Once a true hydrate had been heated. Greek prefixes are attached to the word hydrate to indicate the number of water molecules per formula unit for the compound eg BaOH 2 8H 2 O.

When Copper Ii Sulfate Pentahydrate Cuso Clutch Prep
Thus the product is called copper sulfate pentahydrate.
Copper sulfate pentahydrate formula. Ac-CoA Synthase Inhibitor - CAS 508186-14-9 - Calbiochem. What is the percent water in copperII sulfate pentahydrate CuSO 4 5 H 2 O. BaOH 2 8H 2 O.
When the chemical formula for a hydrated ionic compound is written the formula for the ionic compound is separated from the waters of hydration by a centered dot. A tolerance of 1 ppm is established in potable water for residues of copper resulting from the use of the algicides or herbicides basic copper carbonate malachite copper sulfate copper monoethanolamine and copper triethanolamine to control aquatic plants in reservoirs lakes ponds irrigation ditches and other potential sources of potable water. Copper II sulfate pentahydrate 100 -250 231-847-6 029-023-00-4 Acute ToxO 3.
The water in these compounds can be removed quantitatively by heating the compound with a bunsen burner. Because of the presence of copper the compound has a characteristic blue. When all the water has been removed the ionic compound is said to be anyhydrous.
It is found in foods such as organ meats seafood nuts seeds wheat bran cereals grain products and cocoa products. Calculate the formula mass. 8 water molecules octahydrate.
It is produced industrially by treating copper metal with hot concentrated sulfuric. Write hydrate after the prefix 1 mono 2 di 3 tri 4 tetra 5 penta 6 hexa 7 hepta 8 octa 9 nona 10 deca Example. For example the anhydrous compound copperII sulfate also exists as a hydrate containing five water molecules and named copperII sulfate pentahydrate.
A strong base is a base that is completely dissociated in an aqueous solutionThese compounds ionize in water to yield one or more hydroxide ion OH- per molecule of base. H410 7732-18-5 ZC0110000 Water 750 -900 231-791-2 NA No data available. One litre of Benedicts reagent can be prepared by mixing 173 grams of copper sulfate pentahydrate CuSO 45H 2 O 100 grams of sodium carbonate Na 2 CO 3 and 173 grams of sodium citrate in distilled water required quantity.
The body stores copper mostly in the bones and muscles. Copper sulfate pentahydrate CuSO45H2O or CuH10O9S CID 24463 - structure chemical names physical and chemical properties classification patents literature biological activities safetyhazardstoxicity information supplier lists and more. H315 Eye Damage 2.
This dark blue to purple solid is a salt of the metal complex CuNH 3 4 H 2 O 2. BaIO 3 2. Aqueous solution of copper2 sulfate is an acid.
In hydrated CuSO4 the water molecules surrounding the Central metal Cu function as ligands which bring d-d transition and hence emits blue colour in visible. C 27 H 42 N 7 Na 2 O 20 P 3 S xH 2 O. Use a prefix for the number of water molecules see below 3.
This compound can be prepared by adding concentrated solution of ammonia to a. Write the name of the ionic compound 2. Formula K sp 25 C Aluminium hydroxide.
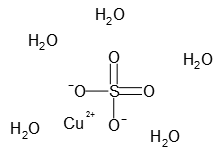
Copper is a mineral. Divide the mass of water in one mole. The most common form of copper sulfate is its pentahydrate given by the chemical formula CuSO 45H 2 O.
Washing soda is the common name for a hydrate of sodium carbonate containing 10 water molecules. Copper sulfate also known as blue vitriol Salzburg vitriol Roman vitriol blue copperas or bluestone is a chemical compound comprised of Copper Sulphur and Oxygen whose formula is CuSO4. 1 Cu6355 gmol 1 S3207 gmol 4 O1600 gmol 15962 gmol Formula mass 15962 gmol 5 H 2 0 1802 g H 2 0mol 24972 gmol 2.
CuSO 4 5H 2 O. Name Formula Systematic Name Common Name Formula Name Formula Methane CH 4 Methanoic acid Formic acid HCO 2H 12-Dichloroethane C 2H 4Cl 2 Ethane C 2H 6 Ethanoic acid Acetic acid CH 3CO 2H Methylamine CH 3NH 2 Propane C 3H 8 Propanoic acid Propionic acid C 2H 5CO 2H. The loss in mass of the compound after losing the water can be used to calculate the amount of water originally in the hydrated sample.
Copper sulfate CuSO 4 is an inorganic compound used in agriculture to control insects plant roots snails bacteria and fungi and its one of the best pool algaecides around. However it is the hydrated form of the salt and has 5 water molecules. It is closely related to Schweizers reagent which is used for the production of cellulose fibers in the production of rayon.
For example copper sulfate is a vernacular name which may refer to copperI sulfate or copperII sulfate. Stable at room temperature in closed containers under normal storage and handling conditions. The pentahydrate CuSO 4 5H 2 O the most commonly encountered salt is bright blue.
2286 SG 156 C 4 C 148gkg 0 C 736gkg 100 C Basic copper sulfate. Copper Sulphate is an odorless crystalline substance electric blue in color highly toxic and not safe to work with. CuSO4 5H 2 O Naming a Hydrate 1.
An illustration describing the. Empirical Formula Hill Notation. Blue crystals granules or powder.
The systematic name is. 95562 anhydrous basis Compare Product No. However it can be noted that the anhydrous form of this salt is a powder that is white.
Incompatibilities with Other Materials. Here the copperII sulfate acts as a source of Cu 2 ions the sodium carbonate provides an alkaline medium and the sodium citrate forms complexes with the Cu. This form is characterized by its bright blue colour.
2O copper II sulfate pentahydrate. Common Covalent Binary Inorganic Compounds. The pentahydrate x.
Hold eyelids apart and flush eyes with plenty of water for at least 15 minutes. When determining the formula mass for a hydrate the waters of hydration must be included. Write the formula of the anhydrous followed by a dot then the of moles of water and H 2O Example.
Some examples are copper II sulfate sodium sulfate nickel II sulfate iron II sulfate cobalt II chloride magnesium sulfate sodium. CopperII sulfate also known as copper sulphate are the inorganic compounds with the chemical formula CuSO 4 H 2 O x where x can range from 0 to 5. CopperII sulfate also known as copper sulphate are the inorganic compounds with the chemical formula Cu SO 4 H 2 O x where x can range from 0 to 5The pentahydrate x 5 is the most common form.
Archaic Name An archaic name is an older name for a chemical that predates the modern naming conventions. Specific gravity density 67. Ac-CoA Synthase Inhibitor - CAS 508186-14-9 - Calbiochem.
The ratio of water mol ecules to the anhydrous salt is constant. H319 Aquatic A 1. Vapor pressure mmHg at 25 C 1.
A hydrate is an ionic compound that has a definite amount of water molecules attached to its crystalline structure. CopperII sulfate pentahydrate CuSO 45H 2O 11 Undeca. Molecular FormulaCuO4S5H2O Molecular Weight24968 Section 10 - Stability and Reactivity Chemical Stability.
High temperatures dust generation exposure to moist air or water. The CuSO 4 molecule consists of an ionic bond between the copper cation Cu 2 and the sulfate anion SO 4 2-. Many compounds exist as hydrates.
Molecular weight gmol 3. 3CuOH 2 CuSO 4. Why is CuSO4 blue.
Public health information CDC Research information NIH SARS-CoV-2 data NCBI Prevention and treatment. Copper sulfate pentahydrate. H400 Aquatic C 1.
When copper sulfate CuSO4 reacts with water H2O the product is still copper sulfate. Older names for this compound include blue vitriol bluestone vitriol of copper and Roman vitriol. Determining the Formula of a Hydrate.
TetraamminecopperII sulfate is the salt with the formula CuNH 3 4SO 4 H 2 O. First Aid Measures Description of First Aid Measures.
H410 7732-18-5 ZC0110000 Water 750 -900 231-791-2 NA No data available. For example the anhydrous compound copperII sulfate also exists as a hydrate containing five water molecules and named copperII sulfate pentahydrate.

