Mg Immune Support Blend from ferment media European Elder. In aqueous solution an octahedral complex ZnH 2 O 6 2 is the predominant species.

Zinc Sulfate Chemistry Project Home
Contains sodium lauryl sulfate which may cause skin reactions particularly in children with eczema.

Is zinc sulfate aqueous. This is a single displacement reaction of a non-metal by a metalThe zinc will react with sulphuric acid forming zinc sulphate dissolved in solution and releasing hydrogen gas. However the conventional aqueous electrolytes are not capable of working at low temperature. NH 4 OH was added to the emulsion in order to obtain the zinc cation.
With a mild-acidic triflate electrolyte here the authors show a high. Ionic bonds also melt at high temperatures. Lactic acid Potassium sulfate Urea Lactose 1-Propanol Zinc sulfate REFERENCES 1.
Benefiting from high safety low cost and competitive energy density aqueous zinc-ion batteries AZIBs have become a very promising technique for grid-scale energy storage. However they often require the use of a surfactant to ensure ease of handling and suspension in aqueous solutions. The Journal of Endodontics the official journal of the American Association of Endodontists publishes scientific articles case reports and comparison studies evaluating materials and methods of pulp conservation and endodontic treatmentEndodontists and general dentists can learn about new concepts in root canal treatment and the latest advances in techniques and instrumentation in the one.
Here we report a frigostable cost-effective safe and eco-friendly hybrid electrolyte with high. The resulting ZnO calcinates were analyzed by XRD IR and. Zinc as zinc oxide from ferment media 15.
Villalpando S Rivera JA King JC. Zinc administered in aqueous solutions to fasting subjects is absorbed quite efficiently at a rate of 60-70. The component that does the dissolving Generally the component present in the greatest quantity is considered to be the solvent.
Ionic bonds are atomic bonds created by the attraction of two differently charged ionsThe bond is typically between a metal and a non-metal. Genetically modified plants for improved trace element nutrition. Skin barrier disruption by sodium lauryl sulfate-exposure alters the expressions of involucrin transglutaminase 1 profilaggrin and kallikreins during the repair phase in human skin in vivo.
Aqueous solution of ammonium hydroxide. 3 When dissolved sodium hydroxide reacts with sulfuric acid aqueous sodium sulfate water and heat are formed. V Aqueous Solutions and Body Fluids Hoeber 1966.
The structure of the bond is rigid strong and often crystalline and solid. Sıhnel O and Novotny P Densities of Aqueous Solutions of Inorganic Substances Elsevier Amsterdam 1985. In aqueous solution all forms of zinc sulfate behave identically.
Aqueous Solutions Solution - a homogeneous mixture Solute. A white coating appears on the copper electrode almost immediately and after a. Refer to sizes for USA standard sieves ASTM E11 for ranges of particle sizes.
Mineral form of basic copper carbonate. The absorption of zinc from soluble zinc acetate zinc sulfate. The development of rechargeable aqueous zinc batteries are challenging but promising for energy storage applications.
The component that is dissolved Solvent. Bioavailability of zinc from the various forms was compared on. The volatilization of zinc in combination with zinc chloride at temperatures above 285 C indicates the formation of Zn 2 Cl 2 a zinc compound with a 1 oxidation state.
Aromatic spirit of ammonia. Cardio Support Blend Organic Fenugreek seed from ferment media Organic Oregano leaf from ferment media Grape Seed extract Organic Hawthorn berry leaf and flower from ferment media. With a reduction potential of 076 V zincII reduces only with difficulty.
2-88 2-25 Partial Pressures of H 2 O over Aqueous Solutions of Sodium Carbonate. When heated above 680 C zinc. And insoluble zinc oxide was compared in ten human volunteers who were dosed orally with 50 mg Zn in various forms separated by two weeks intervals.
Similar to other Zn 2 H synergistic intercalative batteries zinc hydroxide sulfate ZHS was also found to be formed in the Znpoly44-TDP system caused by the precipitation of residual OH with Zn 2. Copper as copper sulfate anhydrous from ferment media 15. Solid polymer spheres in dry powder form.
Aqueous solutions are those in which water is the solvent. Reported a novel aqueous Zndiquinoxalino 23-a23-c phenazine ZnHATN battery which was enabled by the reversible H uptakeremoval behavior via. Solute Mass mmol kg-1 cmol L-1 ρg cm-3 n C ηmPa s Acetic acid 05 0084 0083 09989 13334 016 1012 CH3COOH 10 0168 0.
ZnSO 4 BaCl 2 BaSO 4 ZnCl 2. Zinc sulfate is also obtained as a hexahydrate ZnSO46H2O and as a heptahydrate ZnSO47H2O. Barium sulfate forms when these solutions are treated with solutions of barium ions.
