Write the metal first and the non-metal second. Ammonium sulfide formula NH 4 2 S.
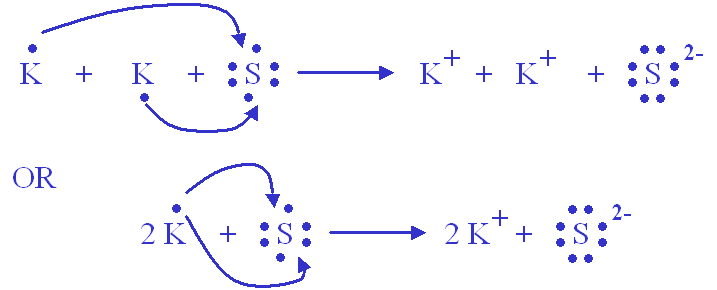
A Potassium Ion Has A Charge Of 1 A Sulfide Ion Has A Charge Of 2 Wha Tis The Chemical Formula For Potassium Sulfide Socratic
People can smell it at low levels.
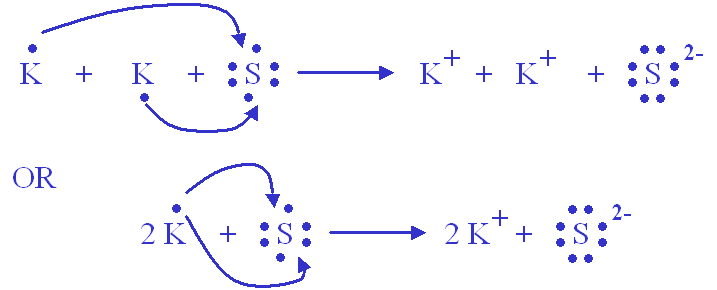
What is the formula for potassium sulfide. Section D Write the formula for the ionic compounds containing transition metals BE CAREFUL TRANSITION METALS MAY HAVE ROMAN NUMERALS and NICKNAMES 1. NH42S ammonium sulfide K3PO4 potassium phosphate ZnNO32 zinc nitrate Fe2SO43 ironIII sulfate or ferric sulfate CuCO3 copperII carbonate or cupric carbonate Note. The chemical formula calculator also contains the names of a range of.
Potassium was first isolated from potash the ashes of plants from which its name derives. Potassium alum is commonly used in water purification leather tanning dyeing fireproof textiles and. The colourless solid is rarely encountered because it reacts readily with water a reaction that affords potassium hydrosulfide KSH and potassium hydroxide KOH.
KMnO4 potassium permanganate Write the chemical formula for each of the following ionic compounds. The names are found by finding the intersection between the cations and anions. N potassium nitride 67 SO 2 sulfur dioxide 68 CuOH copper I hydroxide 69 ZnNO 2 2 zinc nitrite 70 V 2 S 3 vanadium III sulfide Write the formulas for the following chemical compounds.
ZnCH3COO2 zinc acetate 58. 33 potassium hydroxide KOH 34 lead IV sulfate PbSO42 35 silver cyanide AgCN 36 vanadium V nitride V3N5 37 strontium acetate SrC2H3O22 38 molybdenum sulfate MoSO43 39 platinum II sulfide PtS 40 ammonium sulfate NH42SO4 ICP March 23 and 24-Solutions pg. Most commonly the term potassium sulfide refers loosely to this mixture not the anhydrous solid.
Change the final syllable of the non-metal to say ine. 8 7 3 2. The double sulfate of potassium and aluminium with chemical formula KAlSO 4 2.
Barium sulfide BaS 72. Lead IV oxide PbO2 10. S 2 O 3 2-Carbonate.
Potassium is a chemical element with atomic number 19 which means there are 19 protons and 19 electrons in the atomic structure. Mixtures with any perchlorate can explode on impact ACS 146211-212. Hydrogen sulfide can also result from industrial activities such as food processing coke ovens kraft paper mills tanneries and petroleum refineriesHydrogen sulfide is a flammable colorless gas with a characteristic odor of rotten eggs.
