58 Monday March 26 2012 Rules and Regulations. Hydrochloric Acid 37 ww.
Sulfuric acid is a colorless oily liquid.
Hydrochloric acid melting. No data available Freezing point. Long term exposure to low concentrations or short term exposure to high concentrations can result in adverse health effects from inhalation. Hydrochloric Acid 6M Page 1 of 4 Hydrochloric Acid 6M Section 1 Product Description Product Name.
The hydrochloric acid present in the gastric acid helps in the proper digestion of. It is classified as a strong acidIt is a component of the gastric acid in the digestive systems of most animal species including humans. 7647-01-0 12 Relevant identified uses of the substance or mixture and uses advised against Identified uses.
Uses for Hydrochloric Acid A. Science education applications Synonyms. Hydrochloric Acid - Safety Data Sheet Hydrochloric Acid Product Code RC17021 Date of Issue 02082017 Version 10 Page 4 of 8 Conditions for safe storage including any incompatibilities.
Hydrochloric acid HCL is a clear colourless and pungent solution created by dissolving hydrogen chloride gas in water. Tennant investigated this residue and from it he eventually isolated osmium and iridium. Hydrochloric acid 32-38 solution ACC 11155 Section 1 - Chemical Product and Company Identification.
H ClIt is a colorless solution with a distinctive pungent smell. Hydrochloric acid is corrosive to the eyes skin and mucous membranes. They can be removed using vacuum filtration.
Hydrochloric Acid 31 36. There are four types of. Posted by 16 days ago.
Separation of bismuth from its oxide or carbonate ores can be effected by leaching with concentrated hydrochloric acid. Reacts with sulfides carbides borides and. 15780 Mean or Weighted MP VPmm Hg25 deg C.
Stomach acid is composed of hydrochloric acid HCl potassium chloride KCl and sodium chloride NaCl. Sitting and casually smoking after stabbing and spraying hydrochloric acid on passengers on a train in Tokyo and then setting. Chemsrc provides Hydrochloric acidCAS7647-01-0 MSDS density melting point boiling point structure formula molecular weight etc.
3695C 6683F 3968 K Block. Prevents Infections Hydrochloric acid acts as a barrier against foreign microorganisms and helps prevent infection. KOWWIN v167 estimate 054 Boiling Pt Melting Pt Vapor Pressure Estimations MPBPWIN v142.
The amount of stick togetherness is important in the interpretation of the various properties listed above. Hydrochloric acid concentration in the stomach is. Hydroxides amines alkalis copper brass zinc Note.
Reacts exothermically with organic bases amines amides and inorganic bases oxides and hydroxides of metals. We are a leading supplier to the global Life Science industry with solutions and services for research biotechnology development and production and pharmaceutical drug therapy development and production. Hydrochloric Acid 31 367.
Identification of the substancemixture and of the companyundertaking 11 Product identifiers Product name. It is corrosive to metals and tissue. Hydrochloric acid the aqueous solution of hydrogen chloride is also commonly given.
HYDROCHLORIC ACID CAS NO 7647-01-0 MATERIAL SAFETY DATA SHEET SDSMSDS SECTION 1. 1963C 3565F 2236 K Period 5 Boiling point. The physical properties of melting point boiling point vapor pressure evaporation viscosity surface tension and solubility are related to the strength of attractive forces between molecules.
The refinement process involves combining hydrochloric acid with metallurgical-grade silicon to turn it into what are called trichlorosilanes. Hydrochloric Acid 6M Recommended Use. In a chilled environment both compounds will not be soluble in water because the solubility of benzoic acid and 2-naphthol in water at 25C is 034g100mL and 0074g100mL respectively.
800-227-1150 8am-5pm ET M-F Chemtrec. The compound hydrogen chloride has the chemical formula H Cl and as such is a hydrogen halideAt room temperature it is a colourless gas which forms white fumes of hydrochloric acid upon contact with atmospheric water vaporHydrogen chloride gas and hydrochloric acid are important in technology and industry. 45619 Adapted Stein Brown method Melting Pt deg C.