To address this issue this work employs cotton-derived cellulose film prepared by a facile. 2 NaOH aq H 2 SO 4l Na 2 SO 4 2 H 2 O l Double Replacement 4 When fluorine gas is put into contact with calcium metal at high temperatures calcium fluoride powder is created in an exothermic reaction. A dark coating of copper metal appears on the zinc within two minutes and when 45 minutes have elapsed there is a thick coat of copper metal powder on the zinc strip and the blue color of the solution has lightened considerably.
Aqueous zinc-based energy storage ZES devices are promising candidates for portable and grid-scale applications owing to their intrinsically high safety low cost and high theoretical energy density. These aqueous solutions consist of the metal aquo complex ZnH 2 O 6 2 and SO 2 4 ions. 2-24 Total Vapor Pressures of Aqueous Solutions of NH 3.
No compounds of zinc in oxidation states other than 1 or 2 are known. Banana oil artificial isoamyl acetate. Custom color and density polyethylene microspheres can be manufactured by request.
To stabilize the water-in-oil emulsion the surfactant Span 80 was added to the heptane. When zinc metal is immersed in a solution of 01 M aqueous copperII sulfate solution c opper metal plates out on the zincThe solution is initially blue in color. Free radicals are highly reactive and unstable chemicals generated during normal body activities that require oxygen eg respiration digestion blood circulation immune system response increased physical activity etc and after exposure to UV.
Zinc administered in aqueous solutions to fasting subjects is absorbed efficiently. However the life span of AZIBs is severely influenced by the uncontrolled zinc dendritic growth and undesirable side reactions. All forms are soluble in water.
Recently Niu et al. Immediate steps should be taken to limit its spread to the environment. The precipitate was dried and then calcined at 7001000 C.
Vitamin C and zinc together help the bodys natural defense against damaging free radicals antioxidant effect and help boost immune function. A zinc electrode and a copper electrode are placed in a beaker containing an aqueous solution of zinc sulfate ammonium citrate and ammonium chloride. The aqueous phase of the system was zinc acetate dissolved in de-ionized water and the organic phase was heptane.
The primary hazard is the threat posed to the environment. Calculations indicate that a zinc compound with the oxidation. Zinc reacts with dilute sulphuric acid to form zinc sulphate and hydrogen gas is evolved.
Zinc absorption from zinc oxide zinc sulfate zinc oxideEDTA or sodium-zinc EDTA does not differ when added as fortificants to maize tortillas. Anhydrous zinc sulfate is a colorless crystalline solid. The electrodes are connected to a DC power supply and a voltage is applied - the negative lead on the copper electrode and the positive lead on the zinc electrode.
Rechargeable zincmanganese dioxide batteries that use mild aqueous electrolytes are attracting extensive attention due to high energy density and environmental friendliness.
Sodium hydroxide Na OH also known as lye or caustic soda is a caustic metallic base. The G-value was automatically calculated by the program-controlled jar test apparatus ZR4-6 Zhongrun Water Industry Technology Development Co.
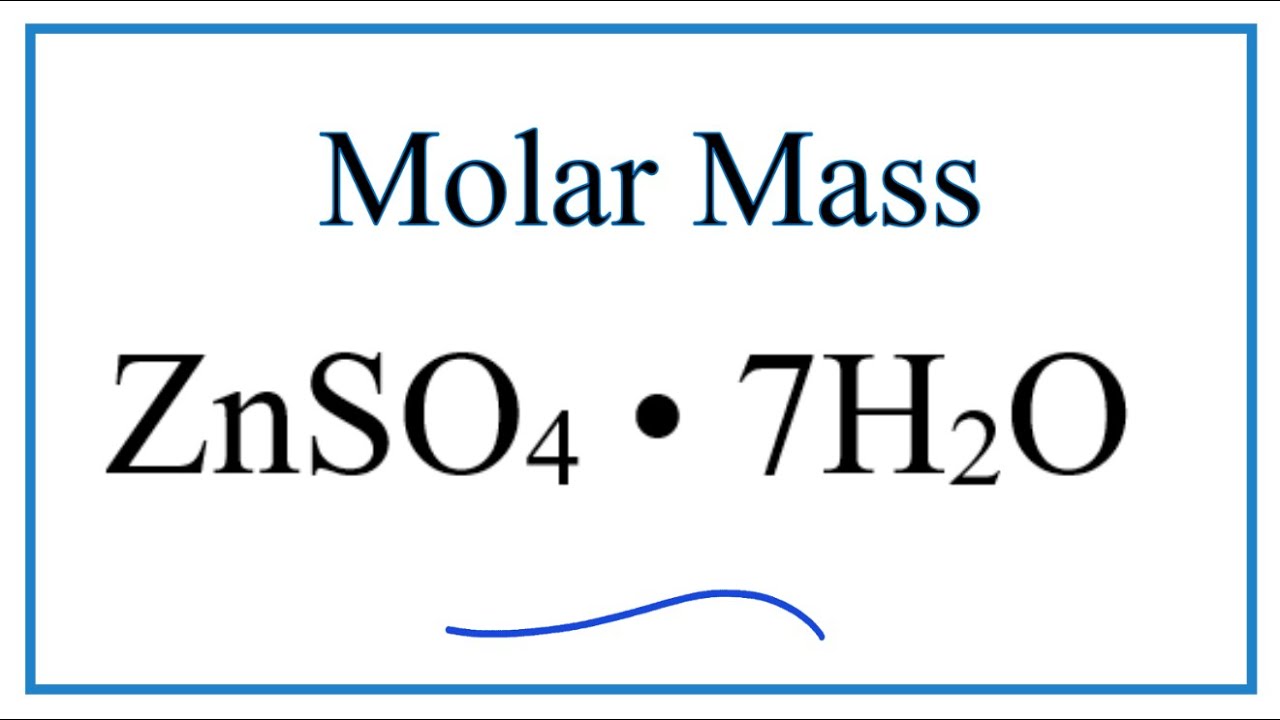
Molar Mass Molecular Weight Of Znso4 7h2o Zinc Sulfate Heptahydrate Youtube
104 gmol 1302 gmol 799 or 8.

