If a gas is given off note the color and odor of the gas. 235-543-4 CAS no.

10099 74 8 Lead Ii Nitrate 124 00612 128 00615 Detail Information Common Chemicals Lab Tools Laboratory Chemicals Fujifilm Wako Chemicals U S A Corporation
Rutherford 104 267 Db.
Lead nitrate hydrate. Lead hydroxide nitrate EC List no. Radon 86 222 Fr. C 27 H 42 N 7 Na 2 O 20 P 3 S xH 2 O.
Supercooling brings extra cooling capacity to release latent heat and phase separation may lead to the failure of inorganic hydrate to maintain its phase change characteristics in long-term thermal cycle. Bori 107 270 Hs. Sign up and be the first to learn about new products for your laboratory and product specials.
Chassius 108 277 Mt. The added word begins with a Greek prefix denoting the number of water molecules see Table 5 and ends with hydrate For example the anhydrous compound copperII sulfate also exists as a hydrate containing five water molecules and named copperII sulfate pentahydrate. Cooling systems for.
Health based guideline by the WHO. Al 2 SO 4 3 3 BaNO 3 2 2 AlNO 3 3 3 BaSO 4. Greek prefixes are attached to the word hydrate to indicate the number of water molecules per formula unit for the compound eg BaOH 2 8H 2 O.
Guidance for Issuing and Rescinding Drinking Water Avoidance Advisories in Emergency Situations. The Report Titled Global and Chinese Holmium Nitrate Market 2016-2021 Industry Research Report is a efficient and comprehensive study on. Acetyl coenzyme A sodium salt.
Copernicius 112 285 Nh. PETG Chemical Resistance Chart. Francius 87 223 Ra.
It may also lead to an increased cancer risk due to the formation of the carcinogen substances N-nitrosamines from ingested nitrate and nitrite 44. Dubny 105 268 Sg. Guidance for Issuing and Rescinding Boil Water Advisories in Canadian Drinking Water Supplies.
Press release - QYResearch Inc. The crystallized product is a hydrate Na 2 SO 4 10H 2 O commonly known as Glaubers salt. Incompatible with combustible material strong reducing agents most common metals organic material.
Storfjordrenna is located south of the Svalbard Archipelago in the red box on the regional map. Guidance on Chloral Hydrate in Drinking Water. Urea is essentially the waste produced when the body metabolizes protein.
65997-18-4 Petrolatum petroleum oxidized lead salt EC List no. Documents for Public Comment - Water. Cobalt chloride as an example of a solid hydrate.
Common Covalent Binary Inorganic Compounds of atoms Prefix element closest to fluorine goes on rightCommon Examples 1 Mono H 2 Hydrogen N 2 Nitrogen 2 Di O 2 Oxygen NH 3 Ammonia 3 Tri O 3 Ozone NO Nitrogen monoxide Nitric Oxide 4 Tetra H 2O Water Dihydrogen Monoxide NO 2 Nitrogen dioxide 5 Penta F 2 Fluorine N 2O Dinitrogen monoxide Nitrous oxide 6 Hexa HF Hydrogen fluoride N. Meters below sea level. It acts by dissolving the part of the silver salts coated onto film which remain.
Ferric nitrate nonahydrate 7782-61-8. Seaborgy 106 269 Bh. Notice that the water molecules shown.
Radium 88 226 Rf. Because the temperature of building heating is usually below 100 C it is valuable to study hydrated salt PCMs. DL-3-Hydroxy-3-methylglutaryl coenzyme A sodium salt hydrate.
We are a leading supplier to the global Life Science industry with solutions and services for research biotechnology development and production and pharmaceutical drug therapy development and production. 8 water molecules octahydrate. Acetaldehyde acetic acid.
Box 550 1560 Industry Road Hatfield PA 19440 Phone. View our virtual catalog. It is not only produced by humans but also by many other mammals as well as.
Anhydrous cobalt chloride upper left and its crystal lattice structure lower left compared with cobalt chloride hexahydrate upper right and its crystal lattice lower right. 7782-61-8CAS DataBase Reference EPA化学物质信息. Guidance on Potassium from Water Softeners.
2 PbNO 3 2 s heat 2 PbOs O 2 g 4 NO 2 g brown CaCO 3 s heat CaOs CO 2 g colorless odorless CaSO 3 s heat CaOs SO 2 g colorless. Polonium 84 209 At. C 12 H 8 Cl 6.
Sodium thiosulfate sodium hyposulfite Na 2 S 2 O 3 is used by photographers to fix developed negatives and prints. May be light sensitive. Temperatures substantially greater than the normal range can lead to severe stress or even death.
Strong oxidizer - contact with combustible material may lead to fire. Methane clathrate CH 4 575H 2 O or 4CH 4 23H 2 O also called methane hydrate hydromethane methane ice fire ice natural gas hydrate or gas hydrate is a solid clathrate compound more specifically a clathrate hydrate in which a large amount of methane is trapped within a crystal structure of water forming a solid similar to ice. C 14 H 20 Cl N O 2.
Guidelines and Technical Documents Documents for Public Comment. Maytnerium 109 278 Ds. A safety data sheet material safety data sheet or product safety data sheet are documents that list information relating to occupational safety and health for the use of various substances and products.
