HX-878 Tepanol Hydroxyethyl Cellulose. Avoid all of these.

Potassium Chromate Solution 50 G L 5 In Water Th Geyer
Take 3 g of oxalic acid dihydrate H 2 C 2 O 42H 2 O into a 50 mL beaker.
Potassium and chromate. Potassium chromate K 2 CrO 4 is used in inks dyes stains bright yellowish-red color. Rare potassium chromate minerals and related compounds are found in the Atacama desert. In explosives and fireworks.
Potassium chloride is usually prescribed in the form of a capsule or a tablet. As the silver nitrate solution is slowly added a precipitate of silver chloride. On heating potassium dichromate decomposes to form potassium chromate chromic oxide and oxygen.
There are thousands of uses of various potassium compounds. Le dichromate de potassium aussi appelé bichromate de potassium est un solide ionique orange de formule K 2 Cr 2 O 7. Weight of Cis-potassium di aqua bis oxalato chromateIII dihydrate 1071.
In this experiment you will use a standard solution of potassium dichromate K 2 Cr 2 O 7 to determine the percent by weight of iron as Fe 2 in an unknown solid. What color is the precipitate between silver nitrate and potassium chromate. When silver nitrate and potassium chromate solutions are mixed together then there is formation of silver chromate and potassium nitrate.
The number in parentheses is the metric equivalent in kilograms. Crocoite PbCrO 4 which can occur as spectacular long red crystals is the most commonly found chromate mineral. In this experiment students add dilute sulfuric acid to an aqueous solution of potassium chromateVI.
Place one drop of water in a small depression in the mixture and cover the dish with a watch glass. The structure of the bond is rigid strong and often crystalline and solid. What is the complex.
Lion chromate de couleur jaune a tendance à se dimériser en solution aqueuse pour former lion dichromate de formule Cr 2 O 7 2-de couleur orange. CS PowderCrystal Riot Control Agent DHE. Cr 2 K 2 0 7 the hexavalent form of chromium Cross-reactions.
Answer 1 of 4. The reaction is as follows. This substance is a known human carcinogen and is associated with an increased risk of developing lung cancer and cancer of the sinonasal cavity.
For example nitrate ion NO 3- contains one nitrogen atom and three oxygen atomsThe atoms in a polyatomic ion are usually covalently bonded to one another and therefore stay together as a single charged unit. NCI05 NCI Thesaurus NCIt. Chromate containing minerals are rare.
Unfortunately there is a problem here. Potassium chloride is a great source of potassium and is therefore used to treat hypokalemia. The Complete ionic equation shows all the ions present in solution separately.
Hydroxyl Terminated Polybutadine HTPB Isodecyl Pelargonate IDP Isophorone di-isocyanate IPDI Microcrystalline Wax Desensitizing. This is described above if you have forgotten. Dans leau il se dissocie en ions dichromate et en ions potassium.
They observe the resulting colour changes before reversing the reaction using aqueous sodium hydroxide. A regulated marked area should be established where this chemical is handled used or stored in compliance with OSHA Standard. It is a powerful oxidizing agent.
You may remember that that is done by adding acid. Potassium oxalate K2C2O4H2O was added. Prepare a solution of 1g of potassium dichromate K 2 Cr 2 O 7 dissolved it with 1ml of hot distilled water.
Cr 2 O 7 2- 14H 6I rightarrow 2Cr 3 7H 2 O 3I 2. Ionic bonds also melt at high temperatures. Dark green solution was formed and.
Ionic bonds are atomic bonds created by the attraction of two differently charged ionsThe bond is typically between a metal and a non-metal. Lead chromate must be stored to avoid contact with oxidizers such as perchlorates peroxides permanganates chlorates and nitrates and chemically active metals such as potassium sodium magnesium zinc and ferric ferrocyanide since violent reactions occur. In fact it can even be taken to prevent hypokalemia.
Prepare an intimate mixture of finely ground potassium dichromate 2 g and oxalic acid dihydrate 6 g and heap the powder in a 15 cm evaporating dish. 2Potassium di chromate. 4K 2 Cr 2 O 7 rightarrow 4K 2 CrO 4 2CrO 3 3O 2.