C 2 H 4 5. Zinc iron II iron III gallium silver lead IV chloride ZnCl 2. Lead II chloride PbCl2 5.
Sb 3 Cl. C 18 H 35 NaO. The compound is the most important member of the generic class of compounds called alums and is often called simply alum.
Zinc iodide ZnI2 3. Cr 2 O 7 2-Thiosulfate. C 6 H 8 O 6.
C 2 O 4 2-Peroxide. Ionic Compounds Naming and Formula Writing. Sodium sulfate Na 2 SO 4.
The chemical symbol for Potassium is K. It is commonly known as hydrosulfuric acid sewer gas and stink damp. For example the molecular formula of glucose is C_6H_12O_6 and we do not simplify it into CH_2O.
2138 g of a solid mixture containing only potassium carbonate FW 1382058 gmol and potassium bicarbonate FW 1001154 gmol is dissolved in distilled water. Potassium alum potash alum or potassium aluminium sulfate is a chemical compound. A mixture of damp sulfur and calcium hypochlorite produces a brilliant crimson flash with scatter of molten sulfur Chem.
Ionic compounds tend to form crystals with high melting temperatures. Potassium sulfide is an inorganic compound with the formula K 2 S. Compound Name Type of Compound.
Ammonium phosphate formula NH 4 3 PO 4. Learn the properties structure and uses of Potassium permanganate KMnO4 Here. Potassium carbonate 2K CO 3 2- K 2 CO 3.
A mixture with potassium nitrate and arsenic trisulfide is a known pyrotechnic formulation Ellern 1968 p. The first box is the intersection between the zinc cation and the chloride anion so you should write ZnCl 2 as shown. In the periodic table potassium is one of the alkali metals.
It has a role as a bacterial xenobiotic metabolite a marine metabolite an EC 3514 amidase inhibitor an algal metabolite and an Escherichia coli metabolite. Cadmium sulfide CdS 2. O 2 2--3 ions.
Its condensed formula is CH 2 CCH 3CHCH 2. Ionic Compound Formula Writing Worksheet Write chemical formulas for the compounds in each box. What is the correct molecular formula for the compound dinitrogen.
C 5 H 8 is the chemical formula for isoprene which polymers are the main components of natural rubber. 3336 mL of a 0767 M HCl stand. Mg3N2 magnesium nitride 49.
In a formula parentheses are used around a polyatomic ion only when there are 2 or more of that polyatomic ion in a formula unit ie when the subscript is not 1. And for each compound they all have a molecular formula but some can be similar and those are called isomers which are common in organic chemistry. This activity includes every compound formula and name that can be formed from the list 44 Ions provided in Chemistry A at Pickerington High School Central.
Copper II bromide CuBr2 8. Use the atomic number to indicate the number of atoms of each type present in the compound. This way students can see that the ions combine in whole number ratios in order to produce a neutral chemical species.
KCIO potassium hypochlorite 48. Silver nitride Ag3N 9. In a molecular formula it states the total number of atoms of each element in a molecule.
Co2SO43 cobalt III sulfate 60. It is produced naturally by some marine algae. Takes fire spontaneously in chlorine dioxide and may produce an.
Melting Point C Physical Form. Formula writing rules to write the correct chemical formulas for each compound. BaC 2 H 3 O 2 2.
It adopts antifluorite structure which means that. Copy this to my account. Ammonium oxide formula NH 4 2 O.
K2SO4 potassium sulfate 59. Analytical 63 ACS reagent 44 Puriss 35 Cell Culture 21 Reagent 18 Purum 17 Anhydrous 13 BioXtra 13 ReagentPlus 11 Plant 7 Show More. Iron II fluoride FeF2 7.
What is the correct molecular formula for the compound antimony trichloride. Al 2 S 3. B the chemical formula of the compound appears after the arrow.