Zinc silicate molar mass. Mole concept molar mass and percentage composition. 8 will serve as a multiplier MOLECULAR FORMULA IS C 8H 8 357 Hydrofluoric acid HFaq cannot be stored in glass bottles because compounds called silicates in the glass are attacked by the HFaq. Heavy metals as Pb 00005.
Cu Copper 00002. Al Aluminium 0001. Na Sodium 05.
An introduction to atomic number isotopes and isobars. Molar mass M is the mass in grams of one mole of a chemical substance. Variations of molar conductivity.
Sulfate SO₄ 00005. Difference between atom and ion. Atomic mass molecular mass.
SiFe molar ratio of 14. Determine the mass of gold that can be extracted if 250 g of sodium cyanide is used. The vapor pressure of pure water at this temperature is 238.
Hafnium dioxide is an intermediate in some processes that give hafnium metal. Molar heat capacity. Atomic mass unit amu a unit of mass equal to 112 the mass of the carbon isotope with mass number 12 approximately 16604 x 10E-24 gram.
The present study measured strontium Sr isotopes with hydro-geochemistry data of 56 water samples in detail in the MRB in northeast Thailand. Molar Mass of Gas. View Answer When does the.
Stoichiometry is the chemistry that mathematically relates all substances in a reaction quantitatively relating the amount of reactants and products in a chemical reaction. Calcium sulfate or calcium sulphate is the inorganic compound with the formula CaSO 4 and related hydratesIn the form of γ-anhydrite the anhydrous form it is used as a desiccantOne particular hydrate is better known as plaster of Paris and another occurs naturally as the mineral gypsumIt has many uses in industry. Zinc silicate powder ZS was mixed with multi-walled carbon nanotubes MWCNTs at various mass fractions 0 1 2 and 3 wt followed by argon sintering.
X-ray diffraction XRD analysis. Thereafter the mass ratios of the four. Atomic mass the mass of an atom expressed in atomic mass units amu.
For example the molar mass of carbon is 12 g mol1. As the largest and most representative tributary of the Mekong River the Mun River Basin MRB provides critical understanding of regional hydro-geochemical features and rock weathering processes on a basin scale. Atomic mass and molecular mass.
269815385 7 Standard state. That is while the fractional mass ratio of the asphaltenes remained the same at 10 the rest of the components decreased with aging ie from 27 to 10 for saturates 31 to 10 for aromatics and 39 to 11 resins. Worldwide production in 1998 was around 45 million tonnes.
Reaction mass of 2-chloroethyl chloropropyl 2-chloroethylphosphonate mixture reaction mass of isomers and 2-chloroethyl chloropropyl 2-chloropropylphosphonate reaction mass of isomers 401-740-0 015-144-00-X reaction mass of pentyl methylphosphinate and 2-methylbutyl methylphosphinate 402-090-0 87025-52-3 015-145-00-5. All forms are white solids that are poorly soluble in water. For monatomic ideal gases the ratio of those at constant pressure and volume is 53.
Collecting Gases Over Water. Daltons Law of Partial Pressure. Cyclohexanone-formaldehyde resin produced when 1 mole of cyclohexanone is made to react with 165 moles of formaldehyde such that the finished resin has an average molecular weight of 600-610 as determined by ASTM method D2503-82 Standard Test Method for Molecular Weight Relative Molecular Mass of Hydrocarbons by Thermoelectric Measurement of Vapor Pressure which is incorporated by.
Pb Lead 00005. Relative atomic mass A r. Total nitrogen N 00005.
93347 66032 C 122058 F K. Gas Diffusion and Effusion. Therefore the molar mass of an element or compound is the relative atomic molecular or formula mass amount expressed in grams.
Sodium hydroxide is also the most common base used in chemical laboratories. An alkali caustic soda is widely used in many industries mostly as a strong chemical base in the manufacture of pulp and paper textiles drinking water and detergents. Van der Waals Equation.
