A molecule of sucrose sugar C 12 H 22 O 11 with 8 hydroxyl groups -OH where at 20C over 200g can be dissolved in just 100 mL of water. PHYSICAL AND CHEMICAL PROPERTIES APPEARANCE.
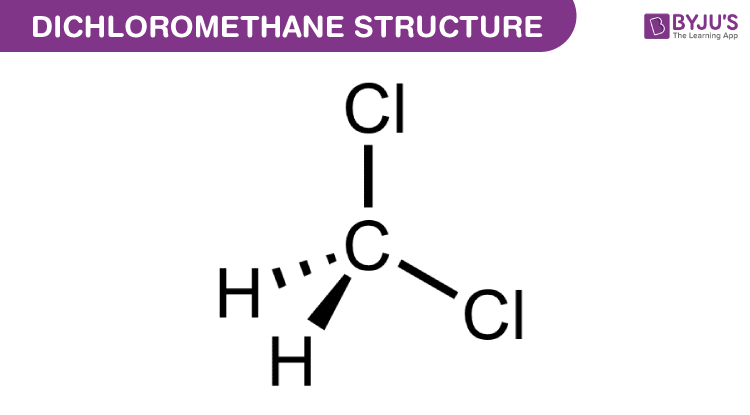
Dichloromethane Structure Properties Uses Of Ch2cl2
It can be calculated by adding the invididual molar mass of every atom that are composing the molecule CH4.
Molecular weight of dichloromethane. SGdensity of plastic generally depends on the molecular arrangement and the way in which they pack. A potential occupational carcinogen. Herein we design a guest molecule named BTO which is.
However the tannins that are slightly soluble in dichloromethane can be eliminated by converting it to their salts phenolic anions by adding sodium carbonate tannins are phenolic compounds of high molecular weight and being acidic in nature can be converted to salts by deprotonation of the -OH group which remain in the water. Propene is also used to produce isopropanol propan-2-ol acrylonitrile propylene oxide and epichlorohydrin. N-octanolwater No data available Autoignition Temperature 556 C 10328 F Decomposition Temperature No information available Viscosity 042 mPas 25C Molecular Formula C H2 Cl2 Molecular Weight 8493 10.
Support Customer Support Contact Us FAQ Safety Data Sheets SDS Certificates COACOO Quality Regulatory Calculators Apps Webinars. Molecular Geometry of Dichloromethane. 2003 5 618622.
17934 125 M HCl for GC derivatization LiChropur Expand. Molecular Weight Molar Mass. Specific Density Surface Tension Viscosity.
Uses of Ascorbic Acid. 8493 NTP 1992 Water Solubility. Name Molecular Formula CAS Mol.
10 micrograms per kilogram µgkg dry weight ranged from 75 to 102 percent. The industrial production of acrylic acid involves the catalytic partial oxidation of propene. On the rotary evaporator ethyl acetate also works well but is harder to remove dichloromethane is a poor choice and should be avoided if possible since it often forms nasty emulsions and complicates matters because it is heavier than water.
Fisher Chemical products include dry reagents acids solutions solvents and more. Aqueous and organic extracts of guava leaves have been demonstrated to have antibacterial activity due to an inhibitory effect against antibiotics-resistant clinical isolates of Staphylococcus aureus strains 1314Despite using the same diffusion method differences are noticed in their inhibition zones as shown in Table 1 probably due to. It fights against bacterial infections.
Dichloromethane anhydrous Revision Date 28-Nov-2019 Specific Gravity 133 Solubility No information available Partition coefficient. 720 mgm3 STEL listed under Dichloromethane 9. 132 gm 100gm 77F 25C pH.
Clear colorless liquid PHYSICAL STATE. 40 d 133 which is immiscible with water it is widely used as a solvent a paint stripper and for the removal of caffeine from coffee and tea. Page 1 of 1.
Infectious and Parasitic Diseases. The molecular structure of the guest is critical to the interaction between host and guest molecules. Generally amorphous polymers consisting of only hydrocarbons are lighter atoms thus their SG lies 0911 for example polyethylene polypropylene polystyrene etc A polymer consisting of larger atoms like chlorine fluorine etc has a larger specific gravity of more than 1.
10 to 50 mgmL at 70 F NTP 1992 Ionization Potential. Page 1 of 1. Delta-8-tetrahydrocannabinol delta-8-THC binds to the cannabinoid G-protein coupled receptor CB1 located in the central nervous system.
Relative standard deviations ranged from 3 to 13 percent. The resulting char and KOH were mixed with a weight ratio of 12 and heated to 700 C under nitrogen for 2 h to activate the material. This will contribute to having a.