Washing soda is the common name for a hydrate of sodium carbonate containing 10 water molecules. When the chemical formula for a hydrated ionic compound is written the formula for the ionic compound is separated from the waters of hydration by a centered dot. Significant quantities were used in the manufacture of tetraethyl lead as a gasoline additive.
X-ray 111 282 Cn. Darmstadty 110 281 Rg. C 12 H 8 Cl 6 O.
Through comparison and selection magnesium nitrate hexahydrate MNH with. 12268-84-7 maleatotrioxotetralead EC List no. Aluminium nitrate is a strong oxidizing agent.
The nitrate carbonate and sulfite ions may decompose as illustrated by the reactions. - Holmium carbonate hydrate Market 2021- Analysis And In-Depth Research On Market Size Trends Emerging Growth Factors And Forecast To 2027. UreaNH 2 2 CO is a colorless organic chemical compound also known by the International Nonproprietary Name rINN carbamide as established by the World Health OrganizationIt is highly soluble in water and has a pKa close to zero.
C 10 H 12 N 2 O 3 S. Aluminium nitrate may also be prepared a metathesis reaction between aluminium sulfate and a nitrate salt with a suitable cation such as barium strontium calcium silver or lead. Empirical Formula Hill Notation.
C 7 H 14 N 2 O 4 S. This is probably water indicating that the compound is a hydrate. Astat 85 210 Rn.
Originally thought to occur only in the outer. 266-047-6 CAS no. EC List no.
C 8 H 14 Cl N 5. 95562 anhydrous basis Compare Product No. Gas hydrate mound GC.
Apply a cotton applicator dipped in solutionointment on the affected area 2-3 times per week for 2-3 weeks. A 100-mgL standard solution would be appropriate for medium- and high-range tests.

Lead Ii Nitrate Solution Acc Ph Eur Chapter 4 2 2 Ep Ref No 3003100 Th Geyer
3 10 grams of potassium fluoride is dissolved to make 010 mL of solution.

Lead nitrate solution. 2 is commonly synthesized via a precipitation reaction between potassium iodide KI and leadII nitrate Pb NO 3 2 in water solution. Ammonium nitrate N2H4O3 is one such compound and has an NPK rating of 34-0-0. Lead Reference Standard Solution.
If your skin comes into contact with silver nitrate solution immediately wash thoroughly with soap and water. We regularly produce chemical solutions to specifications designed by government and regulatory bodies commercial and trade associations and the specific needs of individual users and businesses. Take a sample of water from your aquarium add a couple of drops of the solution included in the test kit and watch the water change color.
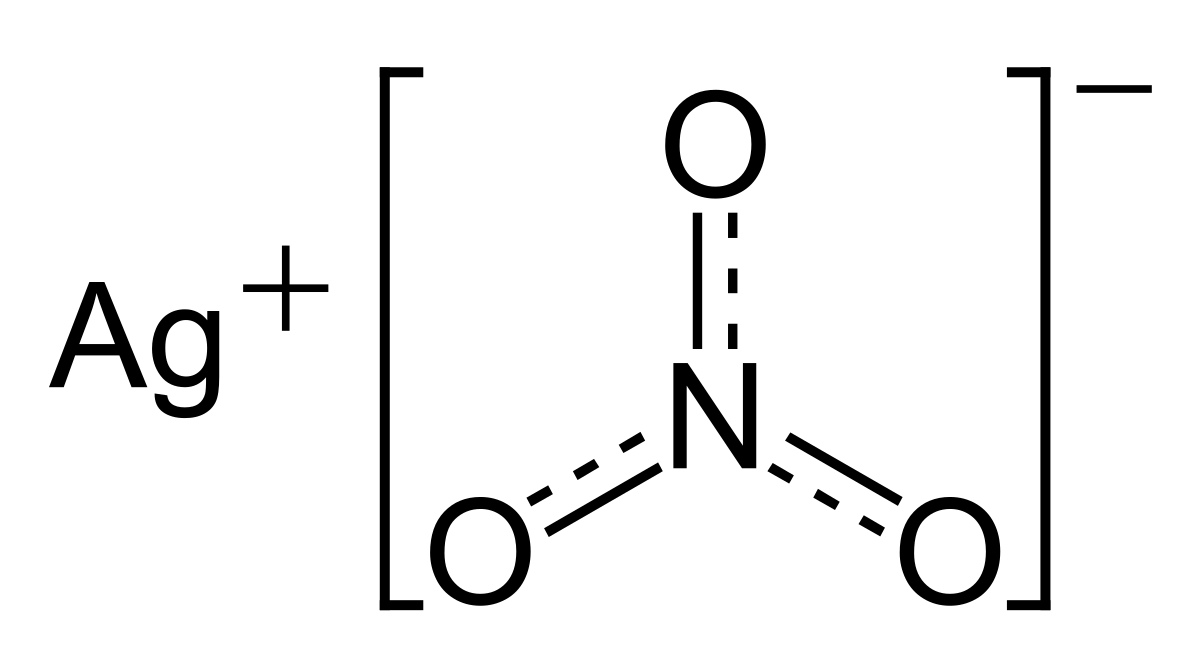
Nitrate Ion OF2 Oxygen Difluoride P2O3 Diphosphorus Trioxide P2O5 Phosphorus Pentoxide P4O10 Phophorus Pentoxide P4S3 Phosphorus Sesquisulfide PbC2H3O22 LeadII Acetate PbC2H3O24 LeadIV Acetate PbNO32 LeadII Nitrate PbCl2 LeadII Chloride PbCl4 LeadIV Chloride PbCrO4 LeadII Chromate PbI2 LeadII Iodide PbO LeadII Oxide PbO2. LeadII nitrate is an inorganic compound with the chemical formula PbNO 3 2. It commonly occurs as a colourless crystal or white powder and unlike most other leadII salts is soluble in water.