Carolina Biological Supply Company 2700 York Road Burlington NC 27215 1-800-227-1150 Chemical Information. What is Hydrochloric Acid. ρ-sulfur is unstable eventually reverting to orthorhombic sulfur α-sulfur.
It is a by-product of chlorine manufacture along with sodium hypochlorite and sodium hydroxide. Store in a dry clean cool well ventilated place away from sunlight. Hydrochloric acid has many uses.
Hydrochloric acid 32-38 solution Catalog Numbers. Hydrochloric acid is the main component of the gastric juices produced in the stomach and it maintains the stomach pH of 1 to 2 by acidifying the stomach contents. HYDROCHLORIC ACID is an aqueous solution of hydrogen chloride an acidic gas.
Reacts exothermically with carbonates including limestone and building materials containing limestone and hydrogen carbonates to generate carbon dioxide. Hydrochloric acid also known as muriatic acid is an aqueous solution of hydrogen chloride chemical formula. Box 858 Ashtabula Ohio 44005 Phone.
118 20 degrees Celsius. Laboratorychemicals Industrial for professional use only. Acute short-term inhalation exposure may cause eye nose and respiratory tract irritation and.
It is used in the production of chlorides fertilizers and dyes in electroplating and in the photographic textile and rubber industries. Hydrochloric acid helps the body break down foods such as calcium digest and drink them. Density 15 lb gal.
2 2 2 2. Hydrochloric Acid 37 ww 7647-01-0 Hydrochloric Acid 7647-01-0. Hydrochloric Acid 37 ww.
The alleged attacker in Japan in a Joker costume. Hydrochloric acid will effectively replace the original proton benzoic acid and 2-naphthol lost. D Density g cm 3.
Metallic bismuth is used principally in alloys to many of which it imparts its own special properties of low melting point and expansion on solidification. It also eliminates stomach bacteria and viruses thereby protecting your body from infection. No data available Flash point.
The first step in the process was to dissolve ordinary platinum in aqua regia nitric acid hydrochloric acid. Sitting and casually smoking after stabbing and spraying hydrochloric acid on passengers on a train in Tokyo and then setting everything on fire. Store in the original labelled container and keep container tightly cl osed when.
Melting point degrees Celsius. After extraction and purification the percent recovery and. 416E-008 Modified Grain method Subcooled liquid VP.
Hydrochloric acid is highly corrosive to most metals. It is used to. -30 C Boiling point.
Articles of Hydrochloric acid are included as well. This on heating with lime and charcoal produces metallic bismuth. Helps in digestion of food.
In the rhombohedral allotrope designated ρ-sulfur the molecules are composed of rings of six sulfur atoms. Hydrochloric Acid 37 ww Safety Data Sheet according to Federal Register Vol. Hydrochloric acid is an important laboratory reagent and industrial chemical.
Dilution then precipitates the oxychloride BiOCl. It is soluble in water with release of heat. Not all of it went into solution and it left behind a black residue.
A142-212 A142P-19 A142P-20 A144-212 A144-212LC A144-500 A144-500LB A144-500LC A144-612GAL A144C-212 A144C-212EA A144P-19 A144P-20 A144S-212 A144S-212EA A144S-500 A144SI-212 A466-1 A466-2 A466-250. Acid etching steel pickling oil and gas ore and mineral food processing pharmaceutical organic chemical synthesis. Boiling Pt deg C.
This form is prepared by treating sodium thiosulfate with cold concentrated hydrochloric acid extracting the residue with toluene and evaporating the solution to give hexagonal crystals. These attractive forces are called Intermolecular Forces. It will char wood and most other organic matter on contact but is unlikely to cause a fire.
Enter Na 2 B 4 O 7 10H 2 O mass in the upper input. The weight of 1 liter of HCl is determined by multiplying the solution concentration Wt by its specific gravity SpGr and by 1000 assuming that 1 liter of water weighs 1000 grams.