Zinc-free floor finish water-based styrene acrylic polymer emulsion for high traffic applications such as schools hospitals and retailMor-Glo G128 is recommended for low maintenance floor finishes. 5611 gmol Pricing Availability. Van der waals forces.
Ni Nickel 00005. What quantity for ten points is defined as the heat capacity per mass. Determine the mass of glucose molar mass 180 gmol needed to add to 5000 g of water to change the vapor pressure to 231 torr.
The oxygen in these oxides and in other compounds mostly silicate minerals and calcium carbonate in limestone makes up nearly half of the Earths crust by mass. The Effect of Intermolecular Forces. Besides the hexagonal wurtzite structure of ZnO expands its volume around 16 times higher than the metallic zinc and hence its deposition on the anode surfaces may cause.
Sodium silicate Na 2SiO 3 for. Many molecules in living things have oxygen in them such as proteins nucleic acids carbohydrates and fats. The molar mass of a substance also often called molecular mass or molecular weight although the definitions are not strictly identical but it is only sensitive in very defined areas is the weight of a defined amount of molecules of the substance a mole and is expressed in gmol.
Solid at 298 K. The Effect of the Finite Volume. Since CH has a formula weight of 1302 divide molar mass by that.
Most living things use oxygen in respiration. In the coagulation tests we just needed to set the G value and the program. P is the rate of energy dissipation per unit mass of water W m 3 and μ is the dynamic viscosity of water Pa s.
The molar mass of an element or compound is given the unit grams per mole or g mol1. Silicate SiO₂ 0005. For water at 15 degrees Celsius it is defined as 1 calorie per gram per degree Celsius.
4Au 8NaCN O_2 to 4NaAuCN_2 4NaOH. Ca Calcium 0001. Atomic molar mass gmol Symbol Electronegativity Atomic number Key iron 26 5585 Fe 18 3 2 europium 63 15196 Eu 3 2 americium 95 243 Am 3 4 gadolinium 64 15725 Gd 12 3 curium 96 247 Cm 3 terbium 65 15893 Tb 3 berkelium 97.
235 W m 1 K 1. It can be calculated by adding the invididual molar mass of every atom that are composing the molecule CH4. A higher molar volume in comparison with zinc metal due to its hexagonal wurtzite structure is the main reason for drastic effects of the deposited layers of ZnO on the anode surface from the structural point of view.
It allows the chemist to determine the amount of product that will form from a given amount of reactants or the amount of one reactant that is needed to react completely with some specific amount of the other reactant. Difference between elements and atoms. It can be easily formulated into EPA Safer Choice Green Seal and Ecologo certifiable floor.
Distribution of Molecular Speeds and Collision Frequency. The total number of protons and neutrons in the nucleus. 2792 2519 C 4566 F K.
150 g of NaCl completely dissolves producing Na and Cl ions in 100 kg of water at 250 C. HafniumIV oxide is the inorganic compound with the formula HfO 2Also known as hafnium dioxide or hafnia this colourless solid is one of the most common and stable compounds of hafniumIt is an electrical insulator with a band gap of 5357 eV. Kinetic Molecular Theory and Gas Laws.
The vapor pressure of water at 250 C is 238 torr. Oxygen is a part of water which all known life. One form is the partial derivative of the enthalpy per unit mass with respect to temperature taken at constant pressure.
Atomic number the number of protons in the nucleus of an atom. Fe Iron 00005.
These electrons are transferred through a series of molecules and the energy released during these transfers is used to generate a gradient of _____ or _____. Health-care leaders in the United States warn about the consequences of Thanksgiving-holiday travel and social mixing.

Figure 1 From Mammalian Zinc Transporters Nutritional And Physiologic Regulation Semantic Scholar
Biotin is widely found in food and good dietary sources include egg yolk liver whole-grain cereal and some vegetables.

Zinc transport in the mammalian intestine. Stearman et al 1996. Learn more about the composition form and physical adaptations of the human body. Absorption is efficient and occurs by an active transport mechanism.
More information Long-term anticonvulsant anti-seizure therapy may increase the dietary requirement for biotin because anticonvulsants can interfere with the intestinal. Glycogen stores are used to fuel prolonged exertions such as long. Zinc is a chemical element with the symbol Zn and atomic number 30.
This website uses cookies to help provide you with the best possible online experience. Biotin cannot be synthesized by mammalian cells and must be obtained from exogenous sources. Glycogen stores are used to fuel prolonged exertions such as long.
Characteristic of the vertebrate form the human body has an internal skeleton with a backbone and as with the mammalian form it has hair and mammary glands. A zinc and copper alloy is used to make pennies in the United States. Here we analyze the role of Catsup the Drosophila ortholog of the mammalian zinc transporter SLC39A7 ZIP7 in regulating dopamine levels.