Note that acetate is often abbreviated OAC. For example suppose you have a near-boiling solution of potassium nitrate in water. Learn about the health effects of lead who is at risk how to test for lead in paint or other areas of your home how to find or become a lead-safe certified firm and more about the Lead Renovation Repair and Painting RRP rule.
Avoid getting silver nitrate solution on your skin. If you get silver nitrate solution in your eyes flush with water for 15 minutes and seek medical help. The terms Nitrite and Nitrate are frequently used interchangeably owing to their similar pronunciations and spellings.
A few drops of silver nitrate solution are added to the solution and a pale yellow precipitate forms. LeadII nitrate 99999 trace metal basis EINECS 233-245-9. Ninety percent of synthetic urea produced now is for fertilizers.
But dont worry too much this solution is. Respectively these are the nitrate acetate nitrite chlorate and perchlorate ions. Silver nitrate is a strong oxidizer but the solution is fairly dilute.
Use a nitrate nitrogen standard solution of appropriate strength for the range in which you are working. PbNO 3 2 2 KI PbI 2 2 KNO 3. At all temperatures above that marked on the graph about 57C 100 g of water will dissolve more than 100 g of potassium nitrate.
Sodic soils tend to have poor structure with unfavorable physical properties such as poor water infiltration and air exchange which can. 10 MnNO 33 is manganese III nitrate 11 FePO 4 is iron III phosphate 12 CoCO 3 is cobalt II carbonate Name to formula problems. Below are two of the best nitrate test kits around.
Irrigating from salt-impacted wells or saline industrial water may lead to the formation of saline soils. At 57C you hit the. Known since the Middle Ages by the name plumbum dulce the production of leadII nitrate from either metallic lead or lead oxide in nitric acid was small-scale for direct use in making other lead.
Then since 02M is such a higher concentration than x we can safely rewrite it as 71. Although nitrates are stable dietary nitrate is converted to nitrite through a non-enzymatic process and nitric oxide NO by symbiotic bacteria in the oral cavity and stomach therefore performing. Now let the solution cool.
These are mixtures. Nitrites are known to hold one nitrogen atom along. High nitrate levels can lead to disease.
Nitrate NO 3 and nitrite NO 2 widely exist in water soil air and plants The main source of absorbed nitrate in the body is food with green vegetables contributing the major portion. Other soluble salts containing leadII and iodide can be used instead for example. LeadII nitrate 99 for analysis.
API Nitrate Test Kit. The Quest Learning and Assessment System uses an extensive knowledge bank of over 60000 questions and answers covering Math Biology Chemistry Computer Science Physical Science and Physics. For example if our lead iodide compound was being dissolved in a solution with 02 M lead chloride PbCl 2 we would rewrite our equation as 7110 9 02Mx2x 2.
Before implementing land disposal of silver nitrate waste consult with environmental regulatory agencies for guidance. Other chemical compounds have been used as popular fertilizers over the last century. Nitrate de plomb French LeadII nitrate 12 Nitric acid lead2 salt.
A 1-mgL nitrate nitrogen NO3-N solution would be suitable for low-range 0 to 10 mgL tests. For spills of silver nitrate in bodies of water apply sodium sulfide solution then neutralize with agricultural lime crushed limestone or sodium bicarbonate and use mechanical dredges to removed immobilized masses. Lasts up to 90 tests If you walk into your local fish store this will likely be the test kit they keep on the.
Which one of the following salts was dissolved in the original solution. Soil sodicity on the other hand is caused by high sodium levels in soils at concentrations greater than 15 percent of the cation exchange capacity. Well take a solution containing 100 g of potassium nitrate and 100 g of water.
Urea on the other hand has an NPK grade of 46-0-0 making it more economical to transport. Our product line consists of chemical solutions prepared to exact quality standards and certified for use in laboratories and production processes. Potassium nitrate sulfur and carbon Gunpowder Oxygen and water Sea foam Petroleum hydrocarbons and fuel additives Gasoline Minerals organic materials air water and living organisms Soil Chlorine caustic soda and water Bleach Plasma white blood cells red blood cells and platelets Blood The opposite of heterogeneous mixtures is homogeneous mixtures.
Write the balanced equation for the reaction that took place between the salt and silver nitrate. While the potassium nitrate KNO 3 is soluble the lead iodide PbI 2 is nearly insoluble at room temperature and thus precipitates out. 5 982 grams of lead IV nitrate are dissolved to make 465 mL of solution.
All the potassium nitrate will stay in solution. In the following example it is assumed that a set of standards for a 0 to 50 mgL range is being prepared. Its quite an experience hearing the sound of your voice carrying out to a over 100 first year.
