to describe molecular bonds in terms of their distances angles and relative arrangements in space.
The total emitted quan- tities of these substances are therefore directly related to the sulfur content and. TVFDJXOCXUVLDH-UHFFFAOYSA-N CAS Registry Number. Disulfide bonds in proteins are formed between the thiol groups of cysteine residues by the process of oxidative foldingThe other sulfur-containing amino acid methionine cannot form disulfide bondsA disulfide bond is typically denoted by hyphenating the abbreviations for cysteine eg when referring to ribonuclease A the Cys26Cys84 disulfide bond or the 2684 disulfide bond or.
Rank the following samples in number of atoms from least to greatest. What is the molecular formula of the compound. Science ASSIST has an expert national advisory team with extensive collective experience across all school laboratory management and safety.
I Deduce two features of this molecule that can be obtained from the mass spectrum. It is this team that will help with your enquiry. HF NaF CaCl2 MgO.
Determining relative molecular mass by weighing gases. Rank the following samples in mass from least to greatest. Magnesium dichloride magnesium chlorate monomagnesium chlorate magnesium chloride.