12 Qué les sucede al anhídrido sulfúrico y al ácido sulfúrico cuando entran al medio ambiente. Reacts violently with phosphorus trioxide Chem.

How To Make Sulfur Trioxide Youtube
At power plants flue gas is often treated with a series of chemical processes and scrubbers which remove pollutants such as sulfur dioxide and sulfur trioxide Speight 2013a.
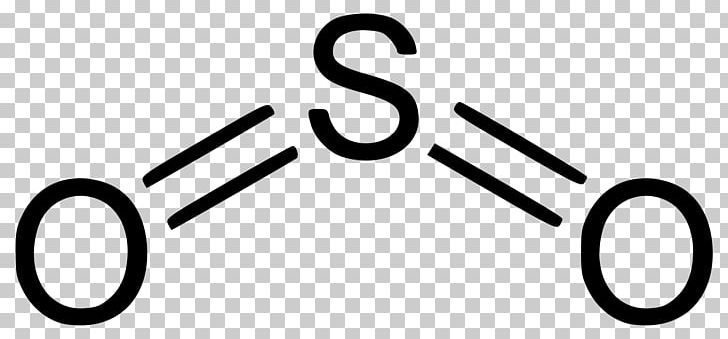
Sulfur trioxide gas. Sulfur trioxide alternative spelling sulphur trioxide also known as nisso sulfan is the chemical compound with the formula SO 3It has been described as unquestionably the most important economically sulfur oxide. Read More on This Topic. The SO 3 gas is removed with cellulose wool.
In all cases the alkalis react with the SO 2 in the presence of water eg a spray of slurry containing the sorbent to produce a mixture of sulfite and sulfate salts. Because it is the ratio of two quantities that have the same dimensions mass per unit volume specific gravity has no dimension. Clarification neededOn other planets sulfur dioxide can be found in various concentrations the most significant being the atmosphere of Venus where it is the third-most abundant atmospheric gas at 150 ppmThere it reacts with water to form clouds of sulfuric acid and is a key.
On average more than 95 percent of the fuel sulfur is converted to SO2 about 1 to 5 percent is further oxidized to sulfur trioxide. The ELEMENTRAC CS-i elemental analyzer can be equipped with up to 4 independent infrared cells. Sulfate aerosols can lower the Earths temperature by reflecting away solar radiation negative.
The gas flow continues past a heated catalyst where carbon monoxide CO is converted to CO 2 and where SO 2 is converted to sulfur trioxide SO 3 which is subsequently removed by a filter. To capture the acidic sulfur compounds from the flue gas. Attacks aluminum and zinc with evolution of.
Flue gas desulfurization units capture the sulfur dioxide and the sulfur trioxide if present and electrostatic precipitators or fabric filters remove particulate matter produced by burning fossil fuels. Handled at elevated temperature typically 290F to prevent solidification and makes transfers easier. Sulfur trioxide dissolves in water to form sulfuric acid which is a strong acid capable of corroding or destroying many materials.
Sulfur molten appears as a pale yellow crystalline solid with a faint odor of rotten eggsInsoluble in waterA fire and explosion risk above 450 F. Plants in turn utilize carbon dioxide as a source of carbon and return the oxygen to the atmosphere. Carbon is then detected as CO 2 by a second pair of NDIR cells.
Oxidation of both carbon monoxide to carbon dioxide and sulfur dioxide to sulfur trioxide follow the sulfur measurement. Specific gravity G is defined as the ratio between the weight of a. But sulfur dioxide is sometimes classified as indirect greenhouse gas.
It is the most potent greenhouse gas currently known with a global warming potential of 23900 times that of CO2 over a 100 year period SF6 has an estimated lifetime in the atmosphere of between 800 and 3000 years. The gas carbon dioxide which has a density of 1976 grams per litre under standard conditions has a specific gravity of 153 1976129. News 272144 1949.
Two molecules of hydrogen peroxide will produce two molecules of water and one molecule of oxygen. Reacts rapidly and exothermically with organic and inorganic acids with organic and inorganic acid anhydrides including oxides of nonmetals such as sulfur dioxide sulfur trioxide phosphorus trioxide phosphorus pentaoxide and with organic and inorganic acid chlorides. Mekanism adds various tiers of ore processing for better ingot yields from raw ores.