Supralumbar Kertaconjunctivitis Off-label Apply 05-1 solution to anesthetized upper tarsus allow it to fall back into place over affected. 4 952 grams of ammonium carbonate are dissolved to make 1750 mL of solution. Chlorine water and carbon tetrachloride were added which resulted in a purple solution.
It is the number of oxygen atoms that bring out the difference in the shape chemical structure and properties of these compounds. Both of these compounds are inorganic compounds composed of nitrogen and oxygen. Lead standard solution for AAS 1 mg-ml Pb in 2-5 HNO3.
13 potassium fluoride is KF 14 ammonium sulfate is NH 42SO 4 15 magnesium iodide is MgI 2 16 copper II sulfite is CuSO 3 17 aluminum phosphate is AlPO 4 18 lead II nitrite is PbNO 22.
Nitrate ion NO 3-5 Nitrogen is of concern to agriculture both as an essential plant nutrient for building proteins and amino acids and as a potential water pollutant. Substance Substance name.

Acros Organics Ac217560010 Magnesium Nitrate Hexahydrate P A 1kg From Cole Parmer United Kingdom
Magnesium Bromide Hexahydrate LR 500GM.

Magnesium nitrate hexahydrate. Magnesium Magnesium Chloride Hexahydrate Contract. Les rapports Global Nitrate de magnésium hexahydrate Market Research fournissent un aperçu détaillé et une analyse approfondie des dernières tendances du marché et de létendue de lexamen la recherche est détaillée. Magnesium nitrate has been reported to undergo spontaneous decomp in dimethylformamide possibly as a result of hydrolysis of the hexahydrate above its mp 90 C to liberate nitric acid.
What is Magnesium nitrate. Magnesium Carbonate Light LR 500GM. The heat storage device has two groups of coils the inner side which carries water and the outer side which is the phase change.
Cd 3 AsO 4 2. Pore size 02 μm pore size 05 μm cartridge nominal length 5 in. About the list.
Active ingredients including adrenaline and noradrenaline changes that will require dual labelling hydration changes and other significant and minor changes. Magnesium oxide All Grades SDS. Magnesium Chloride Hexahydrate SDS.
MgNO 3 2 is an inorganic nitrate salt of magnesium with a chemical name Magnesium nitrate. Magnesium Sulfate 05M Solution SDS. Preparation of the metathesis method potassium nitrate ammonium nitrate and potassium chloride raw materials products for potassium nitrate and ammonium chloride recycled mother liquor wherein the device is provided close to the production of a shunt purify production line production line set up in addition to sodium unit and the unit except magnesium which in addition to the sodium.
The list below shows the medicine ingredient names that are changing. Magnesium Chloride Hexahydrate CAS-No. We regularly produce chemical solutions to specifications designed by government and regulatory bodies commercial and trade associations and the specific needs of individual users and businesses.
It is highly soluble in water. Magnesium dinitrate is a hygroscopic crystalline solid white. Nitrogen as nitrate or ammonium is highly soluble and moves rapidly in runoff and in soil solutions.
Magnesium nitrate refers to inorganic compounds with the formula MgNO 3 2 H 2 O x where x 6 2 and 0. A common derivative is the tetrahydrate MnNO 3 2 4H 2 O but mono- and hexahydrates are also known as well as the anhydrous compound. The loss of the individual waters of hydration upon heating the hexahydrate can be studied and the existence of the anhydrous covalent compound NiNO32.
The anhydrous material is hygroscopic quickly forming the hexahydrate upon standing in air. Handbook of Reactive Chemical Hazards. Lead Nitrate Crystal ACS Grade.
Magnesium nitrate is also called Magnesium dinitrate or Magniosan or Nitromagnesite hexahydrate. All are white solids. Some of these compounds are useful precursors to the oxides of manganese.
Our product line consists of chemical solutions prepared to exact quality standards and certified for use in laboratories and production processes. A new type magnesium nitrate hexahydrate-based phase change material has been studied to improve the cooling degree and crystallization difficulty. Of the Federal Food Drug and Co smetic Act.
Can be observed before it decomposes using differential thermal analysis and thermogravimetric analysis techniques. All of the salts are very soluble in both water and ethanol. Barium nitrate BaNO 3 2 464 103 Barium selenate BaSeO 4 340 108 Barium sulfate BaSO 4 108 1010 Barium sulfite BaSO 3 50 1010 Beryllium hydroxide BeOH 2 692 1022 Bismuth arsenate BiAsO 4 443 1010 Bismuth iodide BiI 3 771 1019 Cadmium arsenate Cd 3 AsO 4 2 22 1033 Cadmium carbonate CdCO 3 10 1012 Cadmium fluoride CdF 2.
ManganeseII nitrate refers to the inorganic compounds with formula MnNO 3 2 H 2 O nThese compounds are nitrate salts containing varying amounts of water. The heat storage device has two groups of coils the inner side which carries water and the outer side which is the phase change. Lithium Chloride Anhydrous SDS.
125 cm Code 7 EPDM rubber seal. 11 11082017 EN English US Page 1 SECTION 1. Bulk Drug Substances Nom inated for U se in Com pounding Under Section 503B.