All of the alkali. E-mail to a friend. Using this program will help you to learn how to write ionic compound names and formulas for Chemistry A.
Potassium carbonate K 2 CO 3 2. 71 silicon dioxide SiO 2 72 nickel III sulfide Ni 2 S 3 73 manganese II phosphate Mn 3 PO 4 2 74 silver acetate AgC 2 H 3 O 2. Potassium Permanganate - KMnO4 is the chemical formula of Potassium permanganate which is most commonly used as an oxidising agent in volumetric analysis.
Copy this to my account. What is the correct molecular formula for the compound boron trifluoride. Dimethyl sulfide is a methyl sulfide in which the sulfur atom is substituted by two methyl groups.
Ionic or Covalent Chemical Formula 1 copper II chlorite 2 sodium hydroxide 3 nitrogen dioxide 4 cobalt III oxalate 5 ammonium sulfide 6 aluminum cyanide 7 carbon disulfide 8 tetraphosphorous pentoxide 9 potassium permanganate 10 manganese III chloride Compound. Ammonium sulfate formula NH 4 2 SO 4. Magnesium nitride Mg3N2 6.
E-mail to a friend. Iron III oxide Fe2O3 4. NH 4 NO 2.
NaMnO4 sodium permanganate 50. Ionic Compound Naming and Formula Writing List 1.
Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structureThe chemical symbol for Hydrogen is H. Chemical Compound Formulas Chemical formulae provide a way to represent any chemical substance using the symbol of the elements present in it.

Phosphorus Pentasulfide 98 1314 80 3
16 magnesium sulfate heptahydrate MgSO 47H 2O.
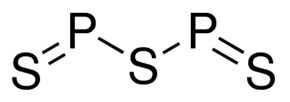
Phosphorus sulfide chemical formula. C 2 H 4 5. Write the correct chemical formula for the following chemical compounds. Sb 3 Cl.
Gum arabic 30 g. An ionic compound is composed of a metal and a non-metal. What is the correct molecular formula for the compound boron trifluoride.
These are limited to a single typographic line of symbols which may include. A chemical formula is a way of presenting information about the chemical proportions of atoms that constitute a particular chemical compound or molecule using chemical element symbols numbers and sometimes also other symbols such as parentheses dashes brackets commas and plus and minus signs. C 5 H 8 is the chemical formula for isoprene which polymers are the main components of natural rubber.
Chapter 9 Honors Chemistry Compound Naming Race - Solutions Be the first team in the room to correctly get all the names on this sheet right. S Calcium sulfide S Silver sulfide Zn P Zinc phosphide Aluminum oxide Sr I Strontium chloride 4. Name _____ Date _____ Period _____ Ionic_Covalent Names.
17 potassium carbonate K 2CO 3. Its condensed formula is CH 2 CCH 3CHCH 2. 11 silicon dioxide SiO 2.
Metals lose electrons to produce positve ions called cations. With a standard atomic weight of circa 1008 hydrogen is the lightest element on the periodic table. Its monatomic form H is the most abundant chemical substance in the Universe constituting roughly 75 of all baryonic mass.
Ionic or Covalent Compound Name 31 VSO 4 2 32 Ag 2CO 3 33 N 2S 3 34 FeSO 3 35 ZnNO 2 2 36 C 6H 12O 6 37 PCl 3 38 MnOH 7 39 NiNO 3 2 40 O 2. Na Mg 2 Non. Under normal conditions sulfur atoms form cyclic octatomic molecules with a chemical formula S8.
Calcium carbonate 5 g. Chemically sulfur reacts with all elements except for gold platinum iridium tellurium and the noble gases. Elemental sulfur is a bright yellow crystalline solid at room temperature.
Ground glass 22. Circle the symbol for the metal in each of the compounds in Model 2. 13 manganese II phosphate Mn 3PO 4 2.
Antimony sulfide 110 g. VanadiumIII sulfide Write the formulas for the following chemical compounds. Chemical formulae provide insight into the elements that constitute the molecules of a compound and also the ratio in which the atoms of these elements combine to form such molecules.