Molar Mass Molecular Weight Of Hcl Hydrochloric Acid Youtube
3 Calculate the molar mass of a metal using experimental data.

Hydrochloric acid molar mass. As solvent for each of reagent 1 M HCl was diluted by analytical reagent-grade 36 38 hydrochloric acid with distilled water. A 135g sample of dilute hydrochloric acid is placed in a copper calorimeter with a mass of 465. 2 Calculate the molar volume of a gas at STP using experimental data.
The water vapor pressure at 210 C is 186 mm Hg and the atmospheric pressure is 755 mm Hg. How many grams of aluminum reacted. The precipitate was filtered dried and weighed to a constant mass of 243 g.
Molar Enthalpy of Neutralisation. The molarity of hydrochloric acid is determined by titrating it against the standard solution of sodium carbonate using methyl orange as indicator. Mass of HCl 1180 365100 4307 g.
H 2 SO 4 Uses Sulfuric Acid It is used in making fertilizers. Weight fraction ww data is used. Take a burette and wash it with water.
It is used to produce phosphoric. The gas was collected over water at 210 C. B What mass of hydrogen gas is produced.
The partial pressure of water at 27 C is 268 mmHg. Weak Acid Strong Base. And oxygen 159994 gmol.
Benzoic acid b ɛ n ˈ z oʊ. 2 A 00677 g sample of magnesium metal reacted with excess hydrochloric acid to produce 699 ml of hydrogen gas. The K a value for HCl is reported at 13 x 10 6 where sulfuric acid is 10 x 10 3.
3 HCI - AICI 32 H C. Set the graduated cylinder in your sink and fill it to the top with tap water. A mass of 500 g of aluminum metal is reacted with the acid.
Hydrochloric acid is corrosive to the eyes skin and mucous membranes. Mass of HCl solution 1180 g. ɪ k is a white or colorless solid with the formula C 6 H 5 CO 2 H.
Multiply the elements atomic mass by the number of atoms of that element in the. If 1081 g of N2H4 is used what mass of nitrogen is produced. The temperature of the acid and calorimeter is 117C.
Mass of HCl Mass of solution ww Mass of HCl 1180 365100. Hydrochloric acid has many uses. It is the simplest aromatic carboxylic acidThe name is derived from gum benzoin which was for a long time its only sourceBenzoic acid occurs naturally in many plants and serves as an intermediate in the biosynthesis of many secondary metabolites.
Hydrochloric acid 7647-01-0 151327-08-1. Wt HCl is first converted to molar concentration to determine the H. Hydrochloric acid concentration in the stomach is.
With the tube in. Hydrochloric acid helps the body break down foods such as calcium digest and drink them. Sulfuric Acid Structure H 2 SO 4.
Hydrochloric acid is a monoprotic molecule with an acid-dissociation equilibrium constant K a three orders of magnitude greater than sulfuric acid indicating HCl is both strong and effective as an acid. Of HCl one molecular mass per liter yields a one molar solution of H. It can be calculated by adding the invididual molar mass of every atom that are composing the molecule CH4.
The relative atomic masses of the elements in hydrochloric acid are. 1 Answer Nathan L. 2 106 g of magnesium react with excess hydrochloric acid.
Specific gravities for very dilute HCl solutions may be. A Write the balanced equation for the reaction. The tests were installed in a thermostatic water bath which contained 100 mL of various of concentration 010 mmol L 1 with -sparteine and -sparteine at 298 K for one day.
Calculate the molar enthalpy change for the reaction. As the next step find the mass of that solution 1 dm 3. Molecular Weight Molar Mass.
Strength of the acid is determined by multiplying its molarity with its molecular mass which is 365. The experiment described above is repeated using 500 mL of 10 mol L-1 sodium hydroxide a strong monobasic base and 10 mol L-1 hydrogren cyanide HCN a weak monoprotic acid K a 6 10-10 instead of 10 mol L-1 hydrochloric acid a strong monoprotic acid. Enter a chemical formula.
