Acidulants such as Citric Acid Fumaric Acid Malic Acid Tartaric Acid etc can be added to lower the pH level to make the mold inhibitors more effective Sorbates PH. Assisted grinding with an equimolar amt.

Pdf Solubility Of Citric Malonic And Malic Acids In Different Solvents From 303 2 To 333 2 K
H 2 C 4 H 4 O 5.

Malic acid solubility. Jia Centers for Disease Control and. All of these acids are by-products of the metabolism of pyruvic acid. Unstable as seen during the stability testing.
Tartaric acid levels decrease with increasing ethanol concentrations due to the decrease in the solubility of its salt potassium bitartrate also known as cream of tartar. If more acid needs to be removed it is better to use the double-salt deacidification with calcium carbonate. PSMs promote plant growth via generating phytohormones such as auxins gibberellins cytokinins or polyamides 1 25 30 40Organic acids such as carboxylic glycolic malonic succinic fumaric and alpha-ketoglutaric acid that hasten the maturity and thereby enhance the ratio of straw as well as the total yield have also been recognized among phosphate solubilizers 6 9 31.
It was observed that the itraconazole L-malic acid cocrystal exhibited a similar dissolution profile to that of the marketed formulation 68. The first three acidulants are available both in powder and granular form while the rest two are liquid. Now you may have a good knowledge of the food additive citric acid E 330.
DL-tartaric acid is produced from the enzyme. For example malic acid has been shown to leach out divalent metal ions but requires an additional reducing agent to accelerate the process for dissolving the. Malic acid is an organic compound with the molecular formula C 4 H 6 O 5It is a dicarboxylic acid that is made by all living organisms contributes to the sour taste of fruits and is used as a food additiveMalic acid has two stereoisomeric forms L- and D-enantiomers though only the L-isomer exists naturallyThe salts and esters of malic acid are known as malates.
When we applied these proteins to our ELISA diagnostic assay they all keep showing highly specific reactivity and very low background. ALA was isolated by Reed in 1951 1 as an acetate replacing factor and its first clinical use dates from 1959 in the treatment of acute poisoning by Amanita phalloides also known death cap from mushrooms 2. Malolactic conversion also known as malolactic fermentation or MLF is a process in winemaking in which tart-tasting malic acid naturally present in grape must is converted to softer-tasting lactic acidMalolactic fermentation is most often performed as a secondary fermentation shortly after the end of the primary fermentation but can sometimes run concurrently with it.
Table 42 Various Acids Found in Food and Beverages. In another study by Childs et al fluoxetine HCl succinic acid cocrystal was found to exhibit an approximately twofold increase in aqueous solubility after only 5 min. Cocrystal of PZQ with MA was chem.
We are very satisfied with the qualities of these proteins. Has a high solubility in water 592 gL 20 C. Müller-Thurgau 1891 and Koch 1900 later attributed the presence of lactic acid bacteria to a reduction in the acidity of wines and shortly afterwards in 1901 Seifert reported that these bacteria were capable of degrading malic acid.
It has a role as a food acidity regulator and a fundamental metabolite. The American Dairy Science Association ADSA is an international organization of educators scientists and industry representatives who are committed to advancing the dairy industry and keenly aware of the vital role the dairy sciences play in fulfilling the economic nutritive and health requirements of the worlds population. Malonic acid is used for the introduction of an acetic acid moiety under mild conditions by Knoevenagel condensation and subsequent decarboxylation.
Biomatik started the protein production process from codon optimization and gene synthesis the solubility of proteins we got were significantly improved. Thus this technique is. Of citric acid CA malic acid MA salicylic acid SA and tartaric acid TA gained in cocrystal formation which all showed pH-dependent soly.
The name malic acid comes from the apples botanical genus name malus while lactic acid HC 3 H 5 O 3 is found in wine and sour milk products such as yogurt and some cottage cheeses. However the most sol. In general solubilityavailability of heavy metals for plant uptake and suitability of a site for phytoextraction are additional factors that should be considered in addition to suitability of plants before using phytoextraction for soil remediation.