The focus of this study is on the heat charging and discharging characteristics of this new phase change material. The concentration limit in drinking water is 10 mg per liter. Calcium nitrate icsc calcium polysulfide icsc chlorosulfonic acid icsc fluoroboric acid aqueous solution 25 icsc magnesium nitrate icsc paraldehyde icsc polystyrene icsc sodium bicarbonate icsc sodium chlorite icsc sodium perborate tetrahydrate icsc sodium sulfide anhydrous icsc tributylamine icsc 124.
Magnesium Hydroxide Powder SDS. A progressive and dynamic company Buradon is one of India s leading manufacture and exporter of fine chemicals to the rigid standard of GR AR EL ACS and LR grades due to its industrial exposure commitment and market fixing capabilities from India. Nickel nitrate hexahydrate loses water on heating and eventually decomposes forming nickel oxide.
Updated June 07 2021. Magnesium Carbonate Light AR 500GM. Being highly water soluble magnesium nitrate occurs naturally only in.
Magnesium Carbonate Light LR 250GM. IP BP EP Ph Eur USP NF JP Analytical Reagent FCC Food Grade Chemicals Excipients Shale Gas Fracturing Chemicals Manufacturers. It is widely used in pyrotechnics.
Magnesium Carbonate Lab Grade. Ce rapport de recherche. This list is split into.
Magnesium Chloride Hexahydrate Safety Data Sheet according to Federal Register Vol. Buildup of nitrate in groundwater is a health concern. 58 Monday March 26 2012 Rules and Regulations Date of issue.
AMERICAN INTERNATIONAL CHEMICALS 395 orders FRESENIUS MEDICAL CARE NA 260 orders FRESENIUS MEDICAL CARE 62 orders THE COCA COLA EXPORT CORP 37 orders MEDISYSTEMS CORP 37 orders CARIBBEAN REFRESCOS 36 orders ATLANTA CONSOLIDATE WAREHOUSE 36 orders AMPOINT DISTRIBUTION. 27 -21 931 0823 68 Industrial Ring Rd Parow Industrial Cape Town scienceworldscienceworldcoza. Magnesium Bromide Hexahydrate LR 25KG.
The focus of this study is on the heat charging and discharging characteristics of this new phase change material. Muby Chemicals of Mubychem Group established in 1976 is the original manufacturers of Specialty Chemicals Pharmaceutical Excipient Fragrance Food Flavor chemicals Reagent Grade Chemicals Shale Gas Fracturing Chemicals in India. Cobalt Ii Nitrate Hexahydrate AR 100GM Cobaltous Nitrate Cobalt Ii Nitrate Hexahydrate AR 250GM Cobaltous Nitrate.
Identification Product form. A new type magnesium nitrate hexahydrate-based phase change material has been studied to improve the cooling degree and crystallization difficulty. Il protège les avancées technologiques mondiales de manière authentique tout en étant soumis à une zone industrielle globale critique.
2 kg calcium hydrogen phosphate to moles. How many moles of silver are in the ring.
Notified of all changes on this drug.
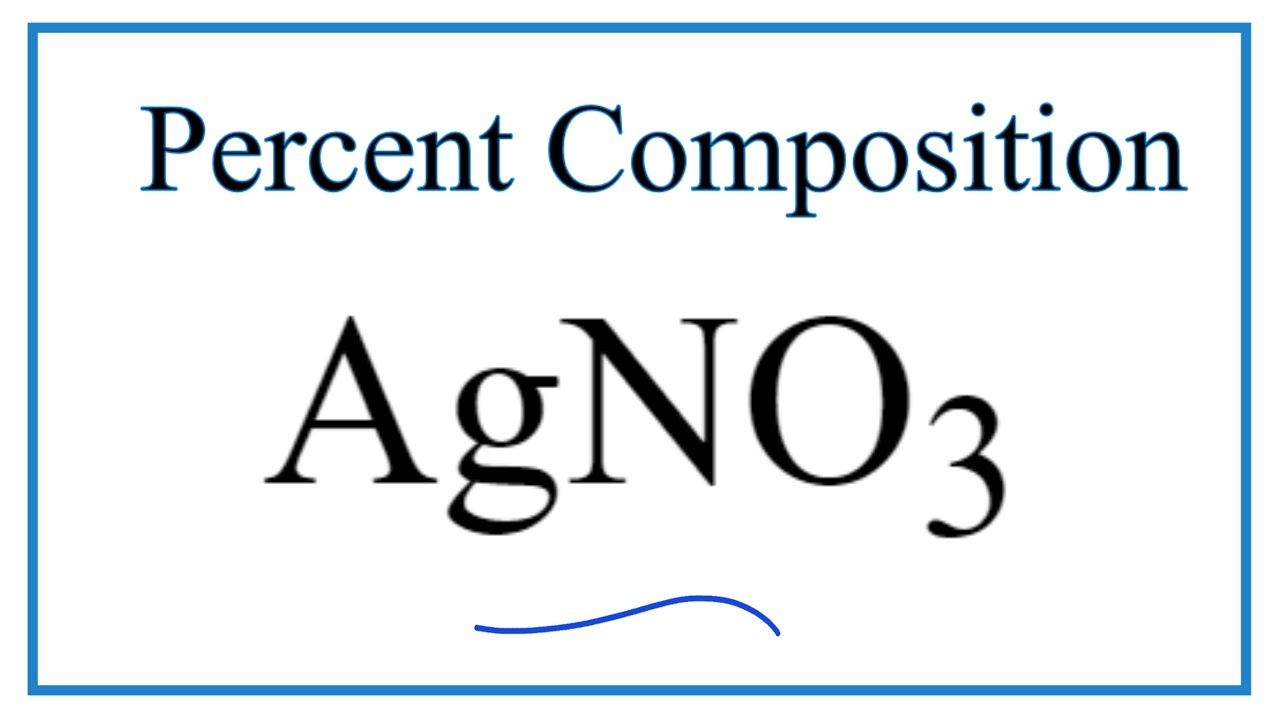
Silver nitrate mass. They are each made of a ca. The actual yield was 220g how do you calculate percent yield. A photon of light hitting the negative nitrate anion frees an electron which ultimately combines with the positive silver ions to make neutral silver metal darkening the surface of the material.