It is the first element in group 12 IIB of the periodic tableIn some respects zinc is chemically similar to magnesium. We found that Catsup is a functional zinc transporter that regulates intracellular zinc distribution between the ERGolgi and the cytosol. The re-oxidation of ferrous to ferric iron is apparently an obligatory step in the transport mechanism although the coupling mechanism of oxidation and membrane transport is unclear.
In humans these molecules provide the fiber required for moving waste through the large intestine and a healthy colon. For example hydrogen magnesium lead and uranium are all chemical elements. Metabolism promotes excellence in research by publishing high-quality original research papers fast-tracking cutting-edge papers research brief reports mini-reviews and other special articles related to all aspects of human metabolism.
Loss-of-function of Catsup leads to increased DA levels and we. The fraction of zinc absorbed is difficult to determine because zinc is also secreted into the gut. De Silva et al 1995.
Yuan et al 1995. Zinc is one of the most common elements in the earths crust. Human body the physical substance of the human organism.
Element one of the 103 chemical substances that cannot be divided into simpler substances by chemical means. FET3 is strikingly homologous to the mammalian copper oxidase ceruloplasmin. The enzymeÕs catalysis of 180 exchange into inorganic phosphate strongly supported the notion that the phosphoserine is a.
14 GLUTS are encoded by human genome. Please read our Terms Conditions and Privacy Policy for information about. Not until 1961 did Engstrom discover that the intestinal enzyme formed a phosphoseryl residue when incubated with phosphate esters at low pH 27 28.
Soybean meal is commonly heat or chemically treated to reduce ruminal protein degradability and increase the flow of dietary amino acids to the small intestine. It requires an electron-transport chain that operates on the high-energy electrons taken from the activated carrier molecules _____ and _____ that are produced by glycolysis and the citric acid cycle. Both elements exhibit only one normal oxidation state 2 and the Zn 2 and Mg 2 ions.
Zinc is a slightly brittle metal at room temperature and has a silvery-greyish appearance when oxidation is removed. Zinc absorption in the small intestine is one of the main mechanisms regulating the systemic homeostasis of this essential trace element. Zinc is absorbed in the small intestine by a carrier-mediated mechanism.
The excess sugars in the body are converted into glycogen and stored in the liver and muscles for later use. Because their concentration is. The excess sugars in the body are converted into glycogen and stored in the liver and muscles for later use.
Some metabolism of L-arginine takes place in the enterocytes. Zinc has many commercial uses as coatings to prevent rust in dry cell batteries and mixed with other metals to make alloys like brass and bronze. Commercial processes used to produce heat-treated SBM include cooker expeller processing of SB extruder-expeller processing of SB nonenzymatically browning of dehulled solvent extracted SBM and cooker processing of dehulled.
L-arginine not metabolized in the enterocytes enters the portal circulation from whence it is transported to the liver where again some. In particular current knowledge on human intestinal zinc absorption and the influence of diet-derived factors on bioaccessibility and bioavailability as well as intrinsic. Under normal physiologic conditions transport processes of uptake are not saturated.
This study has found that supplementation of zinc lysine and vitamin A for 2 months can increase total TCD4 and the levels of IFN- γ in pulmonary tuberculosis patients. In humans these molecules provide the fiber required for moving waste through the large intestine and a healthy colon. The intestinal flora in the human gut are able to extract some nutrition from these plant fibers.
Zinc administered in aqueous solutions to fasting subjects is absorbed efficiently 60-70 whereas absorption from solid diets is less. Absorbed from the lumen of the small intestine into the enterocytes. Browse the archive of articles on Nature.
The intestinal flora in the human gut are able to extract some nutrition from these plant fibers. Particularly calf intestine became the prototype for investigators exploring the properties of the enzyme itself. Electron transport chain a group of electron carriers in mitochondria that transport electrons to and from each other in a sequence in order to generate ATP.
This review summarizes the key aspects of human zinc homeostasis and distribution. It is found in air soil and water and is present in all foodsPure zinc is a bluish-white shiny metal. Glucose transporters are a wide group of membrane proteins that facilitate the transport of glucose across the plasma membrane a process known as facilitated diffusionBecause glucose is a vital source of energy for all life these transporters are present in all phylaThe GLUT or SLC2A family are a protein family that is found in most mammalian cells.
The ammonia formed may be detected by its characteristic odor and by damp red litmus papers turning blue signalling that it is an alkali very few gases other than ammonia evolved from wet chemistry are alkaline. Other alloys like brass copper and zinc and bronze copper and tin are easy to shape and beautiful to look at.

Zinc Strip In Copper Nitrate Solution Youtube
To learn more about the Structure Physical Properties Chemical Properties Uses and FAQs of Copper hydroxide Visit BYJUS for more content.
Copper reacts with zinc nitrate. 1 mark Question 2 continues on the next page. Titanium is much lighter and less dense than steel but as strong. Copper is a chemical element with the symbol Cu from Latin.