This insoluble solid is the precipitate and it has a red-brown color. For a medical purpose. The first number under the column headed RQ is the reportable quantity in pounds.
The experiment is most appropriate with A-level students given the potential hazards with solutions containing chromateVI and dichromateVI ions. One example is potassium superoxide KO 2 an. It can also be administered orally in the liquid form.
The compound has bright red crystals and is used for dyeing staining and tanning etc. This reaction requires 6 electrons and 14 hydrogen ions. Write the formula and Mw.
Potassium chromate primarily affects the nose throat and lungs causing ulcerations shortness of breath bronchitis pneumonia and asthma but can also affect the gastrointestinal tract liver kidneys and immune system. Polyatomic ions are ions which consist of more than one atom. Covered the beaker with a large.
Otherwise it could be carried out as. Then dissolve the oxalic acid with 2ml of boiling distilled water. Dichromate ion reduces to two chromiumIII ions.
This reaction is also described further up the page. L ion dichromate Cr 2 O 7 2- étant un puissant agent oxydant ce produit est couramment utilisé dans les réactions d oxydoréduction en laboratoire et dans lindustrie. Add this solution in portion to the oxalic acid solution.
8H3H2C2O46K2C2O4H2OK2Cr2O7 6CO29H2O8K2K3CrC2O433H2O Vigorouly orange solution was formed and filtered then leaves for crystallization. At work request a material safety data sheet to help identify alternatives that are safe hence avoiding contact with material containing chromates. All that is left is to convert the yellow potassium chromateVI solution into orange potassium dichromateVI solution.
Our product line consists of chemical solutions prepared to exact quality standards and certified for use in laboratories and production processes. Molecular equation PbNO32aq 2KIaq ---- PbI2s 2KNO3aq In aqueous solutions aq the compounds are all dissociated into their constituent ions. The solution turns yellow as potassium chromateVI is formed.
Potassium dichromate K2Cr2O7 dissoved slightly acid solution excess oxalic acid H2C2O4 is added and the following reactiion take place. Pb2aq 2NO3-aq 2Kaq 2I-aq. Among them is lópezite.
It is important to note that potassium chloride must not be administered to individuals suffering from. It oxidizes iodide to iodine. Theoretical yield in gram KCrC 2 O 4 2 H 2 O 22H 2 O Percentage yield KCrC 2 O 4 2 H 2 O 22H 2 O Draw the structure of Cis potassium di aqua bis oxalato chromateIII dihydrate.
Dans certaines conditions on obtient une polymérisation et la production de polychromates de formule Cr n O 3n1 2-de couleur rouge. Dimeryl-di-isocyanate DDI Dioctyl Adipate DOA Glycerin Glycerol. La synthèse se fait par oxydation de chrome-III en chrome-VI qui peut selon les.
Cr 2 O 7 2- 14H 6 e- 2Cr 3 7H 2 O Only one electron is necessary to. In the tanning of leather in fly paper and safety matches but all these uses are due to the chemistry of the chromate ion rather than the potassium ion. After a short induction period the reaction commences and soon becomes vigorous with the evolution of steam and carbon dioxide.
This activity includes every compound formula and name that can be formed from the list 44 Ions provided in Chemistry A at Pickerington High School Central. We regularly produce chemical solutions to specifications designed by government and regulatory bodies commercial and trade associations and the specific needs of individual users and businesses.
Acetyl coenzyme A sodium salt. Fe 3 No interference for Fe 3 500ppm.
Calcium brines are limited to 1250 ppm chromate and sodium brines are limited to 2500 ppm chromate.

Sds sodium chromate. Apart from structural wood preservation measures there are a number of different chemical preservatives and processes also known as timber treatment lumber treatment or pressure treatment that can extend the life of wood timber and their associated products including engineered wood. The American Society of Refrigeration Engineers has established chromate limits in brine treatments. Use a stick to poke something rather than touching it directly Engineering Use a device to protect you eg.
Potassium Chromate Indicator Potassium Hydroxide 45 ULC Potassium Hydroxide 45 Potassium Iodide 50Â wv Potassium Iodide 20 Solution Potassium Iodide 50 Potassium Permanganate Potassium Permanganate Reagent Powdered Chlorine Bleach Powdered Deep Fry. 001molL水酸化ナトリウム溶液N100 001molL Sodium hydroxide solution N100. Exposure limits Once the information on possible types of exposure has been determined the.