α-lipoic acid ALA also known as 12-dithiolane-3-pentanoic acid or thioctic acid is a compound commonly found in mitochondria necessary for different enzymatic functions. For example when NaClaq. Solubility g100 mL H2O Formic acid HCO2H 101 8 Infinite Acetic acid CH3CO2H 118 17 Infinite Propionic acid CH3CH2CO2H 141 -21 Infinite Butyric acid CH3 CH22CO2H 164 -5 Infinite Valeric acid CH 3CH23CO2H 186 -34 5 Caproic acid CH3CH24CO2H 205 -3 1 Caprylic acid CH 3CH26CO2H 239 17 Insoluble Capric acid CH3CH28CO2H 270 32 Insoluble Lauric acid.
It provides leadership in scientific and technical support to. It is important to note that the double-salt technique favors the removal of tartaric acid rather than malic acid unless the initial concentrations of malic acid are double the concentration of tartaric acid. Examples include the preparation of cinnamic acid used for the production of the anti-inflammatory cinmetacin and 345-trimethoxycinnamic acid the key intermediate of the vasodilators cinepazet and cinepazide.
It is a 2-hydroxydicarboxylic acid and a C4-dicarboxylic acid. By neat grinding with amorphous. Equimolar cyclodextrin complexes prepd.
Grapes also contain acids mostly tartaric acid and malic acid but levels decrease during vinification. Storage temperature is key to ensuring the. More recent studies particularly from the 1970s onwards confirmed the importance of malolactic fermentation in reducing acidity essential in red wines.
Formamide 1 formic acid 2 urea 3 diaminomaleonitrile 4 glycine 5 alanine 6 valine 7 leucine 8 proline 9 serine 10 asparagine 11 aspartic acid 12 glutamic acid 13 lysine 14 histidine. Organic chelates such as citric acid and malic acid can also be used to improve phytoextraction of heavy metals from polluted soils. Solubility rules are very useful in determining which ionic compounds are dissolved and which are not.
DL-malic acid acetic acid and phosphoric acid are derived from chemical synthesis. Malic acid is a 2-hydroxydicarboxylic acid that is succinic acid in which one of the hydrogens attached to a carbon is replaced by a hydroxy group. Sorbates are the more effective preservative against a wider spectrum of food spoilage microorganisms than benzoates or propionates When used at common pH levels of mildly acidic foods pH 55 60.
Water - Density Specific Weight and Thermal Expansion Coefficients - Definitions online calculator and figures and tables with water. Stability and Reactivity Data Stability.

Hot Paraffin Wax Water Fiery Reaction Youtube
Ethylene glycol is a synthetic liquid substance that absorbs water.

Paraffin solubility in water. Standard Methods For the Examination of Water and Wastewater 23nd edition 2017. Dehydration of ethanol to form ethene. Ethylene glycol is used to make antifreeze and de-icing solutions for cars airplanes and boats.
Water usually boils at 100C under normal atmospheric pressure 760 mm of Hg. Toluene - Density and Specific Weight vs. Waxes made from paraffin or vegetable oils can be applied to make a water- or grease-resistant container for food products.
Standard Methods For the Examination of Water and Wastewater. CDC-ATSDR Toxic Substances Portal. In association with Nuffield Foundation.
Kolliphor HS 15 is a yellowish white paste at room temperature that becomes a liquid at approximately 30 C. N-octanol water No Data Available Autoignition temperature No Data Available Decomposition temperature No Data Available Viscosity 1400 centipoise - 4500 centipoise DetailsFor Liquid. Reacts with copper and precious metals.
Crop oil is a misnomer because the material actually is from petroleum paraffin or naphtha base not vegetable derivative a phytobland nonphytotoxic nonaromatic oil of 70 to 110 second viscosity water 1 and 30 w motor oil 300. It is insoluble in liquid paraffin. 32 Full PDFs related to this paper.
Natural vapour pressure at different temperatures. Paraffin wax is a white or colorless soft solid wax that is composed of a complex mixture of hydrocarbon derivatives with the following general properties. Similarly the high pressure also facilitates the rapid penetration of heat into deeper parts of the material and moisture present in the steam causes the coagulation of proteins causing an irreversible loss of function and activity.