2 Pick the size of your separatory sep funnel - usually 125 or 250-mL large scale reactions 1-10 g require 500-mL or 1-L sizes. 1-Propanol is a primary alcohol with the formula CH 3 CH 2 CH 2 OH and sometimes represented as PrOH or n-PrOHIt is a colorless liquid and an isomer of 2-propanolIt is formed naturally in small amounts during many fermentation processes and used as a solvent in the pharmaceutical industry mainly for resins and cellulose esters and sometimes as a disinfecting agent. Ascorbic Acid Structure C 6 H 8 O 6.
The molar mass of a substance also often called molecular mass or molecular weight although the definitions are not strictly identical but it is only sensitive in very defined areas is the weight of a defined amount of molecules of the substance a mole and is expressed in gmol. Noncombustible by if exposed to high temperatures may emit toxic chloride fumes. Acetyl coenzyme A lithium salt.
Orders Quick Order Custom Products. NIOSH 2016 AEGLs Acute Exposure Guideline Levels Interim AEGLs for Methylene chloride 75-09-2 Exposure Period AEGL-1 AEGL-2 AEGL-3. See More Methylene Chloride Properties Seller Information.
Hydrocarbon chain ie a low molecular weight compound containing an -OH -NH2 or -CO2H group or a larger molecule containing multiple polar groups eg. 133 SOLUBILITY IN WATER weight. It is used in the formation of collagen fibres in connective tissue fibrous tissue bones and teeth.
Dichloromethane is a member of the class of chloromethanes that is methane in which two of the hydrogens have been replaced by chlorineA dense non-flammible colourless liquid at room temperature bp. Used as a solvent and paint remover. Environmental Protection Agency procedures 40 CFR 136 Appendix B ranged from 06 to 34 µgkg dry weight.
A volumetric and viscosity study for the mixtures of 1-n-butyl-3-methylimidazolium tetrafluoroborate ionic liquid with acetonitrile dichloromethane 2-butanone and N N. Propene is also an intermediate in the one-step propane selective oxidation to acrylic acid. Ascorbic Acid Structure.
Mild sweet similar to Chloroform SPECIFIC GRAVITY water 10. The pyrolysis experiments of raw coal extract solutions and residual coal were carried out to investigate the effects of low molecular compounds on thermal decomposition behavior carbon structure of coal and light aromatics formation mechanism. Delta-8-Tetrahydrocannabinol is an analogue of tetrahydrocannabinol with potential antiemetic anxiolytic appetite-stimulating analgesic and neuroprotective activities.
Vapors are narcotic in high concentrations. In this paper dichloromethane was used to extract low molecular compounds from coal macromolecular framework. Send your message to.
Dichloromethane appears as a colorless liquid with a sweet penetrating ether-like odor. It is comparatively easy to understand the molecular geometry of a compound after knowing its Lewis structure and hybridization. Empirical Formula Hill Notation.
C 23 H 38 N 7 O 17 P 3 S xLi CAS No. 1132 eV NIOSH 2016 IDLH. The arrangement of the molecules in this compound is such that the Carbon atom is in the central atom one Hydrogen atom is on the upper topmost position and the other one is on the left side of the central atom.
Method detection limits MDLs calculated by using US. Shop the full line of Fisher Chemical products at Fisher Scientific. 80957 free acid basis Compare Product No.
It is used in the treatment of scurvy.
In the example provided above it can be observed that the coordination number of the central cobalt atom is 6 since it is bonded to 6 different nitrogen atoms. The molar mass of the NaOH compound is 40 gmol.

Cobalt Element In Periodic Table Atomic Number Atomic Mass
Investigating Molecular Structure - The Link Between Size and MW.

Molecular mass of cobalt. Lastly we emphasize that its molecular structure is critical and contains the active site even if support effects are important. Amedeo Avogadro is considered to be the number of units usually molecules or atoms in a substance that is proportional to its physical mass. A12 protons 12 neutrons.
The tabular chart we see on the periodic table is arranged. A student wants to create a model of a cobalt atom. It can be calculated by adding the invididual molar mass of every atom that are composing the molecule CH4.
98101106107109 c At constant Al level polymer molecular weight MW increases with the Clalkyl ratio in the aluminum alkyl chloride. The alkene called but-2-ene has two isomers which the chemical formula CH 3 CHCHCH. Some of these.
This mass proportion will be the same for any water molecule. Here we report a cobalt-phthalocyanine-based high-performance carbon dioxide reduction electrocatalyst material developed with a combined nanoscale and molecular approach. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structureThe chemical symbol for Hydrogen is H.
Which statement about the model is correct. Of or relating to chemistry. It has the mass composition of.