Under this regulation DuPont Tyvek 400 Tyvek 400 D Tyvek 500 Tyvek 600 Tyvek 400 FC ProShield ProShield 70 ProShield 50 ProShield 10 ProShield 60 ProShield 6 SFR and. Safety Data Sheets SDS are written in accordance with applicable federal regulations 29 CFR 19101200 to communicate health and safety data on usage and handling of hazardous chemicals. Company About Us Responsibility Events Press Releases Programs Careers Offices.
If we cant find the MSDS on the manufacturers website a NOT FOUND error click on the link next to Is the link to this page broken and our search engine will search the internet for you to find the replacement. Chromium Hexavalent-15-diphenylcarbohydrazide Method 10218. Coenzyme A trilithium salt.
Customer Support Contact Us FAQ Safety Data Sheets SDS Certificates COACOO Quality Regulatory Calculators Apps Webinars. Nitrite is also an effective inhibitor but in open systems it tends to be oxidized to nitrate. K No interference for K 500ppm.
Solutions are being studied to allow Flash games being playable again on browser. Evaluation of risks to health for sodium. C 23 H 38 N 7 O 17 P 3 S.
Chromium for Water and Wastewater. A substance of very high concern SVHC is a chemical substance or part of a group of chemical substances concerning which it has been proposed that use within the European Union be subject to authorisation under the REACH Regulation. Sulfuric Acid SDS.
We are a leading supplier to the global Life. Instead of potassium chromate use food dye to give a yellow colour to solutions for demonstrations Isolation Keep away from it eg. In the method using chelating agents EDTA at pH 44 was the best for separating heavy metal ions 901 for Cd 2 871 for Zn 2 and recovering SDS 655 for Cd 2 685 for Zn 2.
However due to health and environmental con-cerns use of chromate has decreased significantly and will probably be outlawed in the near future. Cr 3 Interference for Cr 3 2 ppm giving low COD. FTP name this rule which states that the more highly substituted alkene will prevail in.
Encourage constructivism inquiry and cooperative learning. Ride-on lawn mowers. Chromium Hexavalent Method 8023.
Where to buy yellow chromate Oh Nooooo. Empirical Formula Hill Notation. Essentially the opposite of Markovnikoff addition it allows for the prediction of chemical products upon the elimination reaction of an alkyl halide.
Our product line consists of chemical solutions prepared to exact quality standards and certified for use in laboratories and production processes. 2009 tested chelation followed by UF and acidification followed by UF for the separation of Cd 2 or Zn 2 from SDS micelles in simulated retentate solution of MEUF and the reuse of SDS. The Color of Art Pigment Database - Pigment Brown PBr page is a complete artists reference on brown and red earth pigments with color Index nameslightfastness specifications and health and safety information for iron oxides natural earths and other brown artists pigments and paint.
Student Safety Contracts and Exams. We regularly produce chemical solutions to specifications designed by government and regulatory bodies commercial and trade associations and the specific needs of individual users and businesses. The all-in-one platform for school district science programs.
Indeed listing of a substance as an SVHC by the European Chemicals Agency ECHA is the first step in the procedure for authorisation or restriction of use of. Wood easily degrades without sufficient preservation. Orders Quick Order Custom Products Commerce Solutions.
In closed systems that continuously run at temperatures below 32F 0C a closed brine system is often employed. Print the MSDS close the window and you will return to this page. From January 2021 many browsers will no longer support Flash technology and some games such as Super Smash Flash 2 may not work.
Carry out an experiment in a fume cupboard Administration Arrange things so that people do not go near something risky eg. Fe 2 Interference for Fe 2 100 ppm giving low COD. Li et al.
01molL炭酸水素ナトリウム溶液N10 01molL Sodium hydrogen carbonate solution N10 容量分析用滴定液. Chromium Total Alkaline Hypobromite Oxidation Method 8024. Cobalt 1-2-Pyridylazo-2-Naphthol PAN Method 8078.