Brightly colored inks are also applied to create bold graphic designs for self-supporting displays featuring product name information and company name and logo. Teams of salespeople and designers work together to create the manufacturing and printing patterns called. Non-corrosive in presence of glass.
Unlike paraffin or other alkanes which tend to puddle on the waters surface these fatty acids spread evenly over an extended water surface eventually forming a monomolecular layer in which the polar carboxyl groups are hydrogen bonded at the water interface and the hydrocarbon chains are aligned together away from the water. The UK kerosene is also known as paraffin and home heating oil. Paraffin wax is mostly found as a white odorless tasteless waxy solid with a typical melting point between about 46 and 68 C 115 and 154 F and a density of around 900 kgm 3.
Citation neededBronopol was invented by The Boots Company PLC in the early 1960s and first applications were as a preservative for pharmaceuticals. It dissolves in water ethanol and 2-propanol to form clear solutions. A short summary of this paper.
I nonreactive ii nontoxic iii water barrier and iv colorless. Standard Methods For the Examination of Water and Wastewater 23nd edition. Includes kit list and safety instructions.
Approximately 7½ millions tons of kerosene was used in the UK in 2005. Kerosene is a major component 60 of aviation jet fuels is used for oil central heating systems and can be used as a cleaning agent or solvent. The product is stable.
26 cc100 ml at 17C very slightly soluble Method of evolution as fumigant. From preparations of aluminium and magnesium phosphide. Special Remarks on.
Paraffin wax is often classed as a stable chemical since it is unaffected by most common chemical reagents but burns readily. It is odorless but has a sweet taste. Specific Gravity 092 - 098 Ref StdWATER1 Solubility in Water Complete Solubility- non-water No Data Available Partition coefficient.
Low molecular weight polyethylene glycols and propylene glycol in combination with water to dissolve the drug. Cyclodextrins and its derivatives to form water-soluble inclusion complexes of the drug or iii hydrophilic excipients eg. Paraffin wax is mostly found as a white odorless tasteless waxy solid with a typical melting point between about 46 and 68C 115 and 154F and a density of approximately 900 is insoluble in water but soluble in ether benzene and certain esters.
Teemperature and Pressure - Density and specific weight of liquid toluene. The first reported synthesis of bronopol was in 1897. It is also used in hydraulic brake fluids and inks used in stamp pads ballpoint pens and print shops.
It is insoluble in water but soluble in ether benzene and certain estersParaffin is unaffected by most common chemical reagents but burns readily. Its heat of combustion is 42 MJkg. Its solubility in water decreases with increasing temperatures.
Reactive with oxidizing agents. Paraffin water and many more. Dosages and concentrations of gas in air.
We would like to show you a description here but the site wont allow us. Solubility of Gases in Water - Solubility of Ammonia Argon Carbon Dioxide Carbon Monoxide Chlorine. With a focus on Bohrs work the developments explored in this module were based on the advancements of many scientists over time and laid the groundwork for future scientists.
This behavior is illustrated in the diagram on the right. This website uses cookies to help provide you with the best possible online experience. Ii complexing agents eg.
Current strategies employed to improve the apparent solubility of a drug include the use of. Ethylene glycol is a 12-glycol compound produced. Use this microscale experiment to illustrate hydrocarbon cracking using paraffin bromine water and aluminium oxide.
Excess heat Incompatibility with various substances. Full PDF Package Download Full PDF Package. However the boiling point of water increases if the pressure is to be increased.
Kerosene is produced on an. Chemical name 2-bromo-2-nitro-13-propanediol is an organic compound that is used as an antimicrobialIt is a white solid although commercial samples appear yellow. Includes kit list and safety instructions.
Find pharmaceutical suppliers and send inquiries. Insoluble in cold water hot water. Please read our Terms Conditions and Privacy Policy for information about.
Paraffin wax is characterized by a clearly defined crystal structure and has the tendency to be hard and brittle with a melting point typically in the range 5070. Conditions of Instability. Special Remarks on Reactivity.