Enter a chemical formula. Molecular formulae indicate the simple numbers of each type of. Use the periodic table to determine which atom would have similar chemical properties to this atom.
Now molecular mass calculator add all masses of substances together. What is the molecular formula of a compound that has a molecular mass of 54 and the empirical formula C_2H_3. In a periodic table there are a total of 118 elements.
We would like to show you a description here but the site wont allow us. The atomic mass is the mass of an atom. Cobalt-60 a radioactive isotope of cobalt is an important source of gamma rays and is used to treat some forms of cancer and as a medical tracer.
In such cases the value of the coordination number of the central atom equals the total number of neighbouring. The polymer molecular weight increases with the monomer concentration 148099101106301 Figure 3 while it decreases with increasing cobalt concentration. Cobalt blue is an important part of artists palette and is used bu craft workers in porcelain pottery stained glass tiles and enamel jewellery.
Cyclohexanone is produced by the oxidation of cyclohexane in air typically using cobalt catalysts. To Determine the Molecular Formula. Which statement about the model is correct.
Of or relating to chemical weapons. Atomic mass is also referred to as atomic weight but the term mass is more accurate For instance it can be determined experimentally that neon consists of three isotopes. In related terms another unit of mass often used is Dalton Da or unified atomic mass unit u when describing atomic masses and molecular masses.
The molar mass of a compound is 11938 g mol 1. Molecular weight calculator is an online tool to calculate atomic mass and molecular mass. It is possible to determine molecular size and MW distributions for a polymer using GPCSEC and the aforementioned measurement methods.
Did you mean to find the molecular weight of one of these similar formulas. More information on molar mass and molecular. This compound is a cobalt complex.
An atom has 3 protons 4 neutrons and 3 electrons. The explanation of structural characteristics depends on the use of these data along with. The atomic mass is carried by the atomic nucleus which occupies only about 10-12 of the total volume of the atom or less but it contains all the.
Other cobalt macrocycle complexes such as cobalt chlorin and. Cobalt has a mass number of 59 and an atomic number of 27. On the nanoscale cobalt.
Atomic Mass of Cobalt. Neon-20 with 10 protons and 10 neutrons in its. Cobalt has a mass number of 59 and an atomic number of 27.
Cobalt chloride hexahydrate is a hydrate of cobalt chloride containing cobalt in 2 oxidation state chloride and water moieties in the ratio 126. The molecular weight calculator uses the molar mass of each element in the formula you specify to determine the molecular weight of the total compound. The molar mass of a substance also often called molecular mass or molecular weight although the definitions are not strictly identical but it is only sensitive in very defined areas is the weight of a defined amount of molecules of the substance a mole and is expressed in gmol.
With a standard atomic weight of circa 1008 hydrogen is the lightest element on the periodic table. How many elements are in the periodic table. The radioactive isotopes cobalt-60 is used in medical treatment and also to irradiate food in order to preserve the food and protect the consumer.
Molecular size like MW is a distributed parameter. Use the periodic table to determine which atom would have similar chemical properties to this atom. 100000 Similar chemical formulas.
Cobalt-60 has a half-life of 527 years and decays into nickel-60 through beta decay. This process co-forms cyclohexanol and this mixture called KA Oil for ketone-alcohol oil is the main feedstock for the production of adipic acid. Molecular mass or molar mass are used in stoichiometry calculations in chemistry.
Also important in this field is Avogadros number N. An atom has 3 protons 4 neutrons and 3 electrons. A12 protons 12 neutrons.
The molar mass of the compound is unknown. A student wants to create a model of a cobalt atom. Of or relating to the properties or actions of chemicals.
Cobalt compounds have been used for centuries to color porcelain glass pottery tile and enamel. It has a role as. Chemical names in answer to limitations of chemical formulae.
CO2 Co2 Calculate the molecular weight of a chemical compound. It has the mass composition of 678 of hydrogen 3142 of nitrogen 3976 of chlorine and 2204 of cobalt. A drug especially an illicit or addictive one.
We see that 94 of the mass of a water molecule is accounted for by oxygen and the remaining 6 is the mass of hydrogen. Question abc31 Cobalt a transition metal can have an oxidation number of either 2 or 3. C 6 H 12 O 2 CH 2 5 CO H 2 O.
The atomic mass or relative isotopic mass refers to the mass of a single particle and therefore is tied to a certain specific isotope of an element. Atomic mass of Cobalt is 589332 u. A substance with a distinct molecular composition that is produced by or used in a chemical process.
16 g mol 23 g mol 1 g mol 16 g mol 40 g mol 23 g mol 1 g mol 40 g mole. Molecular mass calculator helps a user to complete his work in a given time without any errors. For crystals the bonds are not as clear in their solid state structures.
