Chemistry Answer Ion Chemical Bond. The octet rule has been satisfied.
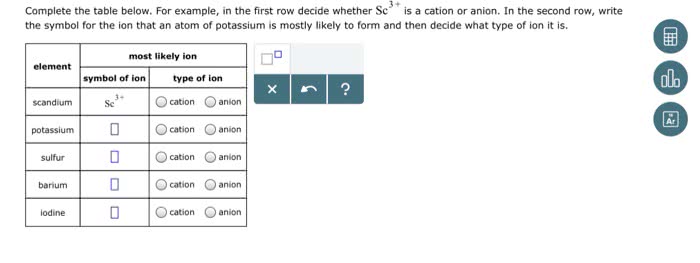
Oneclass 3 Complete The Table Below For Example In The First Row Decide Whether Scis A Cation Or
Where the element is most commonly found in nature and how it is sourced commercially.

Most likely barium ion. From this fact it is apparent that most of the space of a crystal is occupied by the anion and that the cations fit into the spaces between them The terms anion and cation for ions that respectively travel. An ion ˈ aɪ ɒ n-ən is an. Ca Ca 2 2 e-These.
Dilithium monoxide would be the name if Li2O were a covalent compound a compound composed of only nonmetals. Generally found in an ion exchange unit activated aluminum removes magnesium and calcium which are two minerals that cause hard water It can also remove fluoride and arsenic if water spends enough contact time with this media type. Expressed as density of organisms per 100 ml.
This not only improves its resistance to reducing acids such as sulphuric hydrochloric and phosphoric but also raises the critical temperature at which crevice corrosion in sea water can occur. However once the two 3s e- are removed then Mg has an electron configuration of a noble gas Ne. First ionisation energy The minimum energy required to remove.
The energy released when an electron is added to the neutral atom and a negative ion is formed. The outermost shell of the sodium ion is the second electron shell which has eight electrons in it. Why do people need health supplements.
Therefore it is unwilling to give up the next e- as. ASHAs Practice Portal assists audiologists and speech-language pathologists in their day-to-day practices by making it easier to find the best available evidence and expertise in patient care identify resources that have been vetted for relevance and credibility and increase practice efficiency. No ads no money for us no free stuff for you.
High-osmolar contrast medium 2. Seemly Point of view Nen. The two colours in the image represent the two elements that collide to form livermorium calcium and curium.
This table gives an idea of the relative toxicity of various metals. Cations are small most of them less than 10 10 m 10 8 cm in radius. Because lithium is assumed to form 1 ions in compounds we do not need to indicate the charge of the metal ion in the compound.
Depending on the type of activated aluminum it can filter out radium and barium. Notice that sulfate SO42- is a polyatomic ion and that potassium exists as a 1 ion K. 2016 states that most of the research on high-valent ions is done on non-aqueous magnesium ca.
However in barium titanate BaTiO 3 shown in the figure the central Ti 4 cation can be induced to move off-centre leading to a noncubic symmetry and to an electrostatic dipole or alignment of positive and negative charges toward opposite ends of the structure. Get the Best Supplements from Organika. 81 15 with zinc 3 with calcium and 1 with others including aluminum as well as strontium and barium.
Also this is an. Mercury lead and cadmium are not required even in small amounts by any organism. Calcium for example loses two electrons to form Ca 2 ions when it reacts with water.
Most of the chemistry of the alkaline-earth metals. Please do not block ads on this website. The products of these reactions are what we might expect.
Most probable number MPN that number of organisms per unit volume that in accordance with statistical theory would be more likely than any other number to be yielded with the greatest frequency in a specific test. Bas O 2 g BaO 2 s The more active members of Group IIA Ca Sr and Ba react with water at room temperature. Risk of leakage is the same for 18G and 20G 1.
You Better Know These Three Natural Medicines To Help You In Several Ways. Most significant things to know about Tramadol Tramadol COD option. CeNa rightarrow Na e- The cation produced in this way Na is called the sodium ion to distinguish it from the element.
Since barium is a 2 ion the barium phosphate will have a 32 ratio of BaPO4 in order to balance the charge. Barium sulfate is a heavy metal with a high atomic number Z56 and a K shell binding energy K-edge of 374 keV very close to that of most diagnostic x-ray beams. Mg is more likely to become a cation with a 2 charge by giving up 2 e- from the 3s sublevel.