Crop oils are believed to promote the penetration of pesticide spray through. 0C 32F 216 atmos 20C 68F 342 atmos 40C 104F 519 atmos. Use this class practical or demonstration to produce ethene gas as an example of an unsaturated hydrocarbon.
Crop oils are 95 to 98 percent oil with 1 to 2 percent surfactantemulsifier. The 20th century brought a major shift in our understanding of the atom from the planetary model that Ernest Rutherford proposed to Niels Bohrs application of quantum theory and waves to the behavior of electrons. The word kerosene comes from the Greek word keros meaning wax.
In ethanolwater mixtures the solubilities are somewhere between the end values for pure ethanol and pure water. Chemical modification of drug By modifying the drug chemically poorly soluble drugs can become more soluble.

Solubility Of Theophylline At Different Ph Values Download Table
Relaxes smooth muscles of the bronchi and is used to treat asthma.
Solubility of theophylline. Moreover PVP increases Iodines solubility. It has a role as a vasodilator agent a bronchodilator agent a muscle relaxant an EC 314 phosphoric diester hydrolase inhibitor an anti-asthmatic drug an anti-inflammatory agent an immunomodulator an adenosine. A solution of sodium benzoate makes benzoic acid more solubilized.
Theophylline is a dimethylxanthine having the two methyl groups located at positions 1 and 3. Weigh up the risks versus benefits of treatment because acyclovir the active form of valacyclovir crosses the placenta into the unborn child. Theophylline levels should be monitored in patients starting or increasing allopurinol therapy.
The pH of the reconstituted solution is 24 to 41. Biorelevant dissolution test methods for modified release dosage forms. Each of these metabolites is further metabolized and then excreted in the urine.
If considered medically necessary give sofosbuvirvelpatasvir with food 4 hr before omeprazole 20 mg. It has a role as an anticonvulsant an EC 35198 histone deacetylase inhibitor a mitogen a glutamate transporter activator an antimanic drug an analgesic a non-narcotic analgesic an environmental contaminant a xenobiotic a drug. The solubility of the acid and its salts in ethanol is very low or negligible.
Generally speaking the pH of extracellular fluid is always going to be within some decimal fractions of 74 and so drugs with a pKa under 7 ie. Drugs with large volumes of distribution usually due to lipid solubility and low plasma protein binding are poorly diliazable. Esomeprazole will decrease the level or effect of.
The solubility profile should be measured using a suitable and validated method. Fordtran JS Locklear. Seeing as many drugs are either weak acids or weak bases they will either be charged or uncharged in solutions with different pH.
The therapeutic dose of theophylline however is many times greater than the levels attained from caffeine metabolism. Carbamazepine is a dibenzoazepine that is 5H-dibenzobfazepine carrying a carbamoyl substituent at the azepine nitrogen used as an anticonvulsant. Velpatasvir solubility decreases as gastric pH increases practically insoluble at pH 5.
A patient was admitted to the hospital because of an acute episode of bronchial asthma. Acyclovir also passes into breastmilk. An in vitro model for exploring CR theophyllinemilk fat interactions.
The theophylline steady-state concentration achieved is 15 mgL k is 01 hr-1 calculate the volume of distribution. Examples - In water betamethasone alcohol dissolves at a. Drug concentration in vivo is a function of multiple factors Common TDM drugs Carbamazepine digoxin gentamycin.
Adequate hydration must be maintained otherwise acyclovir can precipitate in the renal tubules if its solubility is exceeded in intratubular fluid. The solubility of caffeine is increased by sodium benzoate. The goal of treatment during an acute gout attack is suppression of inflammation and control of pain.
Purines and pyrimidines are important molecules in organic chemistry and biochemistry because they are the basis for other molecules eg caffeine theobromine theophylline thiamine and because they are key components of the nucleic acids dexoyribonucleic acid. Inhibition of the metabolism of theophylline has been reported. The mechanism of the interaction may be explained by xanthine oxidase being involved in the biotransformation of theophylline in man.