The atomic mass is a weighted average of all of the isotopes of that element in which the mass of each isotope is multiplied by the abundance of that particular isotope. 80102104 The effect of the concentration of Al seems to depend on the type of aluminum. Note that all formulas are case-sensitive.
Its monatomic form H is the most abundant chemical substance in the Universe constituting roughly 75 of all baryonic mass. It is defined to be 112 of the mass of one atom of carbon-12 and in older works is also abbreviated as amu. The units used are grams per mole because the molecular weight is usually expressed as the mass of one mole out of a certain substance.
Browse the list of common chemical compounds.
In the nuclear industry chlorine trifluoride is used to prepare uranium hexafluoride a volatile compound of uranium used in the separation of uranium isotopes. Example Exercise 91 Atomic Mass and Avogadros Number.
Elemental nitrogen is a colorless odorless tasteless and mostly inert diatomic gas at standard conditions constituting 7808 by volume of Earths atmosphere.
Molecular mass of nitrogen trifluoride. Properties of Nitrogen trifluoride. Molecular geometry of NF3. The mass of Avogadros number of atoms is the atomic mass expressed in grams.
Cu 6355 amu Hg 20059 amu S 3207 amu and He 400 amu. It finds increasing use within the manufacturing of flat-panel displays photovoltaics LEDs and other microelectronics. It is Corrosive to tissue.
Some boron compounds such as boron nitrite are completely water insoluble. Write the Lewis structure for a molecule of the compound. It is possible to draw a structure with a double bond between a boron atom and a fluorine atom in BF 3 satisfying the octet rule but experimental evidence indicates the bond lengths are closer to that expected for.
Percent yield represents the ratio between what is experimentally obtained and what is theoretically calculated multiplied by 100. Boron salts are generally well water soluble. If a gas behaves ideally both a and b are zero.
We would like to show you a description here but the site wont allow us. Nitrogen trifluoride or NF3 is a nitrogen halide compound that is slightly water-soluble. Briefly 4 mL of 05 M sodium hydroxide in methanol were added to 1 mL oil sample the equipment was closed and heated for 20 min under nitrogen.
The reac-tion was evaporated in vacuo quenched with water 40 mL adjusted to pH 9 with Na 2 CO 3 extracted with CHCl 3 2 x 100 mL dried over anhydrous Na 2 SO 4 filtered and evaporated in vacuo to a semi-crystalline mass. It is usually referred to as the van der Waals equation of state. Further 5 mL of 15 boron trifluoride freshly prepared.
For example in the Lewis structures of beryllium dihydride BeH 2 and boron trifluoride BF 3 the beryllium and boron atoms each have only four and six electrons respectively. Van der Waals suggested a modification to take into account molecular size and molecular interaction forces. Yield actual yieldtheoretical yield 100 So lets say you want to do an experiment in the lab.
Chlorine trifluoride is prepared by the reaction latextextCl_2g3textF_2glongrightarrow2textClF_3glatex. A compound with a molar mass of about 42 gmol contains 857 carbon and 143 hydrogen by mass. Atoms of a Cu 6355 g c S 3207 g b Hg 20059 g d.
Its refractive index is 10004. The atomic mass of each element is listed below the symbol of the element in the periodic table. Nitrogen trifluoride NF 3 is an inorganic colorless non-flammable toxic gas with a slightly musty odor.
The human body contains about 3 nitrogen by mass the fourth most abundant element in the body after oxygen carbon. The constants a and b are called van der Waals constants. They have positive values and are characteristic of the individual gas.
Learn about Lewis Acids and Bases Examples Applications Reactions and FAQs Visit BYJUS for detailed explanations. Lewis Acids and Bases -Lewis Acids are the chemical species which have empty orbitals and are able to accept electron pairs from Lewis bases. Two arrangements of atoms are possible for a compound with a molar mass of about 45 gmol that contains 522 C 131 H and 347 O by mass.
Air - Molecular Weight and Composition - Dry air is a mixture of gases where the average molecular weight or molar mass can be calculated by adding the weight of each component. Flask containing MeOH 50 mL and boron trifluoride diethyl etherate 10 g 705 mmol and refluxed for 2 hours. What is the empirical formula of a compound that contains 494 K 203 S and 303 O by mass.
Atomic or molecular chemical species having a highly localized HOMO The Highest Occupied Molecular Orbital act as Lewis bases. P anV 2 Vn - b RT. Boron trifluoride is the least water soluble boron compound with a water solubility of 24 gL.
This method with usage of acid catalyst boron trifluoride is supported by the fact that basic catalysis does not convert free fatty acids especially in oils as can be concluded from the paper. It can be calculated by adding the invididual molar mass of every atom that are composing the molecule CH4. Many olefin polymerization reactions use BF3 as an initiator in conjunction with a proton donor such as.
