3 Chemical and Physical Properties Expand this section. The ICSC project is a common undertaking between the World Health Organization WHO and.
Determination Of Ascorbic Acid In Vitamin C Tables By Acid Base Titration By Joey Bacus Infographic
Ascorbic acid will increase the level or effect of aspirin by acidic anionic drug competition for renal tubular clearance.
Sodium hydroxide ascorbic acid reaction. In the redox reaction between ascorbic acid and MB the ascorbic acid is oxidized to dehydroascorbic acid while MB was reduced to colorless LMB as shown in the following. 16 days 34 hours in people who have excess levels of vitamin C Clearance. When a strongly electropositive center and hydroxide are attached to each other hydroxide may ionize itself to liberate a hydrogen cation and makes the parent compound as an acid.
1 Structures Expand this section. Sodium carbonate is an organic sodium salt and a carbonate salt. The cards are data sheets intended to provide essential safety and health information on chemicals in a clear and concise way.
The balanced equation for the reaction between ascorbic acid and sodium hydroxide is shown below. Sodium benzoate is best known as a food preservative though it has several other uses. Sodium bisulfate also known as sodium hydrogen sulfate is the sodium salt of the bisulfate anion with the molecular formula NaHSO 4Sodium bisulfate is an acid salt formed by partial neutralization of sulfuric acid by an equivalent of sodium base typically in the form of either sodium hydroxide lye or sodium chloride table salt.
1 Burets Burets are used when it is necessary to deliver a liquid to another container. The results indicate that the fluorescence intensity of the system is a linear function of vitamin C concentration in the range of mol L-1 and. It is a dry granular product that can be safely shipped.
Ascorbic or Citric Acid. People use baking soda in cooking baking cakes. Hydrogen chloride solution Hydrochloric acid solution.
What is ascorbic acid made from. It is a minor constituent of water. The main target users are workers and those responsible for occupational safety and health.
When a strong acid reacts with a weak base the base is unable to fully neutralize the. If a patient has a known allergic reaction to an inactive ingredient they should check for the ingredient in new prescription or over-the-counter medications or check with their pharmacist. The free fatty acid content was measured by acid-base titration meanwhile the reaction rate constant was determined by the equation ln1-XAkt.
Analytical 63 ACS reagent 44 Puriss 35 Cell Culture 21 Reagent 18 Purum 17 Anhydrous 13 BioXtra 13 ReagentPlus 11 Plant 7 Show More. In this application. Scientists also use ascorbic acid in fluorescence microscopy an essential tool to understanding cell biology.
The results showed that the hydrolysis of VCO. Sodium carbonate is formed after the reaction between sodium hydroxide a strong base and carbonic acid a weak acid. The primary aim of the cards is to promote the safe use of chemicals in the workplace.
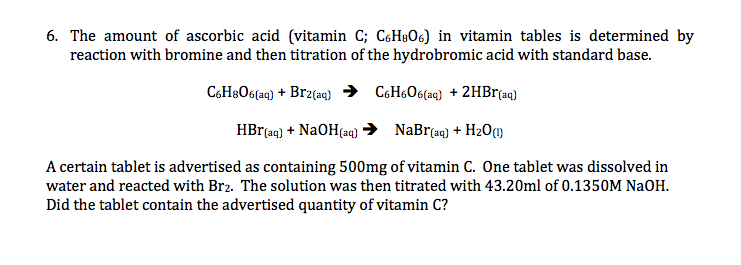
Keyword sigma aldrich Showing 1-30 of 553 results for sigma aldrich within Products. A certain tablet is advertised as co. The amount of ascorbic acid Vitamin C in tablets is determined by reaction with bromine and then titration of the hydrobromic acid with a standard base NaOH.
Aspirin decreases levels of ascorbic acid by increasing renal. Hover over products below to view reaction partners. Sodium Bicarbonate Sodium bicarbonate commonly known as baking soda is the regular item in our kitchens.
Keyword 7647-01-0 Showing 1-19 of 19 results for 7647-01-0 within Products. The ion forms salts where some of them dissociate in aqueous solution to liberate solvated hydroxide ions. Empirical Formula Hill Notation.
2 Names and Identifiers Expand this section. 16 Bulk sodium sulfate is usually purified via the decahydrate form since the anhydrous form. What is ascorbic acid.
Some synthetic vitamin C or ascorbic acid is produced with fermented corn syrup imported from China which may or may not have been genetically engineered. Sample collection and storage Collect samples in clean glass or. As a strong base sodium hydroxide neutralizes gastric acid thereby acting as an antacid.
Sodium acetate is formed after the reaction between a strong base sodium hydroxide a strong base and acetic acid a weak acid. Iron metal concentrated nitric acid HNO 3 was purchased from Fisher Scientific 60 M sodium hydroxide NaOH was purchased from wards science 60 M hydrochloric acid HCl was purchased from Fisher Scientific ascorbic acid was purchased from Acros and triazine present in the Iron Cell Test kit. A solution of baking soda is commonly.
Examples of inactive ingredients that are have been reported to cause reactions in some patients include. To remove the ascorbic acid the corn syrup is then treated with solvents such as acetone sulfuric acid or sodium hydroxide. Reaction of chromium with air Chromium metal does not react with air at room temperature.
This article provides a detailed overview of sodium benzoate including. Not only in soaps all the cleaning products including detergents and washing powder also contain sodium hydroxide. Ascorbic Acid Powder Pillow 2 PAN Indicator Solution 01 12 drops Stoppers for 18-mm tubes and AccuVac Ampul 2 Sample cells For information about sample cells adapters or light shields refer to Instrument-specific information on page 1 2 Refer to Consumables and replacement items on page 5 for order information.
Being a weak base it is safe for human consumption. Applies only to oral form of both agents. Alternatively sodium sulfate is or was formed in the production of lithium carbonate chelating agents resorcinol ascorbic acid silica pigments nitric acid and phenol.
Improve decision support research outcomes. Sort by Relevance. Ascorbic acid increases levels of aluminum hydroxide by enhancing GI absorption.
CrVI is reduced to CrIII by ascorbic acid reductive coefficient 347310 6 M-1 s-1 3. HC 6 H 7 O 6 aq NaOH aq H 2 O l NaC 6 H 7 O 6 aq You will titrate each Vitamin C sample with the standardized NaOH solution to determine the mg of ascorbic acid present in each sample. Analytical 4 ACS reagent 2 BioReagent 2 Technique.
Cause of formation of acidic basic and neutral salts. The calibration curve was made based on the concentration of MB mol L-113. Sulfites benzoates aspartame saccharin oleic acid benzyl alcohol lactose soya lecithin.
Polysorbate 80 is used to water solubilize the oil-soluble vitamins A D E and K. Ascorbic acid is used in a range of industrial and manufacturing applications including as a developing agent and preservative in photo production and in water purification where it is used to help remove the taste of iodine in sterilized potable water. 4 Spectral Information Expand this section.
With 14 polysorbate 80 sodium hydroxide andor hydrochloric acid for pH adjustment and water for injection. Sodium ascorbate is an organic sodium salt resulting from the replacement of the proton from the 3-hydroxy group of ascorbic acid by a sodium ionIt has a role as a food antioxidant a flour treatment agent a coenzyme a plant metabolite a human metabolite a Daphnia magna metabolite and a.