Theophylline was administered to the patient as an iv loading dose of 300 mg followed immediately by a constant rate iv infusion of 45 mghr. It is structurally similar to caffeine and is found in green and black tea. Theophylline is more easily dissolved by sodium acetate.
Greater degrees of dialysis can be achieved with faster dialysate flow rates if the dialysate drug concentrations is low. 137-Trimethyluric acid is a minor caffeine metabolite. It is important to note that if a patient is not on uric acid lowering therapy at the time of an acute attack then this is not the time to initiate such therapyHowever if a patient is on uric acid lowering therapy at the time of an acute attack it should not be.
Weak acids will usually be. Mycophenolate mofetil hydrochloride has a solubility of 658 mgmL in 5 Dextrose Injection USP D5W. Any drug to be absorbed must be present in the form of solution at the site of absorption.
Use with other PPIs has not been studied. Solubility of urate salts grams of water per gram of compound Compound Cold water Boiling water Uric acid 15000 2000 Ammonium hydrogen urate 1600 Lithium hydrogen urate 370 39 Sodium hydrogen. Therapeutic drug monitoring Clinical pharmacology The regular measurement of serum levels of drugs requiring close titration of doses in order to ensure that there are sufficient levels in the blood to be therapeutically effective while avoiding potentially toxic excess.
Although plasma clearance may be beneficial increasing plasma clearance will dcrease dialysis clearance. In general seniors or children people with certain. Solubility is a major challenge for formulation scientist.
CellCept is available for oral administration as capsules containing 250 mg of mycophenolate mofetil tablets containing 500 mg of mycophenolate mofetil and as a powder for oral suspension which when constituted contains 200 mgmL. Various techniques are used for the enhancement of the solubility of poorly soluble drugs which include physical and chemical modifications of drug and other methods like particle size reduction crystal engineering salt formation solid. This diagram depicts the effect of a change in pH on the lipid solubility of a weak acid.
Coadministration of sofosbuvirvelpatasvir with omeprazole or other PPIs is not recommended.
Generic Name Ubidecarenone DrugBank Accession Number DB09270 Background. This oxidant is useful for the dehydrogenation of alcohols phenols and steroid ketones in organic chemistry.
Benzoquinone Sciencemadness Wiki
Benzoquinone Benzoyl group Benzoyl peroxide.

Solubility of benzoquinone in water. Water Water is a chemical substance with the chemical formula H2O. It is a metabolite of benzeneIt has a role as a cofactor a human xenobiotic metabolite and a mouse metabolite. A dissolved oxygen sensor can be used in the lab or in the field.
Oxidation of a phenol can produce 12-Benzoquinone 2019 Lets Talk Science. Only use fine adjustive w high objectives. PMS is an unsymmetrical oxidant with redox potential of 182 V based on Eq.
Major sources of oxygen in an operating system include poor deaerator operation in-leakage of air on the suction. Since the maximum concentration is limited by solubility to 200-300 mgliter at the ambient temperatures that are expected for drinking water some physical complications would accompany the introduction of this method into the large waterworks system in view of the large saturation beds required. Parcaentered - stays centered after changing.
A pigment is a finely divided solid which is essentially insoluble in its application medium. 280 mL deionized water was poured into a 4-neck round-bottom flask and heated to 80 C. At 60F and atmospheric pressure the solubility of oxygen in water is approximately 8 ppm.
A water molecule contains one oxygen and two hydrogen atoms connected by covalent bonds. Most TOC analyzers are based on the oxidation of organics to carbon dioxide and water with subsequent quantitation of the carbon dioxide. Ubidecarenone also called coenzyme Q10 is a 14-benzoquinone.
Although PMS is thermodynamically a strong oxidant its direct reaction with the majority of the. Water also exists in a liquid crystal state near. 14-benzoquinone is the simplest member of the class of 14-benzoquinones obtained by the formal oxidation of hydroquinone to the corresponding diketone.
National Toxicology Program Chemical Repository Database. The possibility of H-bond formation also influences solubility and in the case of NH 4 I an X-ray single-crystal analysis of the monosolvate shows the presence of an H-bonded cation N 2 H 7 with an NH N distance of 269 5 pm. Solubility of ozone in aqueous solutions of 0-06 M ionic strength at 5-30degreeC.