Cadmium Calcium Hardness Carbon Dioxide Cationic Surfactants Chelant Chloride Chlorine Bromine Total Chlorine Demand Requirement Chlorine Dioxide Chlorine Free Chlorine Free Total Chlorine Hypochlorite Chlorine Total Chlorite Chromate Chromium Hexavalent Chromium Hexavalent and Total Chromium Total Cobalt. Chromate Detergents acids alkalis oils solvents or exposure to abrasive caustic or wet environments Onset Begins within hours or days of exposure Begins within minutes or hours of exposure Typical Presenting Symptoms Pruritis papules vesicles edema fissures erythema and oozing May spread beyond contact site. C 21 H 33 Li 3 N 7.
The safety data sheets SDS or other applicable information sources should be used to identify hazardous ingredients and expected products of reaction and decomposition. Chromate is an excellent aqueous corrosion inhibitor particularly from a cost perspective. In the reaction of 2-bromobutane in a solution of sodium ethoxide and ethanol the products will be 2-butene and 1-butene in a ratio of approximately 73.
Preparation of Safety Data Sheets for Hazardous Chemicals. SDSs are a widely used system for cataloging information on. Its hazardous properties relate to exposure via skin or eye contact and inhalation.
Na No interference for Na. Information on electrodes the metals being welded or cut and the specific type of welding process should also be identified. Molybdate and tetraborate dont provide an appropriate corrosion inhibition efficiency in solutions containing aggressive anions due to instability of protective layer formed on the metals surface Mustafa and Shahinoor Islam Dulal 1996.
Mg 2 No interference for Mg 2 500ppm. Chromate Titration using Sodium Thiosulfate Method 8211. Citation White Mineral Oils USP_sds_en Citri Foam Citri Green Citri Suds Citri-Pass Citric Acid Citric Acid 50 FG.
A safety data sheet material safety data sheet or product safety data sheet are documents that list information relating to occupational safety and health for the use of various substances and products. LEARN MORE POGIL. Chromium Total-15-Diphenylcarbohydrazide Method 10219.
Material Safety Data Sheets from the manufacturer or distributor The links below open new windows. Email email protected PAVO. For instance chromate is a toxic compound and also increases the corrosion rate by increase in cathodic reaction rate.
Box 219 Batavia IL 60510. Ag Interference for Ag 10 ppm giving low COD. The pH should be 70-85.
1molL Sodium chloride solution 1N 容量分析用滴定液. In contrast even brief exposures to high concentrations of sodium hydroxide may lead to immediate effects which include irritation and burning of the skin eyes and respiratory tract and blindness. Chemical Safety Data Sheets.
Empirical Formula Hill Notation.
2- Ag2CrO4 2 Ag CrO4 7. Of lead confirmed 2 Potassium chromate test.

Soluble Salts In Water Ki And K 2 Cro 4 Potassium Iodide And Potassium Chromate Are Water Soluble Agi And Pbcro 4 Silver Iodide And Lead Chromate Ppt Download
Any source of H would have caused the same shift in this equilibrium.