11 silicon dioxide SiO 2 12 nickel III sulfide Ni 2 S 3 13 manganese II phosphate Mn 3 PO 4 2 14 silver acetate AgC 2 H 3 O 2 15 diboron tetrabromide B 2 Br 4 16 magnesium sulfate heptahydrate MgSO 47H 2 O 17 potassium carbonate K 2 CO 3 18 ammonium. The empirical formula is the simplest version of a chemical formula for example C3H8. Tin IV selenide dinitrogen trioxide nickel III sulfide lithium acetate silver acetate phosphorus trifluoride silicon dioxide vanadium V oxide manganese II phosphate aluminum hydroxide ammonium oxide zinc sulfide diboron tetrabromide silicon tetrafluoride magnesium sulfate heptahydrate silver phosphate potassium.
Use the table of ions in Model 1 to answer the following questions. In minerals phosphorus generally. If uniformly distributed in a room 10X10X5 m in size the maximum HBr concentration would be 005 mgL 50 mgcu inch.
Phosphorus is a chemical element with atomic number 15 which. Potassium chlorate 482 Antimony sulfide 37. Barium carbonate 15 g.
Write the chemical formulas for the following binary ionic compounds. Phosphorus tribromide reacts with moisture in the air to produce phosphonic acid and hydrogen bromide gas HBr. Use it to calculate the mass percent of each element on a mole.
Write the formulas for the following chemical compounds. Chemical Formula Type of Compound. In the compound zinc phosphide what is the.
Sodium thiosulfate ____Na2S2O3_____ 3. 12 nickel III sulfide Ni 2S 3. Sodium chloride NaCl and magnesium oxide MgO.
The molecular formula contains information on the actual number of atoms. The chemical formula of ionic compounds can be quickly calculated using the chemical formula calculator. Phosphorus sulfide 15 g.
The chemical formula of a compound is a symbolic representation of its chemical composition. Chemically sulfur reacts with all elements except for gold platinum iridium tellurium and the noble gases. Sulfur dioxide ____SO2_____ 2.
Phosphorus Tribromide PbS LeadII Sulfide PCl3 Phosphorus Trichloride PCl5 Phosphorus Pentachloride PF3 Phosphorus Trifluoride PH3 Phosphine PI3 Phosphorus Triiodide PtNH32Cl2 Diamminedichloroplatinum RaOH2 Radium Hydroxide RbOH Rubidium Hydroxide S2F10 Sulfur Fluoride SbCl3 Antimony Tricloride SCl6 Sulfur Hexachloride SF4 Sulfur. Use the stock form for the transition metals. C 18 H 35 NaO.
The transfer of electrons between metals and non-metals produces charged particles called ions. What is the correct molecular formula for the compound antimony trichloride. Magnesium carbonate 12 g.
Under normal conditions sulfur atoms form cyclic octatomic molecules with a chemical formula S8. The combustion of 829 mg produced 225 mg of CO2 and 461 mg of H2O. How many moles and formulas are in 416 g ammonium carbonate.
Elemental sulfur is a bright yellow crystalline solid at room temperature. The molar mass of a substance also often called molecular mass or molecular weight although the definitions are not strictly identical but it is only sensitive in very defined areas is the weight of a defined amount of molecules of the substance a mole and is expressed in gmol. Greek prefixes are used to indicate the number of atoms of each element in the chemical formula for the compound.
It has a concentration in the Earths crust of about one gram per kilogram compare copper at about 006 grams. 14 silver acetate AgC 2H 3O 2. You should complete this by Sunday.
Give the formula for the following. The information in a chemical formula. Manganese II carbide Mn 2C phosphorus V nitride P 3N 5 gold I iodide AuI nickel III phosphide NiP iron II bromide FeBr 2 copper II sulfide CuS aluminum sulfide Aℓ 2S 3 silicon iodide SiI 4 lead IV carbide PbC aluminum fluoride AℓF 3 arsenic V nitride As 3N 5 mercury I phosphide Hg 3P cobalt III.