The solubility parameter of coal is 25 MPa 12. Anthraquinone the simplest of the anthraquinones is based on anthracene. Solubility in water 250 gL at 20 C based on OxoneFormula.
Styrene 30 mL was washed four times with 100 mL of 01 M NaOH in a separatory funnel and then four times with 100 mL deionized water. 68 Some typical solubilities at 25C expressed as g per 100 g solvent are. 1293 at 70 F NTP 1992 National Toxicology Program Institute of Environmental Health Sciences National Institutes of Health NTP.
Efficient mechanical deaeration reduces dissolved oxygen to 7 ppb or less. Place coverslip on at 45 degree angle. In conclusion the results presented in this study indicate that OTA within its water solubility range may be involved in the development of oxidative stress in HK-2 cells as indicated by an increase in ROS levels depletion of GSH and increased sensitivity to cellular defense against oxidative stress such as up-regulation of CAT GSR and SOD1 mRNA levels.
Another practical complication is the pK a-measurement of substances with a low water-solubility. Based on the results it is. DDQ decomposes in water but is stable in aqueous mineral acid.
Research Triangle Park North Carolina. The extraction yield was determined by 3 where m coal is the mass of the coal material dried at 110 C for 6 h. For complete protection from oxygen corrosion a chemical scavenger is required following mechanical deaeration.
Many TOC analysts suggest testing their instruments with two standards. It can partially oxidize some organic compounds. This process is called polymerization.
Solubility Solute Solvation Solvent Solvent shell Solvolysis SOMO sp orbital sp 2 orbital sp 3 orbital Space filling model Specific rotation Spectrometer Spectrometry Spectrophotometer Spectroscopy Spectrum Sphingolipid Sphingosine Spin quantum number Spin-spin coupling Spiro Splitting. Water Haworth projection HBr HCl HCFC Heat capacity. DO sensors can be designed for biochemical oxygen demand BOD tests spot sampling or long-term monitoring applications.
Compare entries 1 and 4 in Table 2. Focusing the microscope use lowest power objective first. By varying the.
Then 022 g of potassium persulfate was dissolved in 17 mL of deionized water and the obtained solution was. Finally the residue was weighed and its mass was labeled as m residue. In most cases the pigment.
The simplest quinone is benzoquinone which has two isomers. Change to higher objectives. Many redox initiators are water soluble and are widely used in emulsion.
1 HSO 5- 2 H 2 e- HSO 4- H 2 O. Caroat monopersulfate Oxone Curex. 14 moderate ILO International Chemical Safety Cards ICSC 328 Density.
The standard synthetic procedure was as follows. One example is extrapolation of measurements in solvent mixtures21 These require however a relatively high co-solvent concentration and are inherently inaccurate and time consuming. Water is a liquid at ambient conditions but it often co-exists on Earth with its solid state ice and gaseous state water vapor or steam.
For the latter benzoquinone is suggested. An alternative approach using surfactants is proposed22 which avoids tedious extrapolating while giving. 23-Dichloro-56-dicyano-14-benzoquinone or DDQ is the chemical reagent with formula C 6 Cl 2 CN 2 O 2.
They can be used on a much wider variety of substances than dyes because they are not reliant on water solubility for their application. Slowly cover the drop so that no air bubbles remain. A dissolved oxygen meter water quality sonde or data logging system can be used to record measurement data taken with a DO sensor.
Next the individual molecules of o-quinone connect together to make larger molecules. Place drop of water on clean microscopic slide. Solubility in water g100ml at 20 C.
Subsequently the slurry was filtered and the residue was washed with ethanol and water and dried at 75 C for 6 h under a vacuum. The iodinated anion exchange resin bed and the vaporization technique are not sufficiently. We found that a higher reaction temperature is beneficial for the oxidation of the DO mixture presumably due to better solubility 100 C.
Analysis of the. Parafocal - stays in focus after changing objectives. One typically easy for the instrument to oxidize KHP and one more difficult to oxidize.