Answer 1 of 7. 2SO 2 O 2 2SO 3 3 SO 3 H 2 O H 2 SO 4 4 When the exhaust gases are discharged from the tailpipe and mixed with air either in the environment or in the dilution tunnel which is used for. Transported as a yellow to red liquid.
The paragraph below will help you in getting better insight. Each tier use a specific machine to process the raw ore for direct ingots dusts clumps shards or crystals Then the product is processed by the machines of the previous tiers. The ionization of SO2 and the sulfuric acid formed from sulfur trioxide SO3 also produces acidity.
A catalyst of potassium permanganate can be used to speed up this process. Reacts to incandescence with heated with thorium Mellor 7208 1946-47. Reacts with calcium phosphide incandescently at about 300C.
May react explosively with maleic anhydride MCA Case History 622 1960. This gas dissolves in rainwater to make acid rain which is a dilute solution of sulfuric acid H 2 SO 4. One word answer would be NO.
Sulfur hexafluoride is a sulfur coordination entity consisting of six fluorine atoms attached to a central sulfur atom. 13 Cómo podría yo estar expuesto al anhídrido sulfúrico o al. Oxygen a colorless odorless tasteless gas essential to living organisms being taken up by animals which convert it to carbon dioxide.
Sulfur trioxide can absorb moisture from the atmosphere. Sulfur dioxide is found on Earth and exists in very small concentrations and in the atmosphere at about 1 ppm. Oxygen forms compounds by reaction with practically any other element.
Help text not available for this section currently. Uncontrolled SOx emissions are almost entirely dependent on the sulfur content of the fuel and are not affected by boiler size burner design or grade of fuel being fired. Sulfur trioxide irritates the mucous membranes of the respiratory tract.
Ignites in fluorine gas at ordinary temperatures Mellor 211-13 1946-47. Sulfate aerosols are injected into the atmosphere from the combustion of fossil fuels and the eruption of volcanoes like Mt. Sulfur trioxide exists in several forms - gaseous monomer crystalline trimer and solid polymer.
2 in the exhaust gas dissolves in the washwater where it is ionized to bisulphate and sulfite which are then readily oxidized to sulfate Hassellöv and Turner 2007. All living things contain sulfur and when fossilised as in fossil fuels the sulfur remains present. Can react with ammonia to form explosive sulfur nitride.
The sulfuric acid in the water then reacts with carbonates and other salts in the seawater to form. If unpurified fossil fuels are burnt sulfur dioxide can enter the atmosphere leading to acid rain. Modern sulfur production is almost entirely from the various purification processes used to remove sulfur from natural gas oil and tar sands.
Oxidation of sulfur dioxide to sulfur trioxide with the subsequent formation of sulfuric acid H 2 SO 4 described by reactions 3 and 4 is perhaps the most important of these processes. The carbon content is detected by infrared cells which can be individually customized. Particulate matter that consists of compounds of sulfur formed by the interaction of sulfur dioxide and sulfur trioxide with other compounds in the atmosphere.
Resúmenes de Salud Pública - Anhídrido sulfúrico y ácido sulfúrico Sulfur Trioxide and Sulfuric Acid English US Páginas relacionadas. It is prepared on an industrial scale as a precursor to sulfuric acid. This reaction may take place in the bulk solution or on the wetted surface of the solid.
Sulfur dioxide is then further oxidised in the atmosphere to sulfur trioxide SO 3. Adding potassium permanganate to the hydrogen peroxide will cause a reaction that produces a lot of heat and water. Any gas in atmosphere that absorbs and emits radiant energy within the thermal infrared range is qualified to be consi.
Potassium permanganate - Hydrogen peroxide will decompose into water and oxygen gas. 11 Qué son el anhídrido sulfúrico y el ácido sulfúrico. These include controls for sulfur trioxide SO 3 hydrochloric acid HCl.
A concentration of 1 volume of SO3 in a million volumes of air one part per million or 1 ppm is enough to cause coughing and choking.