HCl is a very strong acid that is corrosive and hazardous. It is used in petroleum refining. Incline tilt the gas-measuring tube slightly from an upright position and carefully pour in about 10 mL of 6 M hydrochloric acid ___ 5.
Yellow to pink Acid in burette. Chemistry Stoichiometry Equation Stoichiometry. Atomic Mass of Atoms.
Acute short-term inhalation exposure may cause eye nose and respiratory tract irritation and. Now mass of HCl in 1 dm 3 is found. Excess ammonium oxalate solution NH 4 2 C 2 O 4aq was added to the solution to precipitate the calcium ions as calcium oxalate CaC 2 O 4s.
It is used in chemical manufacturing industries. The mass of hydrochloric acid you get in that sample will thus be 1200colorredcancelcolorblackg solution 400 g HCl100colorredcancelcolorblackg solution 480 g HCl Use hydrochloric acids molar mass to determine how many moles you have in that many grams 480colorredcancelcolorblackg 1 mole. Next amount of HCl is determined by dividing HCl amount from molar.
The molar mass of a substance also often called molecular mass or molecular weight although the definitions are not strictly identical but it is only sensitive in very defined areas is the weight of a defined amount of molecules of the substance a mole and is expressed in gmol. These are perfecly equivalent. Hydrogen 1007 gmol and chlorine 35453 gmol.
100794 35453 Percent composition by element. Aluminum reacts with excess hydrochloric acid to form aqueous aluminum chloride and 465 mL of hydrogen gas over water at 27 degrees Celsius and 751 mmHg. Jul 8 2017 00324 g Al Explanation.
C Classify this reaction as single or double displacement decomposition synthesis or. 2764 Calculate the molecular weight of a chemical compound. Perchloric acid is a mineral acid with the formula H Cl O 4Usually found as an aqueous solution this colorless compound is a stronger acid than sulfuric acid nitric acid and hydrochloric acidIt is a powerful oxidizer when hot but aqueous solutions up to approximately 70 by weight at room temperature are generally safe only showing strong acid features and no oxidizing properties.
Calculate the molar mass of each element in the compound. This compound is also known as Hydrochloric Acid. The levels of water inside and outside the gas collecting tube are identical.
Convert grams HCl to moles or moles HCl to grams. It is used in the production of chlorides fertilizers and dyes in electroplating and in the photographic textile and rubber industries. A 200 g sample of limestone was dissolved in hydrochloric acid and all the calcium present in the sample was converted to Ca 2 aq.
Salts of benzoic acid are used as food. Note that to be consistent with the use of decahydrate molar mass and to be able to balance water reaction equation is not the one shown above but. Introduction Metals that are above hydrogen in the activity series will displace hydrogen from an acid and produce hydrogen gas.
Na 2 B 4 O 7 10H 2 O 2HCl 4H 3 BO 3 2NaCl 5H 2 O. Molar Mass of hydrochloric acid HCl Molar Mass of phosphoric acid H3PO4 Molar Mass of ammonium chloride NH4Cl Molar Mass of baking powder NaHCO3 Molar Mass of Sucrose C12H22O11 Molar Mass of Isopropyl Alcohol C3H6CHOH Molar Mass of Carbon Monoxide CO Molar Mass of antimony chloride - 3 SbCl3 Molar Mass of Chloroplatinic acid H2PtCl6 Molar Mass. It also eliminates stomach bacteria and viruses thereby protecting your body from infection.
Mass of 1000 cm of Mg ribbon On board in laboratory _____ ___ 3. The relative atomic masses of the elements in glucose are. To calculate hydrochloric acid solution concentration use EBAS - stoichiometry calculator.
It is used in the production of steel and iron. We will use this to determine the molar volume of hydrogen gas at STP. Stomach acid is composed of hydrochloric acid HCl potassium chloride KCl and sodium chloride NaCl.
After the reaction has ceased. Structure properties spectra suppliers and links for.
Homeopathic Medicines for Acid Reflux Heartburn and GERD. Electro-chemical series also known as electro-activity series Calcium therefore has a greater affinity to bond with electron receptors than hydrogen does and hence it has the ability to replace hydrogen in most of.