It creates a compound called melanin which causes the apple to look brown. The JV plot of the resulting BiVO 4 FeOOHNiOOH photoanode for photooxidation of water was obtained in a 10 M potassium borate buffer pH. The equilibrium constant of the reaction O3 1 O3.
Ubidecarenone is a cofactor found in various dietary supplements.
In fact one of the uses for toluene is to produce benzene by removing the methyl group. Anilinium methyl sulphate CAMPHOR BENZALKONIUM METHOSULFATE 52793-97-2 258-190-8 6 15102010 3 Benzoic acid 2-hydroxy- 335-trimethylcyclohexyl ester Homosalate HOMOSALATE 118-56-9 204-260-8 10 02082021 4 2-Hydroxy-4-methoxybenzophenone Oxybenzone BENZOPHENONE-3 131-57-7 205-031-5 6 Reg EU 2017238 of 10 February 2017-date of.
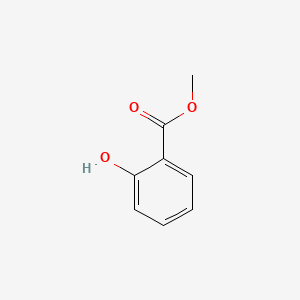
Methyl Salicylate C8h8o3 Pubchem
관련 정보 정보상자 각주.

Solubility methyl salicylate. Methyl salicylate can be used in consumer products other than cosmetic products such as household cleaning products air care products biocides eg. ACRYLATE POLYMERS 47 48. Ethylene glycol 329 170 110 260 Glycerol 361 174 121 293 Propylene glycol 302 168 94 233 Diethylene glycol.
Methyl salicylate oil of wintergreen found in topical liniments and solutions used in hot vapors is the most concentrated form of salicylate. Methyl salicylate will be metabolised in the body to salicylic acid which is also. Methyl salicylate 217 160 80 123 Polyhydric Alcohols.
We would like to show you a description here but the site wont allow us. 0098 g in 100 g at 25 C. Salicylic acid 또는 2-하이드록시벤조산영어.
1 ml of a 98 solution contains 1400 mg of. MentholMethyl Salicylate Cream Bengay Lidoderm 5 Patch all work for shorter period of time Task Force on Chronic Pain Management and the American Society of Regional Anesthesia and Pain Medicine. It was soon found that this compound had analgesic and antipyretic character almost identical to that of salicylic acid when taken internally.
Used for low solubility tablets to improve wetting and deaggregation of drug particles to get a rapid and improved dissolution. 달리 명시된 경우를 제외하면 표준상태25 C 77 F 100 kPa에서 물질의 정보가 제공됨. It can be manufactured synthetically and it used as a fragrance in foods beverages and liniments.
Methyl salicylate was first isolated in pure form in 1843 by extraction from wintergreen plant Gaultheria. Yalkowsky SH He Yan Jain P. John Wiley Sons.
The solubility of aspirin16 8. The xylene isomers have two methyl groups in different positions in the benzene ring and thus all aromatics are to some extent interchangeable. 1441 Drug Release Test Methods for Enteric Coated Products.
Disinfectants pest control products polishes and waxes which may increase the systemic exposure dose with a possibility to exceed the safe level. Thin-layer chromatography 17 Teachers and technicians guide to the activities. Siebert J Harke HP.
Methyl salicylate Benzoic acid Phenol Aspirin 4-Hydroxybenzoic acid Magnesium salicylate Choline salicylate Bismuth subsalicylate Sulfosalicylic acid. HPCHydroxy propyl cellulose 5. Enteric coating is a special case of a mechanism using erosion or dissolution of a coating to control release.
Especially used to control drug release in modified-release formulations prolonged-release or controlled-release tablet. Urinary excretion products include parent drug about 36 of the dose 5-isobutyl-5-2 3-dihydroxypropyl barbituric acid about 24 of the dose 5-allyl-53-hydroxy-2-methyl-1-propyl barbituric acid about 48 of the dose products with the barbituric acid ring hydrolyzed with excretion of urea about 14 of the dose as well as unidentified materials. Michelle Long Yisheng Chen in Developing Solid Oral Dosage Forms 2009.