Calculate the molar mass for each of the following substances. 3 c magnesium nitrate MgNO. Targets 1 Enzymes 1 Transporters 1 Potassium nitrate.
Convert quantities to volumes. Silver nitrate is an inorganic compound with chemical formula AgNO 3This salt is a versatile precursor to many other silver compounds such as those used in photographyIt is far less sensitive to light than the halidesIt was once called lunar caustic because silver was called luna by the ancient alchemists who associated silver with the moon. Find the number of moles from a given mass.
In this case both of the reactants are salts. A 50 reduction in the radioactivity content of spleen occurred after about 70 hr while that of brain took about 84 hr. Silver injected into rats as radioactive silver nitrate had a half-time of 22 days.
Find the equivalent mass of silver. 1030 atoms of helium to liters. Therefore silver sulfadiazines effectiveness as an antimicrobial agent for preventing burn wound infections is due to its tendency.
1 mol C 1201 g C or 12. To calculate chlorides solution concentration use. The mass of A g C l ceAgCl A g C l formed is 7175 g 7175text.
The molar mass of magnesium nitrate equals 14833 gmol. Silver nitrate is used in many ways in organic synthesis eg. Titrate with silver nitrate solution till the first color change.
It can be calculated by adding the invididual molar mass of every atom that are composing the molecule CH4. Calculate the mass of silver bromide which should be produced from 225g of silver nitrate. I will give the lowest level of explanation and then follow up with a high level explanation.
Silver occurs natively and. For instance if you had a 800 g sample of a compound that was 200 g element X and 600 g element y then the percent composition of each element would be. In solid silver nitrate the silver ions are.
Percent composition in chemistry typically refers to the percent each element is of the compounds total mass. For rats given a single ip injection of radioactive silver nitrate a 50 reduction in radioactivity of whole blood plasma kidney liver after approx 40 hr. A silver ring contains 11 x 1022 silver atoms.
The molar mass of silver nitrate is 169872 grams per mole. In this experiment the amount of chloride in an unknown sample was determined by. 245 mol Cu 6022 1023 atoms 1 mol 18 10 148 1024 atoms Cu 11 1022 atoms Ag 1 mol 6022 1023 atoms -2 mol Ag Molar Mass.
7 5 g of A g C l ceAgCl A g C l. Its density in the liquid state at a temperature of 210 o C corresponds to 397 gcm 3. Due to these toxic effects many countries have placed severe restrictions on the amount of their utilisation in processed food products.
The mass of 1 mol of atoms of an element An elements molar mass in gmol is numerically equal to the elements atomic mass in amu. Silver nitrate or lunar caustic the most important silver compound is used extensively in photography. What occurs to the theoretical yield if a sample is incompletely dried.
Los Alamos National Laboratory US. Silver NH 4 ammonium Ca2 calcium Fe2 ironII Cu2 copperII Mg2 magnesium Ni2 nickelII Sr2 strontium Zn2 zinc Sn2 tinII Pb2 leadII Ba2 barium Fe3 ironIII Al3 aluminium Cr3 ChromiumIII F-fluoride Cl-chloride Br-bromide I-iodide HO-hydroxide NO 2-nitrite NO 3-nitrate HCO 3-hydrogencarbonate O2-oxide CO 3 2-carbonate SO 4 2-sulfate HSO 4-hydrogensulfate S2-sulphide N3. The biological half-time of.
Answer 1 of 5. When in 1840 Henry Talbot discovered an additional chemical twist that is so called latent silver image that had been briefly exposed onto a layer of silver iodide could be revealed using gallic. When you get questions like this it helps a lot if you can see the general sorts of patterns.
Discussion The well known Mohrs method in which alkaline or alkaline earth chlorides react with silver nitrate in the presence of a few drops of potassium chromate solution as indicator is a simple direct and accurate method for chloride determination. It is solid white or sometimes white to dirty grey in colour. Calculate the molar mass for each of the following substances.
6 c strontium acetate SrC. Formation of nitrite and propagation of its downstream NO-signaling effects depends on the oral bacterial reduction of inorganic nitrate by a set of bacterial nitrate reductase enzymes NaRs that are largely absent from the human genome In a process referred to as the enterosalivary nitrate circulation or the nitrate-nitrite-NO pathway dietary nitrate is swallowed and rapidly. The melting and boiling points of silver nitrate are 4828 K and 713 K respectively.