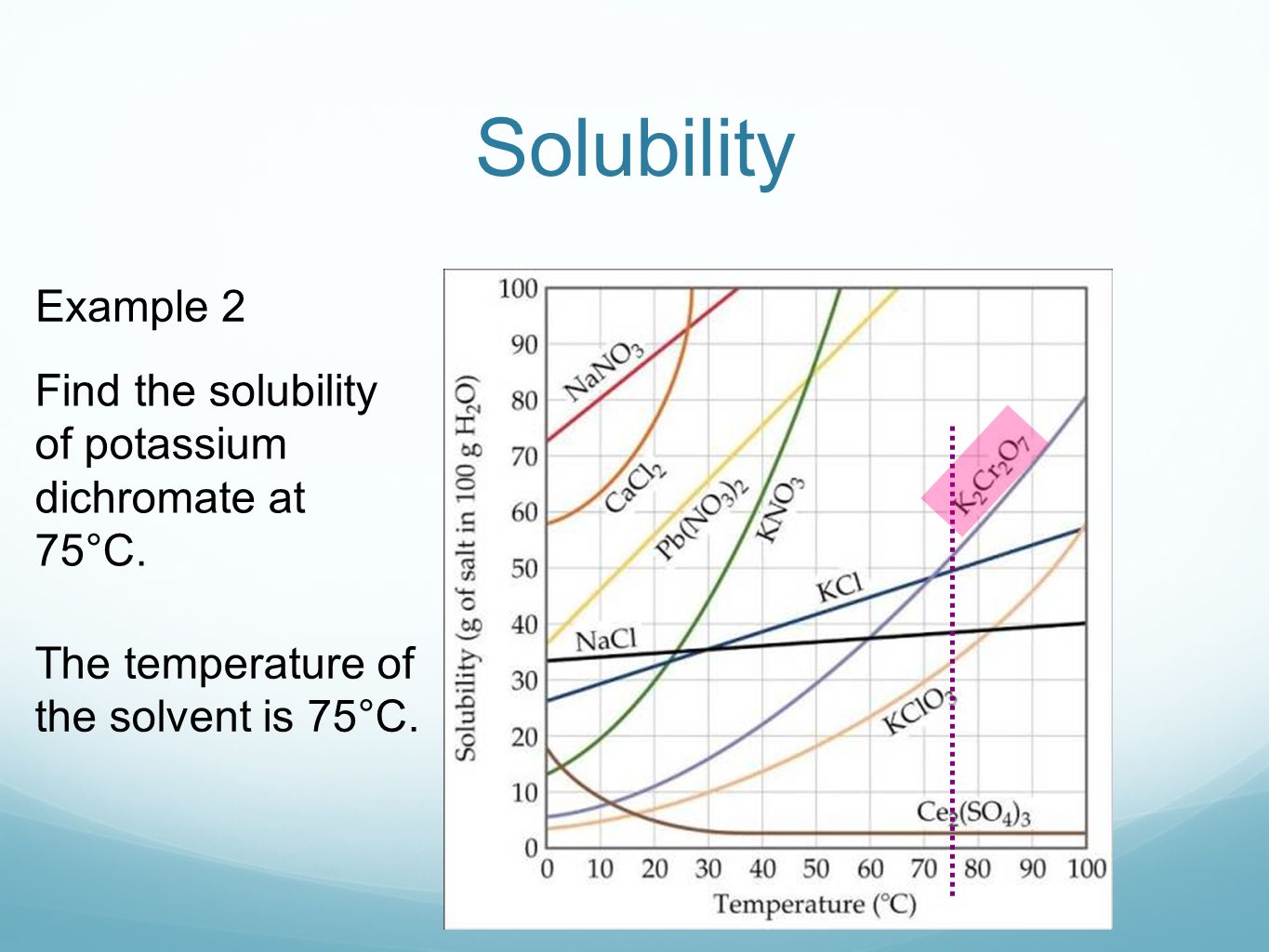
Potassium chromate solubility why. What indication of chemical change will be used to study the equilibrium constant of the reaction in this experiment Aug. The chromate that is the most soluble in water at 25 o C on a molar basis is. Is cacl2 soluble in water email protected.
Its simple reliable and hassle-free. Strontium is water insoluble but it does react with water. Gemfury is a cloud repository for your private packages.
What happened to the color of the K 2 CrO 4 when you did this. Pb2aq 2NO3-aq 2Kaq 2I-aq. Strontium compounds can be water soluble.
The most significant strontium mineral is celestite strontium sulphate. After you have saved the data pair return the test tube to the test tube rack and place. Feb 28 2017 This monograph for Barium Chloride Dihydrate provides in addition to common physical constants a general description including typical.
Solubility Mass of solutesaltsolid x 100 50 x 100 100 g 100g H 2 O Massvolume of watersolvent 500 b Calculate the solubility of potassium cHlorateV if 500 g of the salt is dissolved in 2500cm 3 of water. To the salt solution add KI solution Yellow ppt Pre. Calcium compounds are more or less water soluble.
Calcium carbonate has a solubility of 14 mgL which is multiplied by a factor five in presence of carbon dioxide. Molecular Weight Molar Mass. Ksp solubility product constants of many popular salts at SolubilityOFthings.
Sulfuric acid is a source of H. Add Nesslers reagent Brown ppt Pre. Properties of Potassium dichromate K 2 Cr 2 O 7.