This product rapidly reacts with oxygen in the air to. It is a soft malleable and ductile metal with very high thermal and electrical conductivityA freshly exposed surface of pure copper has a pinkish-orange colorCopper is used as a conductor of heat and electricity as a building material and as a constituent of various metal alloys such as sterling. Sodium chloride lead 11 nitrate lead 11 chloride sodium nitrate 2NaC1 PbC12 2NaN03 iron chlorine iron 111 chloride 2 Fe 2FeC13 barium water barium hydroxide hydrogen Ba 21-120 12 When chlorine gas reacts with methane carbon tetrachloride and hydrogen chloride are produced.
Its also very resistant to corrosion. Zinc oxideeugenol cement Fig. Hazardous Substances Data Bank HSDB ZINC NITRATE EXPLODES WHEN.
Percent yield represents the ratio between what is experimentally obtained and what is theoretically calculated multiplied by 100. Devardas alloy CopperAluminiumZinc is a reducing agentWhen reacted with nitrate in sodium hydroxide solution ammonia is liberated. Van Nostrand Reinhold 1996 p.
First of all zinc helps the plants reach optimal size and maturity as well as the production of leaves stalks stems and branches. When iron reacts with copper sulphate blue a green colour is obtained due to the formation of iron oxide. Zinc silver nitrate --- zinc nitrate and silver metal.
We will be publishing Issue 1 of The School STEM Technician at noon on the 1st December 2021 via the SSERC website. Negative ions that consist of a single atom are named by adding the suffix -ide to the stem of the name of the element. In 12 sentences explain the.
The second part. CC14 4 HCI When sodium oxide is added to water sodium hydroxide is produced. A student wants to make copper chloride crystals.
The zinc oxideeugenol cements are used to provide a sedative. Copper hydroxide reacts with sulfuric acid to form copper sulfate and water. Deficiencies in auxin levels can cause reduced development speeds.
Iron copper nitrate --- iron nitrate and copper metal. Can react violently with carbon copper metal sulfides organic matter phosphorus sulfur. Red-brown copper will be left.
2AlBr 3 3 Cl 2 2AlCl 3 3Br 2 Single Replacement 3. The chemical reaction between lead nitrate and potassium iodide is accompanied by the formation of a yellow precipitate of lead iodide. A salt is any compound formed by the neutralisation of an acid by a base.
Bromine potassium iodide --- potassium bromine and iodine. The student adds excess copper oxide to some hot acid. Zinc and Lead II nitrate react to form Zinc Nitrate and Lead.
ZINC NITRATE is an oxidizing agent. NH 4 ammonium. Zn PbNO 3 2 ZnNO 3 2 Pb Single Replacement 2.
This is our newest publication and has been created to support the school technician profession in Scotland. Before running the experiment the scientist expected the products of the reaction will be a total of 159 grams of copper sulfate Cu2S. The rosin increases fracture resistance and the zinc acetate is effective in accelerating the reaction rate.
1 mark Tick one box. Here colour change is seen. Pour the cool residue into a 100 cm 3 beaker and add a little dilute hydrochloric acid to dissolve the zinc oxide and also any unreacted zinc and copper oxide warming if necessary.
You want to measure how much water is produced when 120 g of glucose C_6H_12O_6 is burned with enough oxygen. Thermal decomposition is used to obtain zinc oxide from its carbonate ore. Metals combined with acids are almost always single replacement chemical.
Instantly reacts with atmospheric oxygen to produce nitrogen dioxide. 0 Suggest how the student would know that excess copper oxide has been 2. Therefore none of zinc copper or lead will displace magnesium ions from solution so that there will be no reaction.
When one chemical reacts to a stimulus by breaking into two simpler elements. The liquid is a preparation of eugenol which reacts with the powder to form an amorphous chelate of zinc eugenolate. In this reaction all of the copper and all of the sulfur react.
Is copperII nitrate soluble or insoluble. This can be rinsed with water and. So here on adding zinc to CuSO4 solution zinc displaces copper from copper sulphate forms zinc sulphate solution.
Bronze is also used frequently in ship-building because it is resistant to corrosion from sea water. The first part comes from the metal metal oxide or metal carbonate. Yield actual yieldtheoretical yield 100 So lets say you want to do an experiment in the lab.
0 2 1 Which acid should the student use. Limiting reagent is the reactant which limits the progress of a chemical reaction. Copper hydroxide is also called cupric hydroxide.
H 3 O hydronium. Copper hydroxide -CuOH2 - Copper hydroxide is a crystalline inert compound used in the preparation of a wide variety of salts. Calcium water --- calcium hydroxide and dihydrogen.
CopperI Cu 2 copperII Polyatomic positive ions often have common names ending with the suffix -onium. Saxs Dangerous Properties of Industrial Materials. At the same time the nitrate ions NO 3 undergo a series of reactions to form nitrogen monoxide NO.