A 10 cc cartridge of PBr3 tested as fire suppressant would react to form an estimate 25 g of HBr. Provided below is a list of the chemical formulas of some common chemical compounds along with their molecular weights. Chemical Formula Nomenclature Practice.
A compound containing only C H and O was extracted from the bark of a sassafras tree. 8 7 3 2. Same for ionic compounds such as potassium sulfide K 2 S.
Potassium chlorate 60 g. Skill 3-3 Mass Percent and the Chemical Formula 12 The formula shows the number of moles of each element. For example the chemical formula of water which is H.
Elemental phosphorus exists in two major forms white phosphorus and red phosphorus but because it is highly reactive phosphorus is never found as a free element on Earth. Phosphorus is a chemical element with the symbol P and atomic number 15. If in a 61.
Which element comes first in the name and formula of the compounds in Model 2the metal or the nonmetal. 15 diboron tetrabromide B 2Br 4. Complete these in lab and on your own time for practice.
Phosphorus is a chemical element with atomic number.
Relevant identified uses of the substance or mixture and uses advised against Use of the substancemixture. As such it readily forms compounds notably oxides with almost all other elements.

Hydrogen Sulfide 99 5 7783 06 4
The infusing of unsaturated or impure hydrocarbons with hydrogen gas at controlled temperatures and pressures for the purpose of obtaining saturated hydrocarbons andor removing various impurities such as sulfur and nitrogen.
Hydrogen sulfide gas formula. It is easily ignited. Hydrogen Sulfide or sour gas H2S is a flammable colorless gas that is toxic at extremely low concentrations. Hydrogen sulfide is.
Oxygen is a member of the chalcogen group on the periodic table and is a highly reactive nonmetallic element. Hydrogen is the lightest element. It is heavier than air and may accumulate in low-lying areas.
Please give easy to follow step by step instruction and the answer. Hydrogen ion concentration the normality of a solution with respect to hydrogen ions H. Hydrogen is a colorless odorless gas.
It is combusted in pure dry oxygen and the p. If you know the quantity in gallons multiply by the number in Column B. É solúvel em água e etanol e pode ser preparado pela ação de ácidos minerais em sulfetos metálicos geralmente em laboratórios pela.
It smells like rotten eggs at low concentrations and causes you to quickly lose your sense of smell. Many areas where the gas is found have been identified but pockets of the gas can occur anywhere. In this article we will discuss the following things- Hydrogen sulfide H2S molecular geometry lewis structure electron geometry its bond angle polarity hybridization and dipole moment.
DOT ID Guide. Sebastian et al 1981. The empirical formula of the gas is CH2.
It is also dangerous for the environment. In its solid form ice water is less. A 515g sample of gas containing only carbon and hydrogen has a volume of 785 mL with a pressure of 1522 torr at a temperature of 28 degrees Celsius.
Weight - the Gravity Force. This occurs mostly in swamps and sewers and the process is known as anaerobic digestion. Hydrogen Sulfide Safety Data Sheet P-4611.
It is poisonous and flammable with a smell like rotten gas. How would you find the molecular formula for a compound with the empirical formula CH2O and the molar mass is 1802 gmol. Synonyms Trade Names Hydrosulfuric acid Sewer gas Sulfuretted hydrogen CAS No.
For example its melting point 0 C 32 F and boiling point 100 C 212 F are much higher than would be expected by comparison with analogous compounds such as hydrogen sulfide and ammonia. Hydrogen sulfide is a chemical compound with the formula H 2 S. It is commonly known as hydrosulfuric acid sewer gas and stink damp.