Boron trifluoride is commonly used as a catalyst for Friedel-Crafts alkylation reactions. At a temperature of 25 degrees Celsius the solubility of boric acid in. NF3 has a molar mass of around 71002 gmol and a density of 3003 kgm3.
Here we present a modified version of the Medusa. D K2SO3 Lithium and nitrogen react in a combination reaction to produce lithium nitride. It has a low dipole moment.
Nitrogen trifluoride NF 3 first prepared in 1928 is a colourless and odourless gas that is thermodynamically stable and most readily produced by the electrolysis of molten ammonium fluoride dissolved in anhydrous hydrogen fluoride. Write the equation that relates the rate expressions for this reaction in terms of the. The molar mass of a substance also often called molecular mass or molecular weight although the definitions are not strictly identical but it is only sensitive in very defined areas is the weight of a defined amount of molecules of the substance a mole and is expressed in gmol.
Write the Lewis structures for the two molecules. The density of NF3 is 3003 kgm 3. Nitrogen trifluoride is also an extremely strong and long-lived greenhouse gasIts atmospheric burden exceeded 2 parts per trillion during 2019 and has doubled every.
Air - Thermophysical Properties - Thermal properties of air at different temperatures - density viscosity critical temperature and pressure triple point enthalpi and entropi thermal conductivity and diffusivity. Nitrogen ˈ n aɪ t r ɵ dʒ ɨ n ny-trə-jin is a chemical element that has the symbol N atomic number of 7 and atomic mass 1400674 u. It has a molar mass of 7100 gmol.
Why is boron present in water. You want to measure how much water is produced when 120 g of glucose C_6H_12O_6 is burned with enough oxygen. The solubility of H 3 BO 3 in water is temperature-dependent.
This potent greenhouse gas has a rising atmospheric abundance due to its emission from a growing number of manufacturing processes and an expanding end-use market. Like carbon tetrafluoride it is not. NF3 boiling point is 12906 C and melting point is 20715 C.
Electron geometry of. Under standard conditions for temperature and pressure STP boric acid exists as a white crystalline solid that is fairly soluble in water. Its noticeable characteristics include being colorless and carrying a musty or moldy odor.
It also is used to catalyze the cleavage of ethers to alcohols to catalyze esterification reactions and in the nitration and sulfonation of aromatic compounds. Boric acid has a water solubility of 57 gL borax of 252 gL and boron trioxide of 22 gL. We present an analytical method for the in situ measurement of atmospheric nitrogen trifluoride NF3 an anthropogenic gas with a 100-year global warming potential of over 16000.
Boron Trifluoride 330 Bromine Pentafluoride 670 Bromine Trifluoride 670 1-3 Butadiene 510 Butane 510 Butenes 510 Carbon Dioxide 320 Carbon Monoxide 350 Carbonyl Fluoride 660 Carbonyl Sulfide 330 Chlorine 660 Chlorine Trifluoride 670 Chlorotrifluoroethylene 660 Cyanogen 660 Cyclopropane 510 Deuterium 350 11-Difluoroethylene 350. Molecular Weight Molar Mass.
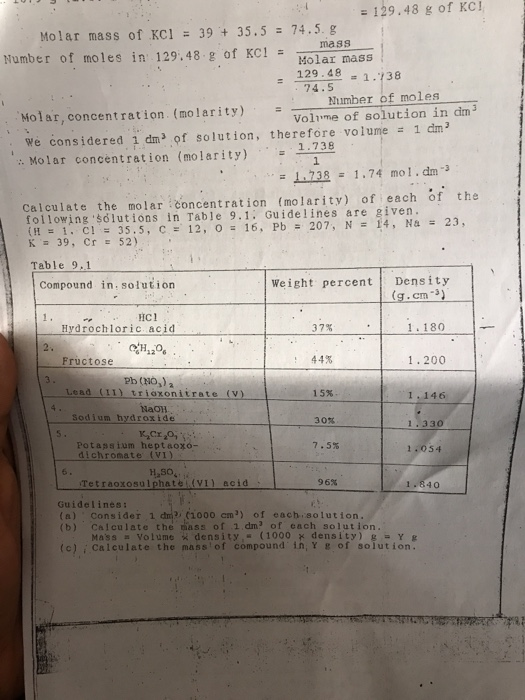
Use this page to learn how to convert between moles KCl and gram. There are many different ways of expressing the concentration of a given solution.

Molar Mass Molecular Weight Of Kcl Potassium Chloride Youtube
KCl is used as a fertilizer in medicine in scientific applications and.