Aluminum Bromide and Chlorine gas react to form Aluminum Chloride and Bromine gas. 2Na 3 PO 4 3 CaCl 2 6NaCl Ca 3 PO 4 2. EPA Pesticide Chemical Code 076102.
Zinc and lead two nitrate make zinc nitrate and lead metal. Iodine crystals and chlorine gas react to make solid iron and carbon dioxide gas. Zn Pb NO 3 2 ZnNO 3 2 Pb.
Zinc Deficiency Zinc has a lot of functions in a plants health. The above reactions should be written in your notebook and all observations should be related to the compounds which appear in those reactions. 2NOg O 2 g.
If two atoms must combine at a 1 to 1 ratio but there is an unequal amount of 1 atom then the reaction will stop when the atom with less quantity runs out. The name of a salt has two parts. Oxidizing Copper Metal with Concentrated Nitric Acid HNO 3 aq The first step involves transforming Cu metal to copperII ions Cu2 using concentrated nitric acid HNO 3 aq.
Sodium Phosphate and Calcium Chloride react to form Calcium Phosphate and Sodium Chloride. Nitric acid copper2 salt. Explanation - Zinc is more reactive than copper.
Cuprum and atomic number 29. And although heavier than aluminum its also twice as strong. Reacts violently with combustible and reducing materials.
Adding sodium oxide to water produces sodium hydroxide. Heat this to show that the white powder zinc oxide is yellow when hot and white when cool. Answer 1 of 17.
Zinc copper and lead do not react with magnesium nitrate because in the activity sequence magnesium is higher because it is more reactive than other metals. CuSO4 solution has a blue colour while ZnSO4 solu. Zinc is a vital nutrient in many enzymes and the growth hormone auxin.
Chlorine gas reacts with methane and carbon tetrachloride to produce hydrogen chloride. Copper Cu reacts with sulfur S to form copper sulfide as shown in the equation. Nitric acid copper2 salt 21 9TC879S2ZV.
This is indicated by colour change from blue to colourless. A scientist adds 127 grams of Cu to 32 grams of S to start the reaction. 212 contains zinc oxide rosin and zinc acetate in the powder.
Formation of copperII hydroxide While stirring the solution from the last step with a glass rod add 30 mL. The student stirs the mixture. Nitric acid copper salt.
15 Pentanedial 50 Solution. Does nitrate ion form precipitates with cations.

Zinc Chloride Solution At Best Price In India
Search results for TISAB SOLUTION.

Zinc chloride solution colour. Fluoride Ionic Strength Adjustor ISA 378 L Contact Us Login. So when anion vary color of preciptate also vary. If the resulting colour does not persist continue to add the reagent dropwise until the observed colour pervades the entire solution.
The test-tube which contains sodium chloride solution is 1 A2 B3 C4 D Asked on 11th Feb 2021 When dilute sulphuric acid is added to granulated zinc placed in a test-tube the observation made is 1 the surface of the metal turns shiny2 the reaction mixture turns milky3 odour of sulphur dioxide is observed4 a colourless and odourless gas evolves with bubbles. Zinc is added to toothpastes and mouthwashes as zinc chloride or zinc citrate. PH Phosphate Phosphorus Potassium Refractometry Relative Humidity.
The pH of an aqueous solution of zinc chloride with a concentration of 6M is 1. When ZnCl 2 is dissolved in water the resulting solution is acidic in nature. 1 Zinc chloride Zinc chloride ZnCl 2 is soluble in excess of ammonium hydroxide.
Hach India Chemistries INQUIRY. Ascorbic Acid Vitamin C Potassium Hydroxide 46-48 wv Solution. Testing by adding barium chloride or nitrate solution ChromateVI ions will give a yellow precipitate of barium chromateVI.
Phenolphthalein turns colourless to pink 2. PbI 2 is yellow colour precipitate and PbCl 2 is white precipitate. The precipitate colour is very similar to the background colour I use on Chemguide pages which makes the last diagram a bit difficult to see.
Brown colour of gas is characteristic of NO 2. Cation Flame test colour caesium blue rubidium violet strontium red tantalum blue Table 1 Table 2 shows the results of three tests used to identify anions in solution. Observe the colour produced when 1 drop of 5 iron III chloride solution is added.
Also this is same for. The core of a1-cbiss success is built with a strong emphasis on engineering design and technical experience supported by the industrys largest service network in the UK. EDTA Disodium Salt.
Exceptions occur when the anion can form a complex such as in the case of zinc sulfate where the complex ZnH 2 O n SO 4 may be formed log K ca. Sodium Aluminate 38 Solution. This compound reacts with ammonia to form complexes.
Pol y Aluminum Chloride becomes unstable when stored or transported for some time at temperatures higher than 40 d egree C. Zinc is a relatively non-toxic noncumulative essential trace element Zinc inhibits the PTS pathway of glucose uptake by Streptococcus mutans Streptococcus sanguis and Actinomyses naeslundii and the metabolism of glucose to lactic acid. Calcium Hypochlorite 70 Granular Calcium Oxide Powder Calcium Nitrate Tetrahydrate Boric Acid Powder Brine Tanks.