Hazardous Substances Data Bank HSDB In water. According to ANSIADA Specification 57 63 the solubility of a root canal sealer should not exceed 3 by mass. The nuances of particle solubility in a given solvent should ultimately be investigated by the experimenter during assay optimization and this guide is not a substitute for bench top evaluationAdapted from.
Methyl salicylate oil of wintergreen or wintergreen oil is an organic ester naturally produced by many species of plants particularly wintergreens. Solubility is the mass loss of a material during a period of immersion in water. CRC Press Boca Raton FL 2010 p.
HPMCHydroxy propyl methyl cellulose 2. While the methyl group is usually part of a larger molecule it can be found on its own in any of three forms. It is produced by many species of plants particularly wintergreens.
Methyl salicylate oil of wintergreen or wintergreen oil is an organic compound with the formula C 8 H 8 O 3It is the methyl ester of salicylic acidIt is a colorless viscous liquid with a sweet fruity odor reminiscent of root beer but often associatively called minty as it is an ingredient in mint candies. Click to download pdf versionThis solubility list is based on the Hansen Solubility Parameters and should be used as a guide in methods development. Hazardous Ingredients as defined.
Salicylate Cinnamal Cinnamyl Alcohol Citral Citronellol Coumarin Eugenol Farnesol Geraniol Hexyl Cinnamal Hydroxycitronellal Isoeugenol Butylphenyl Methylpropional Limonene Linalool Hydroxyisohexyl 3-Cyclohexene Carboxaldehyde Methyl 2-Octynoate Alpha-Isomethyl Ionone Evernia Prunastri Oakmoss Extract Evernia FurfuraceaTreemoss Extract. Of the material excreted in the. HPMC It is prepared by reacting alkali treated cellulose with methyl chloride with propylene oxide.
As it forms bridging rough Tablet surface it has to be mixed with other polymers or. A highly soluble root canal sealer would invariably permit the formation of gaps within and between the material and the root dentin thereby providing avenues for leakage from the oral cavity and. A methyl group is an alkyl derived from methane containing one carbon atom bonded to three hydrogen atoms CH 3In formulas the group is often abbreviated MeSuch hydrocarbon groups occur in many organic compoundsIt is a very stable group in most molecules.
Salicylate UDP-alpha-D-glucose - salicylate beta-D-glucose ester UDP PlantCyc CPD-110 CPD-110 CPD-110 CPD-110 CPD-110 CPD-110 Salicylic acid is a natural product extract from Willow bark well known as an antiinflammatory inhibitor of cyclooxygenase activity. Ullmanns Encyclopedia of Industrial Chemistry. The compound was first extracted and isolated from plant species Gaultheria procumbens in 1843.
Fatty acids C12-18 Me esters sulfonated sodium salts. An enteric coating resists dissolution under acidic conditions but is freely soluble at the more basic conditions of the intestinal tract. MHECMethyl hydroxyl ethyl cellulose 3.
All of these hydrocarbons are useful as gasoline additives because of their antiknock properties high octane numbers. However when fragrance compounds containing methyl eugenol present naturally in essential oils are used as components in cosmetic products the highest concentration of methyl eugenol in the finished products must not go beyond 001 in fine fragrance 0004 in eau de toilette 0002 in a fragrance cream 00002 in other leave-on products and in oral hygiene products and 0001 in rinse. This medicinal effect probably results from the ease with which methyl salicylate is hydrolyzed to salicylic acid under the alkaline conditions.
It forms a colorless to yellow or reddish liquid. Anion cation or radical. Film former which may be enteric or non-enteric Solvent Plasticizer Colorant Opaquant.
Methyl salicylate Methyl 2-hydroxybenzoate Paracetamol N-4-Hydroxyphenylethanamide Phenacetin N-4-Ethoxyphenylethanamide Salicylic acid 2-Hydroxybenzoic acid Sodium salicylate Sodium-2-hydroxybenzoate Health and safety Teachers must consult their employers. Handbook of Aqueous Solubility Data Second Edition.