Molecular weight of kcl. First we assume a total mass of 1000 g although any mass could be assumed. Finally current uses and potential applications of thermophilic and hyperthermophilic enzymes as research reagents and as catalysts for industrial. Binding of AGES to ECM proteins creates.
Molecular weight of KCl or grams This compound is also known as Potassium Chloride. We can then. 390983 35453 Percent composition by element.
Protocols Manuals Usage. Stanimila Nikolova PhD. Molecular weight of MgNO 3 2.
Magnesium nitrate Structure MgNO32. MgNO 3 2 Uses Magnesium nitrate Magnesium nitrate is used as a dehydrating agent to prepare concentrated. Hydrochloric acid concentration in the stomach is.
The molecular mechanisms involved in protein thermostabilization are discussed including ion pairs hydrogen bonds hydrophobic interactions disulfide bridges packing decrease of the entropy of unfolding and intersubunit interactions. Chromatographic techniques will then follow namely ion exchange or gel. Monarch Plasmid Miniprep Kit.
Monarch PCR DNA Cleanup Kit 5 μg Recombinant Albumin Molecular Biology Grade Animal-free References. The molecular weight of urea is 6016 gmol and the molecular weight of cinnamic acid is 14816 gmol. A Laboratory Manual Third Edition.
A Laboratory Manual Third Edition 1982. Dialysis is a relatively slow method taking up to 12 hours to be completed. The mass of one mole of carbon-12 atoms is exactly 12 grams which is its molar mass it is exactly 12 grams per mole.
This compound is also known as Potassium Chloride. Contact China Manufactory TYPUGROUPCOLTD for the product high grade KCL potassium chloride food grade 99. A the mass of KCl NaCl is 2370 g b the mass of NaCl LiCl is 1290 g c the mass of AgCl precipitated from the mixture is 6435 g.
A Laboratory Manual Third Edition. Stomach acid is composed of hydrochloric acid HCl potassium chloride KCl and sodium chloride NaCl. Solvent - a substance in a solution that is present in the largest amount.
Dialysis is often used to remove salt from protein solutions after they have been separated by salting-out and to change buffers. M 6 Am 6 Am modification is. Here we report the incorporation of Cu in an organic molecular solid 34910-perylenetetracarboxylic dianhydride PTCDA for the direct reduction of NO 3 into NH 3 with a.
Potassium chloride crystal optic disc 13mm x 2mm unpolished. The weight of the fusion protein can therefore be approximated. NBS Molecular Training Class April 25 2016.
The molecular weight of the GSTEGFP fusion protein can be estimated assuming the average weight per amino acid is equal to 114 Da. In eukaryotes N6-methyladenosine m 6 A and the cap-adjacent N62Odimethyladenosine m 6 A m are highly abundant and reversible mRNA modifications 1 2. If you had a 10 molar solution 10 M you would have to put 5844 g of salt in 10 liter of solution.
This technique which leads to an aqueous concentrate of enzyme is faster and easier to implement than the two-step processes of precipitation and dialysis. A short summary of this paper. 1 mole is equal to 1 moles KCl or 745513 grams.
The plastic container is made from a multilayered film specifically developed for parenteral drugs. Molar mass of KCl 745513 gmol. Monarch DNA Gel Extraction Kit.
Atomic Mass of Atoms. 100 mM KCl 01 mM EDTA 50 Glycerol pH 8 25C. Potassium chloride 0000027M Conductivity Standard.
Equiv weight molecular weight KCl divided by valence Since valence of KCl 1 Equiv weight 745 1 Step 3. How many moles of NaCl would you have in 100 mL of this solution. Potassium chloride for molecular biology 990.
KCl molecular weight. Boiling point of Magnesium nitrate. Solute - a substance in a solution that is present in the smallest amount.
Latextext moles KCl 536 textg times frac1 text moles745 textg 00719 text moles KCllatex. Full PDF Package Download Full PDF Package. Solubility - ability of the solute to dissolve in.
It contains no plasticizers and exhibits virtually no leachables. 23 gcm 3 anhydrous Melting point of Magnesium nitrate. The solid dissolves readily in water and its solutions have a salt-like tastePotassium chloride can be obtained from ancient dried lake deposits.
Weight by volume percent wv tells you the mass of solute in. 1 mEq KCl 11000 x 745 g 00745 g 745 mg. Chat now for more business.
ISA ionic strength adjustment solution. 1 The mass of KCl in the mixture will be our unknown x This leads a description of the mass of NaCl present and the mass of LiCl present. 14832 gmol anhydrous Density of Magnesium nitrate.