Hydrogen sulfide can also result from industrial activities such as food processing coke ovens kraft paper mills tanneries and petroleum refineriesHydrogen sulfide is a flammable colorless gas with a characteristic odor of rotten eggs. Gas Physical Properties Fast Facts provides physical properties such as molecular weight critical temperature and pressure liquid and gas density and specific gravity. The Gas Cost Estimator provides a budgetary cost estimate and sample system layout and sketch for bulk argon nitrogen or oxygen supply after you provide some basic information regarding your requirements location volume usage etc.
Li J Petelenz D Janata J 1993. Incompatible with most common metals amines metal oxides acetic anhydride propiolactone vinyl acetate mercuric sulphate calcium phosphide. It is related to acidity measurements in most cases by the equation pH log 12.
An explosion occurred upon heating 1-pentol and 1-pentol under hydrogen pressure. Find the name of the compressed gas you want to convert. Shipped as a liquefied compressed gas Skip.
It is poisonous corrosive and flammable. Hydrogen sulphide is most commonly formed due to the microbial breakdown of organic matter in the absence of oxygen. Colourless gas OU Chemical Safety Data No longer updated More details.
Hydrogen sulfide is often produced from the microbial breakdown of organic matter in the absence of oxygen such as in swamps and sewers. Mixtures of hydrogen carbon monoxide or methane and oxygen difluoride are exploded when a spark is discharged Mellor 2 Supp. Hydrogen Sulfide CAS No.
Ribose is an important sugar in RNA with a molar mass of 15015 gmol. Sulfuretted hydrogen sulfur hydride hydrosulfuric acid hepatic gas stink damp 12. Hydrogen - Thermophysical Properties - Chemical Physical and Thermal Properties of Hydrogen - H 2.
The equation of the state. Sulfeto de hidrogénio em nomenclatura antiga sulfureto de hidrogénio citado algumas vezes como gás sulfídrico ou sulfidreto é um gás com odor de ovos podres e carne em decomposição cadáver em solução aquosa chamado de ácido sulfídrico. However it is a highly corrosive flammable and poisonous gas.
The result is the quantity of compressed gas in cubic feet. CHEMICAL GAS NAME FORMULA COLUMN A COLUMN B. NIOSH REL C 10 ppm 15 mgm 3 10-minute OSHA PEL C 20 ppm 50.
If its empirical formula is CH2O what is its. Langmaier J Janata J 1992. Sensitive layer for electrochemical detection of hydrogen cyanide.
What is the molecular formula of the gas. 1 ppm 140 mgm 3. Oxygen is a strong oxidizing agent and.
H2S Other means of identification. If you know the quantity in pounds multiply by the number in Column A 3. It is a colorless chalcogen hydride gas with the characteristic foul odor of rotten eggs.
This is mainly carried out by sulfate-reducing. Suspended gate field-effect transistor sensitive to gaseous hydrogen cyanide. For the prediction of hydrogen solubility of oil fractions under high temperature and high-pressure system the equation of state method has good applicability.
Shipped as a liquefied compressed gas NIOSH MW4025000. At standard temperature and pressure STP two atoms of the element bind to form dioxygen a colorless odorless tasteless diatomic gas with the formula O 2. Torres et al 2013.
Explosive reactions occur upon ignition of mixtures of nitrogen trifluoride with good reducing agents such as ammonia hydrogen hydrogen sulfide or methane. Methane - Thermophysical Properties - Chemical Physical and Thermal Properties of. At standard conditions hydrogen is a gas of diatomic molecules having the formula H 2It is colorless odorless non-toxic and highly combustibleHydrogen is the most abundant chemical substance in the universe constituting roughly 75 of all normal matter.
Hydrogen - Specific Heat - Specific heat of Hydrogen Gas - H 2 - at temperatures ranging 175 - 6000 K. It is flammable over a wide range of vaporair concentrations. Hydrogen sulphide is a colourless chalcogen hydride gas with the formula H 2 S.
People can smell it at low levels. The vapors are lighter than air. Colorless to slightly yellow gas with a pungent irritating odor.