Calcium is a chemical element with the symbol Ca and atomic number 20. Based on the color change you saw which way did the equilibrium shift right or left. Potassium peroxochromate K 3 CrO 2 4 is made by reacting potassium chromate with hydrogen peroxide at low temperatures.
Package install and use your code anywhere. Compounds of chromiumIV are slightly more common than those of chromiumV. Chromate forms a precipilate with Ag but this precipitate has a greater solubility than that of AgCl for example.
BaOH 2 x 8H 2 O. Produced from potassium chromate by the roasting of chromite ore with potassium hydroxide. CrSn4Cr3Sn2 Sn4 2LiSLi2S is a redox reaction.
Select the correct answer below. Therefore AgCl is formed first and after all Cl- is consumed the first drop of Ag in excess will react with the chromate indicator giving a reddish precipitate. Refer to the solubility rules as needed.
Question 8 predict the product for the following reaction. Without the s Potassium sulfate solution is mixed with barium chloride solution. Im using a diamide ligand as the receptor and an anion chromate nitrate and chloride as the.
Help students understand that the graph shows that more sugar dissolves in water as the temperature of the water increases. In this method neutral medium should be used since in alkaline solutions silver will. Im conducting on study of anion complexation.
Solubility of strontium and strontium compounds. Academiaedu is a platform for academics to share research papers. Iron III nitrate and potassium thiocyanate.
In Step A1 on Page 150 you added sulfuric acid H 2 SO 4 to a sample of potassium chromate K 2 CrO 4. S is the oxidizing agent oxidation change from 0 to 2. Stoichiometry is the chemistry that mathematically relates all substances in a reaction quantitatively relating the amount of reactants and products in a chemical reaction.
Solubility of calcium and calcium compounds Elementary calcium reacts with water. As an alkaline earth metal calcium is a reactive metal that forms a dark oxide-nitride layer when exposed to airIts physical and chemical properties are most similar to its heavier homologues strontium and bariumIt is the fifth most abundant element in Earths crust and the third most abundant metal after iron and. It allows the chemist to determine the amount of product that will form from a given amount of reactants or the amount of one reactant that is needed to react completely with some specific amount of the other reactant.
What is negative absorbance and why am I getting it. Some other important properties of potassium dichromate are listed below. Water and undergoes evaporation on heating 37.
Of lead confirmed ANALYSIS OF GROUP 2Copper 1 To the salt solution add few drops of potassium. Sep 22 2021 Part 5. 0020 M FeNO 3 3 aq iron III nitrate dissolved in 1.
The IronIII - Thiocyanate Equilibrium Pages 150-151 and Page 158. Sawdust water sawdust floats on tion and hence carbon and potassium possess strong water iv The solid retained on the filter paper is called a forces of attraction. Calcium chromate solubility is 170 gL and at 0 o C calcium hypo.
Rarely occurring naturally barium chromate 2. See your observation on Page 157. Which will be the solid product when strontium bromide and potassium sulfate react.
Describe what happens when ionic and covalent molecular substances dissolve. Why is strontium present in water. To the salt solution add k2CrO4 solution Yellow ppt Pre.
Soft metals are sodium and potassium Match the following 24. K 2 Cr 2 O 7. The tetrahalides CrF 4 CrCl 4 and CrBr 4 can be produced by treating the trihalides.
90 Barium hydroxide s 944. The solubility of potassium dichromate is 120 g1000 g H_2O at 20 degrees C. This red brown compound is stable at room temperature but decomposes spontaneously at 150170 C.
Access the answers to hundreds of Solution questions that are explained in a way thats easy for you to understand. A mixture has a variable composition. 0011 473 BeF2 Beryllium fluoride 552 1.
Calcium phosphate solubility is 20 mgL and that of calcium fluoride is 16 mgL. Get help with your Solution homework. Identify which of the following is the oxidizing agent and its change in oxidation number.
When potassium chromate K2CrO4 is dissolved in water what ions are produced. K 2 Cr 2 O 7 has a. A solution is prepared by adding 264 g of potassium dichromate to 440 g of water at 20 degrees C.
Identify the oxidizing agent in the following reaction. A Calculate the solubility of potassium nitrateV if 50 g of the salt is dissolved in 500cm 3 of water. Of ammonium confirmed ANALYSIS OF GROUP 1LEAD 1 Potassium iodide test.