Nitrate is also recognized as hazardous on the basis that it can be easily reduced to nitrite in-vivo. Isotopes of silver range in relative atomic mass from 92950 u 93 Ag to 129950 u. A silver metal Ag b ammonia gas NH.
Excessive amount of nitrite and nitrate in food has been a major concern of food safety thus a highly sensitive selective speedy. Notice in Figure 6 how silver sulfadiazine Ag sulfadiazine gradually releases the majority of its silver ions into solution over an extended period of time whereas silver nitrate immediately released all of its silver ions into solution Fox and Modak 1974. 12 lbs of 4-cyanoindole.
It is an alkali metal nitrate because it is an ionic salt of potassium ions K ions and nitrate ions NO 3. Later a displacement reaction takes place when N a C l ceNaCl N a C l is added to A g N O X 3 ceAgNO3 A g N O X 3 to form 7175 g 7175text g 7 1. For deprotection and oxidations.
Atomic mass of Cl- 3545 gmole mmoles of Cl- A M. A 5494 g. Potassium nitrate contains potassium a soft light and silver metal oxygen and nitrogen a colourless and odourless gas.
AgNO 3 as a colourless appearance in its solid state and is odourless. Potassium nitrate is a medication used to cauterize small wounds remove granulation tissue warts and verrucae and treat tooth sensitivity. According to the reaction equation.
What occurs to the reported yield. Compute properties of a chemical solution. Notified of marketing changes.
Silver for centuries has been used traditionally for coinage by many countries of the world. The resulting adduct can be decomposed with ammonia to release the free alkene. Chemical Solutions Explore properties of different solutions which depend on the solvent solute and concentration.
In recent times however consumption of silver has greatly exceeded the output. Ag binds alkenes reversibly and silver nitrate has been used to separate mixtures of alkenes by selective absorption. Ag Cl- AgCl.
500mg of silver nitrate. In a double replacement reaction between silver nitrate and magnesium bromide. In its solid state it has a density of 435 grams per cubic centimetre.
A manganese metal Mn b sulfur hexafluoride SF. That makes calculation especially easy - when we calculate number of moles of AgNO 3 used it will be already number of moles of Cl-titrated. Silver nitrate reacts with chloride anion on the 11 basis.
The molar mass of a substance also often called molecular mass or molecular weight although the definitions are not strictly identical but it is only sensitive in very defined areas is the weight of a defined amount of molecules of the substance a mole and is expressed in gmol. Potassium nitrate is soluble in hot water. 54 g 54text g 5 4 g of silver reacts with H N O X 3 ceHNO3 H N O X 3 to form silver nitrate.
The basic equation mass of element mass of compound X 100.
The ammonia formed may be detected by its characteristic odor and by damp red litmus papers turning blue signalling that it is an alkali very few gases other than ammonia evolved from wet chemistry are alkaline. Other alloys like brass copper and zinc and bronze copper and tin are easy to shape and beautiful to look at.

Zinc Strip In Copper Nitrate Solution Youtube
To learn more about the Structure Physical Properties Chemical Properties Uses and FAQs of Copper hydroxide Visit BYJUS for more content.
Copper reacts with zinc nitrate. 1 mark Question 2 continues on the next page. Titanium is much lighter and less dense than steel but as strong. Copper is a chemical element with the symbol Cu from Latin.
This product rapidly reacts with oxygen in the air to. It is a soft malleable and ductile metal with very high thermal and electrical conductivityA freshly exposed surface of pure copper has a pinkish-orange colorCopper is used as a conductor of heat and electricity as a building material and as a constituent of various metal alloys such as sterling. Sodium chloride lead 11 nitrate lead 11 chloride sodium nitrate 2NaC1 PbC12 2NaN03 iron chlorine iron 111 chloride 2 Fe 2FeC13 barium water barium hydroxide hydrogen Ba 21-120 12 When chlorine gas reacts with methane carbon tetrachloride and hydrogen chloride are produced.
Its also very resistant to corrosion. Zinc oxideeugenol cement Fig. Hazardous Substances Data Bank HSDB ZINC NITRATE EXPLODES WHEN.
Percent yield represents the ratio between what is experimentally obtained and what is theoretically calculated multiplied by 100. Devardas alloy CopperAluminiumZinc is a reducing agentWhen reacted with nitrate in sodium hydroxide solution ammonia is liberated. Van Nostrand Reinhold 1996 p.
First of all zinc helps the plants reach optimal size and maturity as well as the production of leaves stalks stems and branches. When iron reacts with copper sulphate blue a green colour is obtained due to the formation of iron oxide. Zinc silver nitrate --- zinc nitrate and silver metal.
We will be publishing Issue 1 of The School STEM Technician at noon on the 1st December 2021 via the SSERC website. Negative ions that consist of a single atom are named by adding the suffix -ide to the stem of the name of the element. In 12 sentences explain the.
The second part. CC14 4 HCI When sodium oxide is added to water sodium hydroxide is produced. A student wants to make copper chloride crystals.
The zinc oxideeugenol cements are used to provide a sedative. Copper hydroxide reacts with sulfuric acid to form copper sulfate and water. Deficiencies in auxin levels can cause reduced development speeds.