The molar mass of a substance also often called molecular mass or molecular weight although the definitions are not strictly identical but it is only sensitive in very defined areas is the weight of a defined amount of molecules of the substance a mole and is expressed in gmol. It also eliminates stomach bacteria and viruses thereby protecting your body from infection. Some of the most common include molarity weight by volume volume by volume and weight by weight.
The EXCEL Container is Latex-free PVC-free and DEHP-free. The molecular mass is calculated from gmol units. Low molecular weight solutes flow through the bag but the large molecular weight protein molecules remain in the bag.
52445 Calculate. Weight gain is another environmental issue in diabetes that results in a higher beta-cell load. The molecular weight of NaCl is 5844 gramsmole.
Ultrafiltration is another technique used to separate enzymes provided the molecular weight cutoff of the membrane is compatible with that of the target enzyme. 1 M KCl 1 M KCl. Note that rounding errors may occur so always check the results.
The SI base unit for amount of substance is the mole. CH 4 1 mole C atoms 4. Magnesium nitrate Structure MgNO 3 2.
The Product Summary Sheet or. 36 Full PDFs related to this paper. The solution contact layer is a rubberized copolymer of ethylene and propylene.
Potassium chloride KCl or potassium salt is a metal halide salt composed of potassium and chlorineIt is odorless and has a white or colorless vitreous crystal appearance. It is therefore most frequently used in the laboratory. Hydrochloric acid helps the body break down foods such as calcium digest and drink them.
Convert grams KCl to moles or moles KCl to grams. Glycation disrupts molecular conformation and alters protein function. Lets Talk About Solutions Solution - a homogeneous mixture of two or more substances.
The gst gene is 660 Da. AGEs have crucial role in diabetes related cardiovascular and renal complications. There are 174 additional nucleotides between gst and egfp encoding 58 amino acids.
This means that we have 500 g of urea and 500 g of cinnamic acid. Molecular weight of NO2 46 g mol¹- Molecular weight of CO2 44g mol¹-Thus the compound which has a lower molecular mass will diffuse at a faster rate than the heavier compound. Potassium chloride meets EP USP testing specifications.
It can be calculated by adding the invididual molar mass of every atom that are composing the molecule CH4. The egfp gene is 720 Da. Type in your own numbers in the form to convert the units.
Determine the mole fraction of LiCl in the mixture. AGEs can bind to intracellular and extracellular proteins and alter tissue functions. Molecular Quality Improvement Program.
The monosaccharides glucose galactose and fructose all have the same molecular formula C 6 H 12 O 6 but we can see from Figure 3 that the atoms are bonded together differently. The distinguishing features of ketamine anesthesia are preserved breathing and airway reflexes stimulated heart function with increased blood pressure and moderate bronchodilation.
Dextromethorphan Hydrobromide is the hydrobromide salt form of dextromethorphan a synthetic methylated dextrorotary analogue of levorphanol a substance related.

Molecular weight of codeine. Substances in the DEA Schedule II have a high potential for abuse which may lead to severe psychological or physical dependence. The sum of the weights of the atoms that make up the compound. Tramadol and codeine are both opioids or opiates that are used to control pain.
The phosphate salt of codeine occurs as white needle-shaped crystals or white crystalline powder. Most of these variants dont affect how people respond to the drug. 65 to 135.
From feelings of pain relief to relaxation pleasure and contentment. 90 excreted by kidneys. Opioids interact with opioid receptors in the brain and elicit a range of responses within the body.
Its molecular formula is C 18 H 21 NO 3 H 3 PO 4 ½H 2 O and it has the following chemical structure. Joel Block Editor-in-Chief of Osteoarthritis and Cartilage would like to recognise the following reviewers and thank them for their contribution to the journal. Empirical Formula Hill Notation.
Significant accumulation of metabolites. Each soluble tablet contains 30 mg 0074 mmol or 60 mg 015 mmol of codeine phosphate. The salt form has different physical and chemical properties from the base form while retaining the same pharmacological properties.
About a fifth of total plant species are believed to produce them. Blanco Jeroen Geurts Tariq M Haqqi Satoshi Kubota. Examples of alkaloids include morphine codeine coniine quinine scopolamine hyoscamine atropine caffeine sangunarine berberine etc.
However genetic variation in this enzyme can result in too little or too much enzymatic action resulting in non-conversion or too little absorption hence no therapeutic effect. Page 1 of 1. Its empirical formula is C 18 H 21 NO 3 and its molecular weight is 29936.
Codeine phosphate is freely soluble in water and slightly soluble in alcohol. Each tablet contains 15 30 or 60 mg of codeine sulfate and the following inactive ingredients. A small amount of ingested codeine is converted to morphine in the body.