Hydrochloric Acid In The Stomach And Digestive Problems Owlcation
By the way stable Hypochlorous salts can be gotten by dissolving chlorine gas into sodium hydroxide mixture or other.
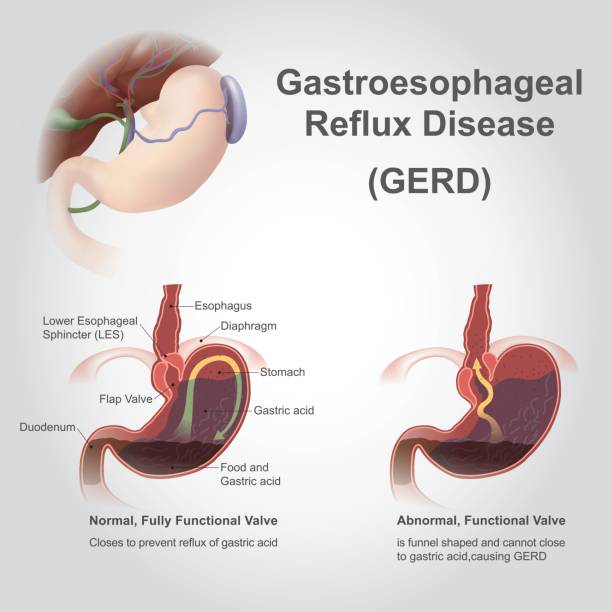
What is bad about hydrochloric acid gas. Hydrochloric and hydrofluoric acid both very caustic substances are used to refine and clean the wafers of silicon which is the main building block of most solar panels. Mg 2 HCl H₂ MgCl₂. The amino acid liquid is then neutralized pressed through a filter mixed with active carbon and purified through filtration.
Decant or siphon into another container neutralize with 6 molar hydrochloric acid before. For example a 2012 study found that drinking naturally carbonated artesian-well alkaline water with a pH of 88 may help deactivate pepsin the main enzyme that causes acid reflux. Zinc Hydrochloric Acid Zinc Chloride Hydrogen Zn s 2HCl l ZnCl 2 l H 2 g Materials Needed.
Notably hydrochloric acid bactericides scale removers and friction-reducing agents. When these acid levels increase it can create an environment for hyperacidity. Stomach acid is crucial to the digestion of food and kills harmful bacteria.
Only maybe a dozen chemicals are used for any given well but the choice of which chemicals is well-specific depending on the geochemistry and needs of that well. Muriatic acid is a form of hydrochloric acid which has a pH of about 1 to 2. The stomach produces the hormone gastrin which in turn creates hydrochloric acid.
The reaction between zinc and hydrochloric acid is Z n 2 H C l Z n C l 2 H 2 This reaction is known as single replacement reaction where zinc metal displaces the hydrogen to form hydrogen gas and zinc chloride a salt. Wear a NIOSH-approved half face respirator equipped with an organic vaporacid gas cartridge specific for organic vapors HCl acid gas and SO2 with a dustmist filter. Bad breath Feeling of Abdominal bloating Cough Laryngitis Worsening of asthmatic complaint.
For example mixing bleach with toilet bowl cleaner which contains hydrochloric acid yields chlorine gas. When bloating occurs as the result it could put a lot of pressure on the muscle between the stomach and esophagus causing acid reflux to occur. Soybeans are boiled with hydrochloric acid for 15 to 20 hours.
For example if you put magnesium into hydrochloric acid youll get the following single-displacement reaction. Other bromine compounds of significance include hydrogen bromide HBr a colourless gas used as a reducing agent and a catalyst in organic reactions. Digestive System vocabulary game to match the names with the picture.
After most of the amino acid is removed the mixture is cooled to stop the hydrolytic reaction. You can get hydrogen gas by reacting hydrochloric acid with zinc. Answer 1 of 6.
R e a s o n. This is what makes it so good at bleaching and disinfecting. Calcium is a metal that is higher than hydrogen in the ecs.