A b Sodium. BaIO 3 2 x H 2 O. Answer 1 of 4.
The Complete ionic equation shows all the ions present in solution separately. Evaporation NaCl water NaCl is soluble in 28. Examples include strontium carbonate with a water solubility of 10 mgL and strontium chromate with a water solubility of 9 mgL.
Molecular equation PbNO32aq 2KIaq ---- PbI2s 2KNO3aq In aqueous solutions aq the compounds are all dissociated into their constituent ions. A Ag 2 CrO 4 b BaCrO 4 c PbCrO 4 d impossible to determine e none of these 4. Solution Questions and Answers.
An ionic compound dissolved in water H2Ol will produce aqueous anions and aqueous cations in solution A covalent compound dissolved in water H2Ol will produce aqueous covalent compound in solution. 2 H2 O Reach Registration Number -1.
Lead chromate and other test swabs are self-contained units with a fiber tip at one end and glass ampoules with reactive materials inside the swab barrel. Given 640 g of HBr.
The anion is named.

Mass of lead ii chromate. MC3 removed a cationic and an anionic dye from monocomponent solutions efficiently. A new bi-functionalized biosorbent MC3 was synthetized from cellulose CModification of C was performed using succinic anhydride and choline chloride. How much should you weigh on the scale.
Uncountable A heavy pliable inelastic metal element having a bright bluish color but easily tarnished. A CuClO₃ B KMnO₄ C PbCrO₄ D CaF₂ E. Similarly under the Cotton Dust standard 29 CFR 19101043 the PEL must be proportionately reduced for extended work shifts for the purpose of determining whether and for how long respirators.
While squeezing gently the tip of the. Atomic number 82 symbol Pb from Latin plumbum countable nautical A plummet or. Of pulse with 5 ml.
What is the mass of exactly one mole of calcium acetate. Li 2 CO 3. 133 Galvanic and electrolytic cells ESCR3 Electrochemical reactions ESCR4.
Bi 3 bismuthIII ion. How many moles are contained in 158 g of the substance. Observation Inference 1mark white ppt persistremains SO 4 2- Cl- IIITo the portion in iv II above Heat to boil.
Observation Inference 1mark white ppt CO 3 2- SO 4 2- SO 3 2- Cl- IITo the portion in iv I above add five drops of dilute Hydrochloric acid. Cut a piece of potato add salt and wait minute and add two drops of lemon juice. Known since the Middle Ages by the name plumbum dulce the production of leadII nitrate from either metallic lead or lead oxide in nitric acid was small-scale for direct use in making other lead.
Write a formula for each of the following ionic compounds. LeadII Acetate PbC2H3O24 LeadIV Acetate PbNO32 LeadII Nitrate PbCl2 LeadII Chloride PbCl4 LeadIV Chloride PbCrO4 LeadII Chromate PbI2 LeadII Iodide PbO LeadII Oxide PbO2 LeadIV Oxide PBr3 Phosphorus Tribromide PbS LeadII Sulfide PCl3 Phosphorus Trichloride PCl5 Phosphorus Pentachloride PF3 Phosphorus Trifluoride PH3. A copperI chlorate B potassium permanganate C leadII chromate D calcium fluoride E ironII phosphate F lithium hydrogen sulfite.
We would like to show you a description here but the site wont allow us. Carcinogenic lead chromate induces DNA double-strand breaks in human lung cells. Both groups have to absorb substantially more iron than is lost from the body and both are at a considerable risk of developing iron deficiency under ordinary dietary circumstances.
In case of common salt there will be no blue colour. Amphetamine methadone benzodiazepine opiate and cocaine metabolite tests are also affected at glutaraldehyde concentration between 1 and 2 using EMIT II immunoassays. The charge on the anion is the group number minus eight.
What is the formula mass of magnesium chloride MgCl₂. Xu B Wakeman T Pelsue SC Singh NP Wise JP. PbC 2 O 4.
Zhitkovich A Song Y Quievryn G Voitkun V. With our money back guarantee our customers have the right to request and get a refund at any stage of their order in case something goes wrong. Pink colour indicates Lead Chromate.