Hydrogen is the chemical element with the symbol H and atomic number 1. Details of the supplier of. This process is commonly known as anaerobic digestion which.
Determination of trace hydrogen cyanide in the presence of large concentrations of hydrogen sulfide. Although its formula H 2 O seems simple water exhibits very complex chemical and physical properties. Note that monthly volumes must be between 20000 and 3500000 SCF for nitrogen and oxygen and between 20000 and 1000000 SCF for argon.
Hydrogen solubility refers to the content of hydrogen in the liquid phase which is a typical gas-liquid equilibrium problem Chao and Seader 1961. Weight - Mass vs. Once ignited it burns with a pale blue almost invisible flame.
Hydrogen sulfide is a colorless gas with a molar mass of 3408 gmol 1.
Message via chat and well immediately start working on your assignment. Trio-Smart is also a breath test but it detects and measures all three of the SIBO gases hydrogen methane and hydrogen sulfide.
Hydrogen Peroxide Market size worth 211 Billion Globally by 2028 at 58 CAGR.

Hydrogen sulfide used in real life. Do you have an urgent order. Please Use Our Service If Youre. Hydrogen bonds are a critical part of many chemical processes and they help determine the properties of things necessary for life such as water and.
Along with our writing editing and proofreading skills we ensure you get real value for your money hence the reason we add these extra features to our homework help service at no extra cost. Sulfuric acid barium hydroxide --- barium sulfate and water. Hydrogen sulfide is the only chemical that my agency and others participating in this investigation have identified as being directly caused by this.
Pure water that contains an equal balance of positive hydrogen ions H and negative hydroxide ions OH - has a neutral pH. A 2019 video by Real Engineering noted that notwithstanding the introduction of vehicles that run on hydrogen using hydrogen as a fuel for cars does not help to reduce carbon emissions from transportation. However at least 03 ppm of dissolved oxygen is needed to carry out the oxidation.
At low concentrations humans recognize its characteristic smell of rotten eggs At higher concentrations the gas can be deadly. And for many that - plus the indisputable fact that molybdenum is a funny word - is often about as far as their knowledge goes of this silvery. Energy used by cells is chemical energy in the form of ATP or adenosine triphosphate.
Sensitive layer for electrochemical detection of hydrogen cyanide. And 42 as every Mendeleev fan knows is the atomic number of molybdenum. Every mouthful of food we eat contains a little mercury.
Tastyart Ltd Rob White Getty Images. Chlorination can be used to remove any level of hydrogen sulfide but it is most often applied in cases where the hydrogen sulfide concentration exceeds 60 mgL. On a spectrum in-between concentrations of hydrogen sulfide can cause headaches nausea eye irritation and dizziness.
We provide solutions to students. Iron is a very important element required by living organisms to carry out numerous metabolic reactions. Best of all we achieve a relentlessly reliable reading without depending on expensive consumables or short life pH sensors.
This direct measurement gives an accurate reading of total hydrogen sulfide bisulfide sulfide in the fluid regardless of the current equilibrium. Halogenated aliphatic compounds are compounds built from straight chains of carbon and hydrogen with varying numbers of hydrogen atoms replaced by halogen atoms. Silver nitrate sodium chloride --- silver chloride and sodium nitrate.
Almost anything you do breaks these bonds such as brushing hair roughly coloring pool water beach water thermal styling etc. Langmaier J Janata J 1992. Iron-oxidizing bacteria are chemotrophic bacteria that derive energy by oxidizing dissolved ferrous ironThey are known to grow and proliferate in waters containing iron concentrations as low as 01 mgL.
The reason for measuring H2S and its dissociated forms is of course to manage how you remove it. Iron sulfide hydrogen chloride --- iron chloride and hydrogen sulfide poisonous gas lead nitrate potassium iodide --- lead iodide and potassium nitrate saltpeter sodium bicarbonate baking soda vinegar --- carbonic acid and sodium acetate. Suspended gate field-effect transistor sensitive to gaseous hydrogen cyanide.