The H 2 generation phase is relatively short because it is complete. Be very careful to avoid contact with the acid. Because hydrogen is a gas bubbles can be seen during this reaction.
This is one of the main ways coffee causes gas as undigested putrefying protein is feed on by the intestinal bacteria that create hydrogen sulfide rotten egg gas for those really smelly farts. NTP 1992 National Toxicology Program Institute of Environmental Health Sciences National Institutes of. Hypochlorous acid is a strong oxidizer.
At this point your pH levels decrease and problems ranging from mild to severe can develop. When there is a shortage of hydrochloric acid for proper digestion you often have problems breaking down protein in particular. Its quite an experience hearing the sound of your voice carrying out to a over 100 first year.
Benzalkonium Chloride USPNF Grade 50 Ethanol 190 Proof 95 Certified Organic Sugar Cane Alcohol FCC Grade Ethanol 200 Proof 100 Denatured Alcohol with Heptane Extraction Grade Ethanol 200 Proof 100 Non-Denatured Alcohol ACSUSPFCC Grade Kosher Hydrogen Peroxide. The above response does not seems in the balance and it is difficult to detach Hypochlorous Acid HOCL from this blend. The conventional treatment for GERD involves the use of antacids which work by changing the pH of the stomach acid to make it less acidic.
Natural gas emits 50 to 60 percent less carbon dioxide CO2. Cl2 H2O Hypochlorous Acid HOCL HCl. Preparations such as Gaviscon or Algicon combine alginate with antacids and are.
If you mix bleach with an acid chlorine gas will be produced. When food is digested slowly the food sits idly in the stomach creating a lot of gas in the stomach. A solution of the gas in water is called hydrobromic acid a strong acid that resembles hydrochloric acid in its.
Many metals will boil when you place them into a strong acid. Ordinary household bleach contains sodium hypochlorite which reacts with ethanol or isopropyl alcohol to produce chloroform hydrochloric acid and other compounds such as chloroacetone or. It gives us the Hypochlorous Acid HOCL with HCL hydrochloric acid as an additional chemical in the process.
Gas could cause indigestion and acid reflux or it could be the result of an acid reflux episode. Figure shows the curve obtained when the volume of hydrogen gas liberated against time is plotted. Long-chain volatile organic acids VOAs are converted to acetic acid C 2 H 4 O 2 CO 2 and hydrogen gas H 2.
Copy the graph and sketch on the same axes the curve that you would expect to. To investigate how the rate of reaction between Sodium Thiosulphate and Hydrochloric acid is affected by changing the concentration. High concentrations of VFAs increase both the biochemical oxygen demand BOD and VOA concentrations which initiates H 2 production by fermentative bacteria which stimulates the growth of H 2-oxidizing bacteria.
When ingested this tasteless and apparently harmless substance floats on gastric fluid to prevent the reflux of acid and pepsin into the esophagus. Let stand for 24 hr. Hydrochloric acid muriatic acid zinc granules or iron filings or strips of aluminum Hydrogen gas bubbles will be released as soon as the acid and zinc are mixed.
The refining process also produces silicon tetrachloride a toxic compound. It is not an antacid. The experiment is repeated using 25 cm 3 of 05 mol dm 3 hydrochloric acid at 800C to replace 50 cm 3 of 05 mol dm 3 hydrochloric acid at room temperature.
Large quantities tens of thousands of gallons for each well of the. This helps decrease the irritation caused by the acid to the stomach esophagus or. When Sodium Thiosulphate reacts with hydrochloric acid sulphur is produced.
Another way coffee makes you gassy and bloated is by stimulating gastric. The only differences between hydrochloric acid and muriatic acid are puritymuriatic acid is diluted to somewhere between 145 and 29 percent and often contains impurities like iron. Alginic acid Prepared from kelp seaweed alginate acts as a physical acid barrier for the esophagus in gastroesophageal reflux.
The sulphur forms in very small particles and causes the solution to cloud over and turn a yellow colour.