Note that phosphate PO43- is a polyatomic ion and will not break apart. Activated aluminum is also referred to as a resin. The image features an abstract form inspired by images from NIF Target Chamber at the Lawrence Livermore National Laboratory.
Uses and properties. Prewarming of high-osmolar 370 mgmL contrast to 37C lowers the viscosity and has been proven to lower the. Chlorine is a highly reactive gas.
It is a naturally occurring element. Cheap levitra prices generic vicodin xanax. Mg more readily gives up these two e- thus requiring low IE to remove them.
A neutral sodium atom is likely to achieve an octet in its outermost shell by losing its one valence electron. The table below gives the name atomic number electronic configuration of the atom the first second and third ionisation energy melting point density and electronegativity of the Group 2 elements alkaline-earth. A recent review by Canepa et al.
A Phenoxide ion is a stronger base than ethoxide ion b Phenoxide ion is stabilized through delocalization c Phenoxide ion is less stable than ethoxide ion d Phenoxide ion is bulkier than ethoxide ion 16. Due to these characteristics barium is an ideal medium for the absorption of x-rays. Results are computed from the number of positive findings of coliform-group.
Table of Data for Group 2 Elements. This principle of palladium additions is now being extended to some of the. The ionization constant of phenol is higher than that of ethanol because.
160 orthorhombic tetragonal and cubic forms depending on temperature. In the idealized cubic unit cell of such a compound the type A atom sits at cube corner position 0 0 0 the type B atom sits at the body-center position 12 12 12 and oxygen atoms sit at face centered. Federal Register 56 110.
Double replacement H2SO4 2KOH 2H2O K2SO4. What is the most likely ion for magnesium to become when it is ionized. Because metals are rather insoluble in neutral or basic pH pHs of 7 or above give a highly misleading picture of the degree of metal pollution.
This dipole is responsible for the ferroelectric properties of barium titanate in which domains of neighbouring dipoles line up. ATOMS MOLECULES AND IONS b. Calcium strontium and barium can also form peroxides.
Likely related to a smaller amount of subcutaneous tissue and the fact that veins are more fragile in these regions. But most anions are large as is the most common Earth anion oxygen. Only if smaller caliber access over 22G is used.
Where the element is most commonly found in nature and how it is sourced commercially. The gauge of intravenous catheter. The correct order of boiling points for primary 1 secondary 2 and tertiary alcohol 3 is.
Barium titanate is an example of a perovskite which can take on the rhombohedral space group R3m no. Barium sulfate is essentially not absorbed from the GI tract nor metabolized in the body. One of the most successful of these involves the addition of small amounts of palladium to the commercially pure material.
The use of high-valent ions increases the number of electrons involved in the electrochemical process and thus in principle leads to high capacity. New Report suggests Facebook CEO Mark Zuckerberg knew. Commercial Kitchen Equipments Manufacturer.
The largest users of chlorine are companies that make ethylene dichloride and other chlorinated solvents polyvinyl chloride resins chlorofluorocarbons and propylene oxidePaper companies use chlorine to bleach paper. Electronegativity Pauling scale The tendency of an atom to attract electrons towards itself expressed on a relative scale.
Properties of Barium Chloride BaCl 2. Barium chloride is poisonous in nature.

How Can Poisonous Heavy Metals Be Perfectly Safe American Council On Science And Health
Barium sulfate is used as an insoluble additive to oil well drilling fluid as well as in a purer form as X-ray radiocontrast agents for imaging the human gastrointestinal tract.
Is barium poisonous. It is an important precursor to other barium compounds including BaCO 3 and the pigment lithopone ZnSBaSO 4. Often your veterinarian will recommend not feeding your dog for about 24. Phosphine PH 3 is a spontaneously flammable and explosive poisonous colorless gas with the foul odor of decaying fish.
Boiling point of Barium Chloride. Barium or bismuth medicines to treat diarrhea oily laxatives such as castor oil. 3856 gcm 3 anhydrous Melting Point of Barium Chloride.
Like other chalcogenides of the alkaline earth metals BaS is a short wavelength emitter for electronic displays. Arsenic despite its poisonous reputation may be a necessary ultratrace element for humans. To determine the toxicity of arsenic.
It has a similarly colored vapor with an offensive and suffocating odor. It is colorless although like many sulfides it. Where paraquat is found and how it is used.
Barium study endoscopy colonoscopy and ultrasound are other options that may be considered depending upon what is seen during the examination. Heavy metals are naturally occurring elements that have a high atomic weight and a density at least 5 times greater than that of water. M The most common element on Earth is oxygen about 50.