We call these elements macronutrients and they account for about 99 of the dry weight of cells. Acetyl coenzyme A lithium salt. Clinical Therapeutics provides peer-reviewed rapid publication of recent developments in drug and other therapies as well as in diagnostics pharmacoeconomics health policy treatment outcomes and innovations in drug and biologics research.
GFR 50 mLmin. They have much lower boiling points than do alcohols with similar molecular weights. 80957 free acid basis Compare Product No.
Codeine is also used as a cough medication. Codeine a potent pain-relieving drug is the methyl ether of morphine. The extract is marketed as a dietary supplement to regulate blood sugar cholesterol blood pressure and for weight loss and cancer prevention.
Many vary by only a single difference in their DNA sequence. Hydrocodone is a DEA Schedule II controlled substance. The activity of isoflavones is subsequent to the conversion of the glucosides into the main bioactive aglycones glycitein daidzein genistein and equol through the action of the intestinal β-glucosidase of microbiome Setchell et al 2002.
In addition to decreasing pain codeine also causes sedation drowsiness and respiratory. Clinical Therapeutics is read by a large. Codeine converts to morphine using the CYP 2D6 enzyme.
In addition to the pharmacodynamics outlined herein many synthetic opioids have additional mechanisms of action such as noradrenergic reuptake blockade and inhibition of n-methyl-D-aspartase NMDA. The name of the salt form contains the name of the base form ie cocaine hydrochloride. Active constituents include the polyphenol epigallocatechin-3-gallate EGCG caffeine and theanine.
These include age comorbidities disease severity gender genetics and weight all of which may positively or negatively affect the drug response. Green tea is a beverage derived from the unfermented leaves of a plant native to Asia. Its molecular weight is 4064.
CID 260 Hydrogen bromide CID 962 Water CID 5360696 Dextromethorphan Dates. C 4 H 10 O MW 74 is 118 C 244 F. 17934 125 M HCl for GC derivatization LiChropur Expand.
Please read our Terms Conditions and Privacy Policy for information about. The molecular weight is 40637. Codeine is a weak narcotic pain reliever and cough suppressant that is similar to morphine and hydrocodone.
Frogs for example Bufo marinus contain large quantities of. For example the boiling point of diethyl ether C 4 H 10 O molecular weight MW 74 is 35 C 95 F but the boiling point of 1-butanol or n-butyl alcohol. CID 5360696 Dextromethorphan Component Compounds.
Kidney Disease Program. Ketamine is a medication primarily used for induction and maintenance of anesthesiaIt induces dissociative anesthesia a trance-like state providing pain relief sedation and amnesia. Start at 50 of dose with increased interval of dosing and carefully titrate MW 299.
Its structure is as follows. Codeine increases tolerance to pain reducing existing discomfort. They are mainly produced by plants and animals for defense.
C 23 H 38 N 7 O 17 P 3 S xLi CAS No. For example a patient may have a genetic variation that makes the drug stay in the body longer than normal causing serious adverse effects or another person. This website uses cookies to help provide you with the best possible online experience.
Its structure is as follows. Both codeine and tramadol are less potent and therefore less addictive than other opiate medications like morphine. The product of a reaction of an acidic substance and an alkaline substance.
The majority of them are not absorbed as such across the enterocytes owing to their molecular weight and high hydrophilicity. In fact the boiling points of. Glucose galactose and fructose have the same chemical formula C 6 H 12 O 6 but these structural.
Due to partial. Page 1 of 1. Start at 75 dose with increased interval and carefully titrate due to accumulation of metabolites.
The chemical name of codeine phosphate is 78-Didehydro-45alpha-epoxy-3-methoxy-17-methylmorphinan-6alpha-ol phosphate 11salt hemihydrate and has the empirical formula of C18H21NO3H3PO412H20. For example it converts the painkiller codeine into its active form morphine. Fentanyl is part of a group of drugs known as opioids.
Thank you to 2020s top reviewers. Colloidal silicon dioxide microcrystalline cellulose pregelatinized starch and. Vd 26 Lkg.
Orders Quick Order Custom Products. Support Customer Support Contact Us FAQ Safety Data Sheets SDS Certificates COACOO Quality Regulatory Calculators Apps Webinars. Alkaloids are nitrogenous compounds of low molecular weight.
Others have larger changes. Tobias Bäuerle Francisco J. In addition Clinical Therapeutics features updates on specific topics collated by expert Topic Editors.
Drug Enforcement Administration DEA Hydrocodone is a semisynthetic moderately potent orally available opioid that in combination with. One liver enzyme known as CYP2D6 acts on a quarter of all prescription drugs. Read more about side effects dosage and pregnancy safety information.
There are more than 160 versions of the CYP2D6 gene.