It is easily fusible forms alloys with other metals and is an ingredient of solder and type metal. IvITo the fourthportion add three drops of LeadIInitrateIVsolution. Of water and add a few drops of HCl.
Glutaraldehyde at a concentration of 075 volume could lead to false-negative screening results for a cannabinoid test using the EMIT II drugs of abuse screen. Different forms of lead and particle size have different rates of absorption in the rat. During pregnancy more iron is needed primarily to supply the growing fetus.
Both malleable and ductile though with little tenacity. The ability of copper to cycle between an oxidized state CuII and reduced state CuI. How many leadII and iodate ions are present in 38 L of a saturated solution of leadII iodate.
AJS is the official journal of 7 major surgical societies and publishes their official papers as well as independently submitted clinical studies editorials reviews brief reports. Answer 1 of 4. Lead Chromate is a yellow orange or red colored crystalline inorganic compound that emits toxic chromium fumes upon heating.
Pb 4 leadIV ion. An inorganic pigment that is the reaction product of high temperature calcination in which lead II oxide silicon oxide tin IV oxide and zinc oxide in varying amounts are homogeneously and ionically interdiffused to form a crystalline matrix of spinel. MC3 is a versatile biosorbent that can be reused in new adsorption cycles.
Lead II acetate C ammonium iodide D potassium chlorate. Pb2aq 2NO3-aq 2Kaq 2I-aq. Its preparation is an entertaining and popular.
Use 4 significant figures. The compound currently has a few specialized applications such as the manufacture of solar cells and X-ray and gamma-ray detectors. Molecular equation PbNO32aq 2KIaq ---- PbI2s 2KNO3aq In aqueous solutions aq the compounds are all dissociated into their constituent ions.
LeadII nitrate is an inorganic compound with the chemical formula PbNO 3 2It commonly occurs as a colourless crystal or white powder and unlike most other leadII salts is soluble in water. The Complete ionic equation shows all the ions present in solution separately. The swabs are activated by squeezing at the crush points marked on the barrel of the swab shaking well to mix the reagents and then squeezing until the reactive liquid comes to the tip of the swab.
Among healthy human beings pregnant women and rapidly growing infants are most vulnerable to iron deficiency Bothwell et al 1979. He American Journal of Surgery is a peer-reviewed journal designed for the general surgeon who performs abdominal cancer vascular head and neck breast colorectal and other forms of surgery. The lead standards for construction 29 CFR 192662 and general industry 29 CFR 19101025 require PEL adjustments with respect to extended work shifts workshifts longer than eight hours.
This substance is used in printing inks paints and to color vinyl rubber and paper. How many moles is this. Pb 2 leadII ion.
If iodized salt blue colour will develop. Observation Inference 1mark white. Will a precipitate form if 34 mL of 88 x 10-3 M leadII nitrate are mixed with.
In Grade 11 you carried out an experiment to see what happens when zinc granules are added to a solution of copperII sulfate. Lead chromate is highly corrosive and is a strong oxidizing agent. Main-Group Nonmetals Groups IVA VA VIA and VIIA Group IVA VA VIA and VIIA nonmetals tend to form anions by gaining enough electrons to fill their valence shell with eight electrons.
LeadII iodide or lead iodide is a salt with the formula PbI 2At room temperature it is a bright yellow odorless crystalline solid that becomes orange and red when heated. Bi 5 bismuthV ion. Lead chromate primarily affects the lungs causing shortness of.
MC3 showed very high adsorption capacity for crystal violet and orange II. PMC free article Google Scholar 160. Non-oxidative mechanisms are responsible for the.
Write the correct formula for calcium acetate and then answer 23 - 25 based on it. You need 00100 mole of lead II chromate. Carbonate and thallate were absorbed to the greatest extent whereas absorption of sulfide chromate naphenate and octoate were absorbed at only 44-67 the rate of absorption for lead acetate Uptake of lead into the femurs of rats was highest for lead acetate intermediate for lead oxide and lowest.
It was formerly called plumbous iodide. What is the maximum concentration of chromate ion that is allowed before silver chromate will precipitate if the silver ion concentration is 78 x 10-4 M. Described a simple fluorometric method for.