It is unfortunately no longer in print and I dont have it my school library. Barium compounds are added to fireworks to impart a green color. It is a really interesting book about the element with arguably the most interesting history.
A deficiency results in inhibited growth. Barium-aluminates are utilized for water purification acceleration of concrete solidification production of synthetic zeolites and in the paper and enamel industries. After determining if there is a health condition causing your dogs vomiting as well as what that condition is your veterinarian will provide treatment recommendations.
It is reported that at present people are using crackers that are made of poisonous chemicals like Sulphur Nitrogen oxide Manganese Barium etc. In fact the metal is a getter in vacuum tubes meaning its used to remove the last traces. It is so reactive that glass metals and even water as well as other substances burn with a bright flame in a jet of fluorine.
And I live very healthily She says her dog has been poisoned by a radioactive metal. It is the only nonmetallic element that is liquid under ordinary conditions it evaporates easily at standard temperature and pressures in a red vapor that has a strong disagreeable odor resembling that of chlorine. Fact sheets on a range of health and safety topics have been developed in partnership by Sydney Childrens Hospital The Childrens Hospital at Westmead and Hunter New England Kidshealth Network.
A chemical compound is a chemical substance composed of many identical molecules composed of atoms from more than one element held together by. Soluble Barium compounds are highly toxic and may be used as insecticides. Fluorine readily forms compounds with most other elements even with the noble gases krypton xenon and radon.
The Elements of Murder book just deals with a few highly poisonous elements used for dastardly purposes. M Pradyu Kannur. Fluorine is an univalent poisonous gaseous halogen it is pale yellow-green and it is the most chemically reactive and electronegative of all the elements.
Nitroglycerin nitroglycerine trinitroglycerin glyceryl trinitrate 來. In the preparation of HNO 3 we get NO gas by catalytic oxidation. Structure of Barium Chloride BaCl 2 Structure of Barium Chloride.
11 Na Sodium Atomic number Symbol Name An element is made up of atoms which are all the same. On heating ammonium dichromate and barium azide separately we get i N 2 in both cases ii N 2 with ammonium dichromate and NO with barium azide iii N 2 O with ammonium dichromate and N 2 with barium azide iv N 2 O with ammonium dichromate and NO 2 with barium azide 18. Example sodium a very reactive metal can combine chemically with chlorine a poisonous gas to form sodium chloride a chemically harmless white powder which we call salt.
Periodic tales The Curious lives of. Sulfuric acid barium hydroxide --. Raising concerns over their potential effects on human health and the environment.
Paraquat from outside the United States may not have these safeguards added. It is a necessary ultratrace element for red algae chickens rats goats and pigs. 20823 gmol anhydrous Density of Barium Chloride.
Attempt Free Mock Test on General Awareness. BaS is the barium compound produced on the largest scale. Although Barium is poorly absorbed orally less than 5 it can be very high in peanuts and peanut butters about 3000 nanogramsgram as.
If you can find The shocking History of Phosphorus by him do get it. Conveniently barium which is a soft silvery metallic alkaline earth metal is never found in nature in its pure form due to its reactivity with air or in water. Toxicity definition the quality relative degree or specific degree of being toxic or poisonous.
It reacts with water acids and. They aim to help you learn about your childs illness and ways you can help improve their overall health and wellbeing Factsheets are for educational purposes only please consult. Your health care provider will give you instructions for picking up your specimen containers for collecting your sample.
Chemical Compounds List Their Common Names and Formulas Chemical Compound. Strychnine is a white odorless bitter crystalline powder that can be taken by mouth inhaled breathed in or mixed in a solution and given intravenously injected directly into a vein. Regular crackers emit poisonous gases while green ones emit water vapour said Dr B L Sujatha Rathod Director of Minto Hospital.
Phosphine is a dangerous fire hazard and ignites in the presence of air and oxidizers. M The least common naturally occurring element is Astatine. Their multiple industrial domestic agricultural medical and technological applications have led to their wide distribution in the environment.
You will also need a clean collection device such as a shallow pan plastic bag or clear plastic wrap to place over the toilet seat in which to collect your sample before. Water-soluble barium compounds are poisonous and have been used as. I was toxic in aluminium barium strontium arsenic manganese.