The soft black mineral molybdenite molybdenum sulfide. Mercuric sulfide vermilion is a high-grade bright-red paint pigment but is very toxic so is now only used with great care. C 6 H 12 O 6 6O 2 6CO 2 6H 2 O energy 36 ATPs Anaerobic Respiration.
Hydrogen Sulfide Scavengers Market By Process Regenerative Non-Regenerative By Type Water-soluble Oil-soluble Metal-based By Application. Hydrogen sulfide is a manure gas that can be dangerous. During a cruise to the OMZ off Peru in January 2009 we found a sulfidic plume in continental shelf waters covering an area 5500 km2 which contained 226104 tons of H2S.
Determination of trace hydrogen cyanide in the presence of large concentrations of hydrogen sulfide. Mercury has no known biological role but is present in every living thing and widespread in the environment. A small chemical feed pump is used to feed the chlorine solution usually sodium hypochlorite into the water upstream from a mixing tank or coil of plastic pipe.
Real-Life Lego Wind Turbines Coming Soon To A Wind Farm Near You. Wishing for a unique insight into a subject matter for your subsequent individual research. Organisms That Cannot Make Their Own Food.
Li J Petelenz D Janata J 1993. Some highly chlorinated representatives of. The 95 of hydrogen still produced from fossil fuels releases carbon dioxide and producing hydrogen from water is an energy-consuming process.
Anaerobic respiration is a set of chemical reactions that allows cells to gain energy from complex molecules without oxygen. These types of organisms transform inorganic molecules such as hydrogen sulfide methane or ammonia into organic molecules that they can use for food. PH is a measure of the hydrogen ion activity in a solution.
Solutions with a higher concentration of hydrogen ions H than pure water are acidic and have a pH less than 7. OMZs can sporadically accumulate hydrogen sulfide H2S which is toxic to most multicellular organisms and has been implicated in massive fish kills. When these are broken they become a single sulfide bond and when.
Think of your hair strand like a ladderthe DNA strand of hair has these hydrogen disulfide bonds that are connected in pairs like ladder steps says celebrity colorist Bianca Hillier. When hydrogen is used. A much lower amount about 10 parts per billion is enough to cause adverse health effects said Jill Johnston an assistant professor of environmental health at USC.
Fermentation is a specific type of anaerobic respiration that is used by yeast and some bacteria. The mixing tank or pipe should be sized to provide at least 20. Get your paper done in less than 4 hours.
Hydrogen sulfide is a toxin and there is little. Hydrogen sulfide is produced anaerobically by bacteria breaking down sulfur. Hydrogen sulfide levels as high as 1000 parts per billion were recorded near the Dominguez Channel earlier this month by the South Coast Air Quality Management District.
The other VSC is dimethyl sulfide which mainly responsible for extra-oral or blood-borne halitosis but it can be a contributor to oral malodorKetones such as acetone benzophenone and acetophenone are present in both alveolar lung and mouth air. Indole and dimethyl selenide are present in alveolar air1621 These compounds are also factors for Halitosis occurrence and it may be simply. Organisms that cannot make their own food are called heterotrophs.
Solutions with a higher concentration of hydroxide ions OH - than water are basic. Downsides of Lab Testing My clinical philosophy has increasingly been to take a cautious approach to diagnosing with functional medicine lab testing because often expensive tests fail to actually add much to the clinical picture or help make a patient. Halogenated aliphatics are effective solvents and degreasers and have been widely used in manufacturing and service industries ranging from automobile manufacturing to dry cleaning.
This was the first time that H2S was measured in the Peruvian OMZ and. Here is the overall equation for aerobic cellular respiration. Twiggy Forrest Sees Big Bright Green Hydrogen Potential.
Verified Market Research Some of the major factors expected to. Our daily intake is less than 001 milligrams about 03 grams in a lifetime and. The answer to the ultimate question - of life the Universe and Everything - is as every Douglas Adams fan knows 42.