Iron copper nitrate --- iron nitrate and copper metal. Can react violently with carbon copper metal sulfides organic matter phosphorus sulfur. Red-brown copper will be left.
2AlBr 3 3 Cl 2 2AlCl 3 3Br 2 Single Replacement 3. The chemical reaction between lead nitrate and potassium iodide is accompanied by the formation of a yellow precipitate of lead iodide. A salt is any compound formed by the neutralisation of an acid by a base.
Bromine potassium iodide --- potassium bromine and iodine. The student adds excess copper oxide to some hot acid. Zinc and Lead II nitrate react to form Zinc Nitrate and Lead.
ZINC NITRATE is an oxidizing agent. NH 4 ammonium. Zn PbNO 3 2 ZnNO 3 2 Pb Single Replacement 2.
This is our newest publication and has been created to support the school technician profession in Scotland. Before running the experiment the scientist expected the products of the reaction will be a total of 159 grams of copper sulfate Cu2S. The rosin increases fracture resistance and the zinc acetate is effective in accelerating the reaction rate.
1 mark Tick one box. Here colour change is seen. Pour the cool residue into a 100 cm 3 beaker and add a little dilute hydrochloric acid to dissolve the zinc oxide and also any unreacted zinc and copper oxide warming if necessary.
You want to measure how much water is produced when 120 g of glucose C_6H_12O_6 is burned with enough oxygen. Thermal decomposition is used to obtain zinc oxide from its carbonate ore. Metals combined with acids are almost always single replacement chemical.
Instantly reacts with atmospheric oxygen to produce nitrogen dioxide. 0 Suggest how the student would know that excess copper oxide has been 2. Therefore none of zinc copper or lead will displace magnesium ions from solution so that there will be no reaction.
When one chemical reacts to a stimulus by breaking into two simpler elements. The liquid is a preparation of eugenol which reacts with the powder to form an amorphous chelate of zinc eugenolate. In this reaction all of the copper and all of the sulfur react.
Is copperII nitrate soluble or insoluble. This can be rinsed with water and. So here on adding zinc to CuSO4 solution zinc displaces copper from copper sulphate forms zinc sulphate solution.
Bronze is also used frequently in ship-building because it is resistant to corrosion from sea water. The first part comes from the metal metal oxide or metal carbonate. Yield actual yieldtheoretical yield 100 So lets say you want to do an experiment in the lab.
0 2 1 Which acid should the student use. Limiting reagent is the reactant which limits the progress of a chemical reaction. Copper hydroxide is also called cupric hydroxide.
H 3 O hydronium. Copper hydroxide -CuOH2 - Copper hydroxide is a crystalline inert compound used in the preparation of a wide variety of salts. Calcium water --- calcium hydroxide and dihydrogen.
CopperI Cu 2 copperII Polyatomic positive ions often have common names ending with the suffix -onium. Saxs Dangerous Properties of Industrial Materials. At the same time the nitrate ions NO 3 undergo a series of reactions to form nitrogen monoxide NO.
Aluminum Bromide and Chlorine gas react to form Aluminum Chloride and Bromine gas. 2Na 3 PO 4 3 CaCl 2 6NaCl Ca 3 PO 4 2. EPA Pesticide Chemical Code 076102.
Zinc and lead two nitrate make zinc nitrate and lead metal. Iodine crystals and chlorine gas react to make solid iron and carbon dioxide gas. Zn Pb NO 3 2 ZnNO 3 2 Pb.
Zinc Deficiency Zinc has a lot of functions in a plants health. The above reactions should be written in your notebook and all observations should be related to the compounds which appear in those reactions. 2NOg O 2 g.
If two atoms must combine at a 1 to 1 ratio but there is an unequal amount of 1 atom then the reaction will stop when the atom with less quantity runs out. The name of a salt has two parts. Oxidizing Copper Metal with Concentrated Nitric Acid HNO 3 aq The first step involves transforming Cu metal to copperII ions Cu2 using concentrated nitric acid HNO 3 aq.
Sodium Phosphate and Calcium Chloride react to form Calcium Phosphate and Sodium Chloride. Nitric acid copper2 salt. Explanation - Zinc is more reactive than copper.
Cuprum and atomic number 29. And although heavier than aluminum its also twice as strong. Reacts violently with combustible and reducing materials.
Adding sodium oxide to water produces sodium hydroxide. Heat this to show that the white powder zinc oxide is yellow when hot and white when cool. Answer 1 of 17.
Zinc copper and lead do not react with magnesium nitrate because in the activity sequence magnesium is higher because it is more reactive than other metals. CuSO4 solution has a blue colour while ZnSO4 solu. Zinc is a vital nutrient in many enzymes and the growth hormone auxin.
Chlorine gas reacts with methane and carbon tetrachloride to produce hydrogen chloride. Copper Cu reacts with sulfur S to form copper sulfide as shown in the equation. Nitric acid copper2 salt 21 9TC879S2ZV.
This is indicated by colour change from blue to colourless. A scientist adds 127 grams of Cu to 32 grams of S to start the reaction. 212 contains zinc oxide rosin and zinc acetate in the powder.
Formation of copperII hydroxide While stirring the solution from the last step with a glass rod add 30 mL. The student stirs the mixture. Nitric acid copper salt.