Used in making explosives and medically as a vasodilator trade names Nitrospan and Nitrostat syn. Iron sulfide hydrogen chloride --- iron chloride and hydrogen sulfide poisonous gas lead nitrate potassium iodide --- lead iodide and potassium nitrate saltpeter sodium bicarbonate baking soda vinegar --- carbonic acid and sodium acetate. Russ claims he found six times the safe.
Unlike barium sulfate barium carbonate dissolves in stomach acid releasing the poisonous barium to do its rather nasty but efficient work. When mixed with combustible materials barium sodium and potassium peroxides form explosives that ignite easily. Go green this Diwali.
Because paraquat is highly poisonous the form that is marketed in the United States has a blue dye to keep it from being confused with beverages such as coffee a sharp odor to serve as a warning and an added agent to cause vomiting if someone drinks it. Reactions of arsenic as the element with air water halogens. Molecular Weight of Barium Chloride.
The liquid can cause frostbite. If fireworks have to be used the hospital recommended. A heavy yellow poisonous oily explosive liquid obtained by nitrating glycerol.
At ambient temperature bromine is a brownish-red liquid. Barium sulfide is the inorganic compound with the formula Ba S.
What mass of titanium can be obtained from 5000 g of ilmenite. AgaqClaq AgCls All spectator ions are removed.

Barium Chloride Formula Definition Concepts And Solved Examples
Barium chromate named barium tetraoxochromateVI by the IUPAC is a yellow sand like powder with the formula BaCrO 4.
Water content of barium chloride formula. A sample of 08360 grams of an unknown compound containing barium ions Ba2 is dissolved in water and treated with an excess of Na2SO4. This chemical with formula of Cl is one of chemical that found in water pollutant especially in waste of salt storage areas sewage effluent animal manure and industrial waste. The decomposition of solid potassium chlorate leads to the formation of solid potassium chloride and diatomic oxygen gas.
What happens when copper sulphate reacts with hydrogen sulphide. Calcium chloride is an inorganic compound a salt with the chemical formula CaCl 2It is a white colored crystalline solid at room temperature and it is highly soluble in water. How many grams of Br are in 195 g of CaBr_2.
It is expressed in milligrams per liter of equivalent calcium carbonate mgl CaCO3. Add ammonium hydroxide with constant stirring until solution is alkaline to litmus let precipitate settle and wash by decantation with water until wash-water gives only slight test for sulphate with barium chloride solution. Solvent is water H 2 O because this is an aqueous solution.
The Process of Dissolving. Step 3 Extract the data from the question mass of solute volume of solution mass solute NaCl 5 g. Equation Type Example 771a Example 771b.
NaI and NH42SO4 are both soluble. In the formula of water oxygen O is written. However the chemical formula does not show the type of particles in the compound.
In this way 8 converts to. What is the percent composition by weight of Al in Al_2SO_43. Erythrosine consists essentially of disodium 2-2457-tetraiodo-3-oxido-6-oxoxanthen-9-yl benzoate monohydrate and subsidiary colouring matters together with water sodium chloride andor sodium sulphate as the principal uncoloured components.
The chemical formula of ilmenite is FeTiO_3. Volume of solution 250 mL. Lead can enter drinking water when plumbing materials that contain lead corrode especially where the water has high acidity or low mineral content that corrodes pipes and fixtures.
It can be created by neutralising hydrochloric acid with calcium hydroxide. 7131 Liquid or Strained Honey. Pour off excess water and store residual cream in stoppered bottle.
In contrast squid ink and other cephalopod inks is composed mainly of melanin and mucus with an assortment of other compounds in a variety of concentrations. G The table shows the formula and colour of three different types of cobaltII chloride. In the case of water dissolving sodium chloride the sodium ion is attracted to the partial negative charge of the oxygen atom in the water molecule whereas the chloride ion is attracted to the partial positive hydrogen atoms.
Products Building Blocks Explorer Technical Documents Site Content Papers Genes. Colorful fireworks often involve the decomposition of barium nitrate and potassium chlorate and the reaction of the metals magnesium aluminum and iron with oxygen. Even though have mild effect on humans body and excessive intake of chloride water could cause poisoning.
It is antagonistic to all muscle depressants no matter whether they act primarily on nerve or muscle. No net ionic equation. In order to suppress the afterglow of phenolic resins use is made of compounds such as aluminium chloride antimony trioxide and organic amides.
Barium is used in many industrial processes as well as in diagnostic testing fireworks and pesticides. If the mass of the BaSO4 precipitate formed is 10366 grams. This chemical mostly found polluted a well water.
