Acetic acid anhydride CH 3. The total gas flow across the bubbler should be as low as possible pressure and concentration of the absorbing substance.
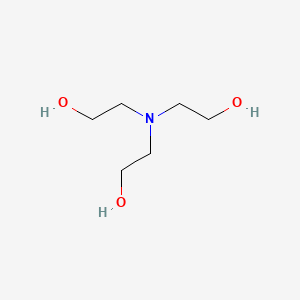
Triethanolamine C6h15no3 Pubchem
Roshan Shahid Zubair ZM.

Triethanolamine vapor pressure. Therefore the plasticizer should have low vapor pressure and a low rate of diffusion in the polymer. The boiling point is defined as the temperature at which the saturated vapor pressure of a liquid is equal to the surrounding atmospheric pressure. Production of Dimethyl Carbonate by Liquid Phase Oxidative Carbonylation of Methanol.
Manganese and potassium both ignite in the vapor Ann. Personal protection equipment should be chosen according to the ANSI standards and in discussion with the supplier of the personal protective equipment. HCFC means a hydrochlorofluorocarbon mentioned in Part 5 of Schedule 10.
Avoid breathing vapor or mist. - the Auto-Ignition Temperature is not the same as Flash Point - The Auto-Ignition Temperature indicates the minimum temperature required to ignite a gas or vapor in air without a spark or flame being present. Pressure Swing Distillation of Benzene and Ethanol.
Basic O-Ring chemical resistance compatibility information is based on isolated generic O-Ring material testing in optimal conditions at room temperature and pressure. MEA has the lowest boiling point and the highest vapor pressure of the amines. Then the amines are separated using a three-step.
Eye contact with concentrated solutions may cause severe eye damage followed by loss of sight. Of positions across the film. Draw vapor-liquid equilibrium VLE phase diagrams for single compounds or binary systems.
The front and back tubes contain molecular sieves impregnated with triethanolamine and the middle or oxidizer tube contains an inert support impregnated with a chromate salt. Handgun has the meaning given in subregulation 4F4. Fight fire from a safe distance with adequate cover.
102-71-6 Triethanolamine - 5 mgm3 TWA - ACGIH Exposure controls Engineering measures. Advice for firefighters. Repeated or prolonged exposures may cause darkening of the skin erosion of exposed front teeth.
A transition from predominantly Ns-doped to NsNi mixts to purely Ni-doped titania was obsd. Direct Insertion Probe. Government Engineering College Valsad.
The majority of plastic products are prepared by so-called hot compounding techniques where the formulated ingredients are combined under heat and shearing forces that bring about a state of molten plastic fluxing which is shaped into the desired product cooled and allowed to. It is important to use a water vapor trap before measuring the outlet gas flow to prevent measurement distortions due to the presence of water in the gas stream after the bubbling process. In accordance with the theory of maturing and aggregation the resulting clusters are susceptible to rapid collision with other clusters of zinc oxide leading to an appropriate concentration of the compound.
After the reaction the distillation of the resulting mixture is carried out first to remove excess ammonia and water. This results in MEA losses of 1-3. Diethanolamine often abbreviated as DEA or DEOA is an organic compound with the formula HNCH 2 CH 2 OH 2Pure diethanolamine is a white solid at room temperature but its tendencies to absorb water and to supercool meaning that it is often encountered as a colorless viscous liquid.
Across an anatase TiO2 thin-film. Siddaganga Institute of Technology Tumkuru. Large Protective Tube Cover.
Bhargav K V And Srivathsa M M. Kamani Parth Bhanushali Miral and Khan Aarif. Specific material compound formulations can.
Production of Methyl Isobutyl Ketone. Exposing rubber O-Ring materials to multiple chemicals and compounding application factors like temperature pressure and gland design can result in significantly different performance. High vapor concentrations may cause skin sensitization.
The boiling point at atmospheric pressure 147 psia 1 bar absolute for some common fluids and gases can be found from the table below. Recycling of flexible PUF from dumped car seating was scrutinized by DEG and potassium acetate KAc under atmospheric pressure at a constant temperature 215225 C. Flash point means the temperature at which petroleum and shale products including kerosene give off an inflammable vapor upon being tested by the Abel Pensky closed test apparatus.
As in any fire wear self -contained breathing apparatus MSHANIOSH approved or equivalent and full protective gear. Residue curves for ternaries. Wear appropriate respirator when ventilation is inadequate.
Cyclohexanone-formaldehyde resin produced when 1 mole of cyclohexanone is made to react with 165 moles of formaldehyde such that the finished resin has an average molecular weight of 600-610 as determined by ASTM method D2503-82 Standard Test Method for Molecular Weight Relative Molecular Mass of Hydrocarbons by Thermoelectric Measurement of Vapor Pressure. An important role in the proposed modification technique is played by TEOH which. Triethanolamine - 5-.
Diethanolamine is polyfunctional being a secondary amine and a diolLike other organic amines. 1atm 1 atm 760 mmHg. Development of hazardous combustion gases or vapor possible in the event of fire.
HANDLING AND STORAGE Handling Put on appropriate personal protective equipment see Section 8. UV-vis λ max 280 nm Refractive index n D 1485 Thermochemistry Heat capacity C 389 J K 1 mol 1. Triethanolamine Uranyl Acetate Dihydrate.
Skin absorption rates as blood concentration-time curves after dermal application of aqueous and neat 14Ctriethanolamine to mouse skin 2000 mgkg bw enclosed by a glass ring showed no significant change with the use of water as the vehicle. Vapor deposition cAPCVD was used to synthesize a film with gradating substitutional Ns and interstitial Ni nitrogen dopant concns. Put on appropriate personal protective equipment.
Exposure to vapor may cause intense watering and irritation to eyes. Temperature classification of equipment in hazardous areas are related. Diethanolamine is formed whereas with three moles of ethylene oxide triethanolamine is formed Fig.
Observe Occupational Exposure Limits and minimize the risk of inhalation of vapors. The Direct Insertion Probe DIP is widely used to introduce low vapor pressure liquids and solids into the mass spectrometer. This is important for analyzing temperature sensitive compounds.
To C 59To F - 32 Note. Eating drinking and smoking should be prohibited in areas where this. Prevent fire extinguishing water from contaminating surface water or the ground water system.
D03AX12 Hazards Safety data sheet. These studies demonstrated that the conversion of NCOO. Vapor pressure diagram p-T bubbledew point xy-T Pressure.
Vapor reacts with barium oxide incandescently Mellor 1940 Vol. Product Boiling Point at Atmospheric Pressure o C Acetaldehyde CH 3 CHO. We believe this to be the first time that such a.
A slow reaction between the vapor and formaldehyde became explosive near 180C Trans. Although the direct insertion probe is more cumbersome than the direct vapor inlet it is useful for a wider range of samples. The Zn 2 ions in reaction with triethanolamine produce clusters of ZnO on the silica surface.
Generating ternary map May take a few minutes Phase diagram. Std enthalpy of formation Δ f H 298 6657 6627 kJ mol 1. The effects of solvent catalyst temperature and the mass ratios on the reaction as well as the properties of the glycolysis products were investigated.
Folding stock has the meaning given in subregulation 4F4. The middle tube is not submitted to the lab for analysis but may undergo a color change indicative of depletion of the oxidizer. Std enthalpy of combustion Δ c H 298 38421 38391 MJ mol 1.
Direct Ionization of Sample. The slow phase half-lives for elimination of triethanolamine in mice after dermal exposure to 1000 and 2000 mgkg bw in acetone were 97 hr and 186 hr. 1 Pa at 20 C Acidity pK a 774.