Lets consider lead 2 ion. Lead Lux Magnesium Manganese Molybdenum Nickel Nitrate Nitrite Nitrogen ORP Ozone. 91 9916040249 - e-Shop.
Alkaline Non Cyanide Zinc Process contains lower concentration zinc metal concentration 6-14 gL 08-19 ozgal or higher zinc metal concentration 14-25 gL 19-34 ozgal provides superior plate distribution from high current density to low current density or throwing power when compared to any acidic baths such as chloride based Low ammonium chloride Potassium chloride Ammonium. We will be publishing Issue 1 of The School STEM Technician at noon on the 1st December 2021 via the SSERC website. Usually only 1 drop of the iron III chloride solution is necessary.
Answer 1 of 5. Copper sulphate blue stone blue vitriol are all common names for pentahydrated cupric sulphate CuSO 4 5H 2 O which is the best known and the most widely used of the copper salts. Colourless White gelatinous ppt.
With an excess of NH 4 OH ppt. Ferric chloride - FeCl 3 - brown or colour. Zinc compounds like those of main group elements are mostly colourless.
At the end point the chromate ions combine with silver ions to form the sparingly soluble brick-red silver chromate. They are each made of a ca. ZnCl 2 2NH 4 OH ZnOH 2 2NH 4 Cl.
On this basis we fabricated PSCs by adding isopropylammonium chloride to the perovskite precursor solution for the one-pot process and achieved a certified power conversion efficiency of 239. CCl 4 l 4. Phenolphthalein remains colourless 4.
Chemical Properties of Zinc chloride. Structure of solid basic zinc acetate Zn 4. I will give the lowest level of explanation and then follow up with a high level explanation.
According to the anion. ZnOH 2 2NH 4 Cl 2NH 4 OH excess ZnNH 3 4Cl 2 4H 2 O. Anion Test and Result Hydrochloric acid added Magnesium chloride solution added Methyl orange added carbonate effervescence white precipitate forms yellow chloride no change no.
Indeed it is often the starting raw material for. In this case both of the reactants are salts. Blue litmus solution turns red 1 Question 3 The compound which is a non-electrolyte.
Place crosses in two boxes to show the names of two other metals that could be safely used to make hydrogen. Watertest Systems will be closed from December 23rd until January 4th. Yellow residue indicates presence of either Cu or Pb or Zn With a few drops of sodium hydroxide solution a white precipitate appeared.
All nitrate compounds are soluble in water. A zinc sulfate B zinc sulfide C zinc sulfite D zinc sulfur ii The student could have used other metals in this experiment. Sodium Benzoate FCC.
This is our newest publication and has been created to support the school technician profession in Scotland. H 2 SO 4 dil 3. A positive test will be a distinct burgundy or magenta.
75GPD under the sink RO Membrane Cartridge. Molten zinc chloride is highly viscous and has a relatively low electrical conductivity value. Each 5ml contains protien hcl 20 0333gm pyridoxine hcl 050mg niacinamide 15mg iron choline citrate 15mg magnesium chloride ip 3333mg manganese chloride usp 00333mg zinc sulphate 44mg lysine hcl 20mg 3333mg5ml magnesium chloride.
In the titration of a neutral solution of chloride ions with silver nitrate a small quantity of potassium chromate solution is added to serve as the indicator. Please place orders before the 5th of December to ensure delivery. When you get questions like this it helps a lot if you can see the general sorts of patterns.
Carbon Dioxide Chloride Chlorine Chlorine Dioxide Chromium COD Colour Conductivity TDS Copper Cyanide Cyanuric Acid. PAC tends to hydrolyse to a white turbid solution and loses effectiveness when it is kept long a diluted solution of less than approximately 3 as Al2O3. This indicates presence of Cu C u 2 a q 2 O H a q C u O H 2 white ppt.
Methyl orange remains orange 3. Uses of Copper Sulphate. That means there are no precipitates of nitrate compounds.
This is a case of fractional precipitation the two sparingly soluble salts being AgCl Ksp 12 x 10 -10 and Ag 2. A1-cbiss sell gas detection equipment and continuous emissions monitoring systems. Dissolved Oxygen Formaldehyde Fluoride Glycol Hardness Hydrazine Hydrogen Peroxide Hypochlorite Iodide Iodine Iron.
Password cAsE sEnSiTiVe Forgot your login info. 2 A copper B iron C magnesium D potassium E silver dilute sulfuric acid hydrogen water zinc P41554A0732 7 Turn over b Describe a test to show that the gas. The colour change observed when the solution of magnesium hydroxide is tested with the following indicators.
Zinc salts are usually fully dissociated in aqueous solution. Polypropylene Filter Cartridge. CH 3 COOH aq 1 2 Question 4 Twice.
Compare the colour with that produced in test a.