Millipore 5 Sigma-Aldrich 697 Supelco 9 Boiling Point C Feature. Stearoyl-CoA Desaturase 1 Inhibitor MF-438 CAS 921605-87-0 is a cell-permeable inhibitor of Stearoyl-CoA Desaturase 1 SCD1. When an ionic salt such as sodium chloride shown in A comes into.
Water is a compound made up of 2 atoms of hydrogen element and 1 atom of oxygen element so the formula of water is written as H 2 O. Salts 63 stable isotopes 48 kits 31 buffers 29 building blocks 29 solvents 29 Show More. In homes with lead pipes that connect the home to the water main also known as lead services lines these pipes are.
32 The Lifetime Health Advisory for manganese contains a precautionary statement that for infants younger than 6 months the lifetime Health Advisory of 03 mgL be used even for an acute exposure of 10 days because of the concerns for differences in manganese content in human milk and formula and the possibility of a higher absorption and lower excretion in young infants. Write the formulas of barium nitrate and potassium chlorate. Given a water analysis strong acids manifest themselves in the presence of non-carbonate anions such like sulfate chloride nitrate.
Write an equation for the reaction. Barium stimulates striated cardiac and smooth muscle regardless of innervation. On the other hand strong bases are indicated by monovalent cations eg.
This chemical is also found in gas drilling waste. It is a known oxidizing agent and produces a green flame when heated a. Zinc and barium salts of boric acid and aluminium hydroxide also find frequent application 22.
FeSO 4 is the chemical formula of ironII sulfate or ferrous sulfate normally found complexed with water as a salt with the formula FeSO 4 xH 2 O and has been used in the manufacture of iron gall ink and other inks for centuries. Melting Point C Physical Form. In the formula H 2 O the subscript 2 indicates 2 atoms of hydrogen.
What is the percent water in the compound barium chloride dihydrate. When an ionic salt such as sodium chloride shown in A comes into. The capacity of water to neutralize acids a property imparted by the waters content of carbonate bicarbonate hydroxide and on occasion borate silicate and phosphate.
Solute sodium chloride NaCl. The most common sources of lead in drinking water are lead pipes faucets and fixtures. The Process of Dissolving.
This enables the compound to interact with the solvent. Formula Chemical Formula Aluminum Oxide Benzene Latex Charcoal Ammonia Ink Boron Trioxide Lithium Hydride Crude Oil Barium Sulfate Iron Sulfide Calcium Bromide Luminol Lye Glue Cyanoacrylate Hydrogen Peroxide Al2O3 C6H6 C5H8 C7H4O NH3 FeSO4 B2O3 LiH C9H20 BaSO4 FeS CaBr2 C8H7N3O2 NaOH C5H5NO2 H2O2 CRAFTABLE COMPOUNDS Here are the compounds you can create. Ships Today 387 Product Category.
IC50 of 23 nM. Initial stimulation of contraction leads to vasoconstriction through direct action on arterial muscle peristalsis through action on smooth muscle tremors and cramps through action on the skeletal muscles and. Barium is a naturally occurring alkaline metalloid element with atomic symbol Ba atomic number 56 and atomic weight 137 that is only found in combination with other elements typically barite barium sulfate and witherite barium carbonate or chemicals.
Formula Colour CoCl 2 blue CoCl 22H 2O purple CoCl 26H 2O pink When water is added very slowly to solid CoC 2 the colour of CoCl 2l changes from blue to purple and then to pink. What is the percentage composition of NH_4I. Calcium chloride is commonly encountered as a hydrated solid with generic formula CaCl 2 H 2 O x where x 0 1 2 4 and 6.
This enables the compound to interact with the solvent. In the case of water dissolving sodium chloride the sodium ion is attracted to the partial negative charge of the oxygen atom in the water molecule whereas the chloride ion is attracted to the partial positive hydrogen atoms. A typical precipitation reaction occurs when an aqueous solution of barium chloride is mixed with one containing sodium sulfate.
I Write a chemical equation for the change from the purple solid to the. Step 4 Check the units for consistency and convert if necessary mass in grams volume in millilitres mass solute NaCl 5 g no unit conversion needed volume. Erythrosine is manufactured by iodination of fluorescein the condensation product of resorcinol and phthalic anhydride.
The mixture of Na2CO3 and NaHCO3 weighs 220g.