When Copper Ii Sulfate Pentahydrate Cuso Clutch Prep
First Aid Measures Description of First Aid Measures.

Formula for copper sulfate pentahydrate. What happens when hydrated copper sulphate is heated. CopperII sulfate pentahydrate CuSO 45H 2O 11 Undeca. Aqueous solution of copper2 sulfate is an acid.
Public health information CDC Research information NIH SARS-CoV-2 data NCBI Prevention and treatment. List of Sulfate Compounds Common Compounds of Sulfate SO4 Formula Molecular Weight. Losing water of crystallization turns.
Stable at room temperature in closed containers under normal storage and handling conditions. BaOH 2 8H 2 O. In hydrated CuSO4 the water molecules surrounding the Central metal Cu function as ligands which bring d-d transition and hence emits blue colour in visible.
1 Cu6355 gmol 1 S3207 gmol 4 O1600 gmol 15962 gmol Formula mass 15962 gmol 5 H 2 0 1802 g H 2 0mol 24972 gmol 2. Molecular weight gmol 3. Copper II sulfate pentahydrate 100 -250 231-847-6 029-023-00-4 Acute ToxO 3.
Copper sulfate pentahydrate CuSO45H2O or CuH10O9S CID 24463 - structure chemical names physical and chemical properties classification patents literature biological activities safetyhazardstoxicity information supplier lists and more. The most common form of copper sulfate is its pentahydrate given by the chemical formula CuSO 45H 2 O. Molecular FormulaCuO4S5H2O Molecular Weight24968 Section 10 - Stability and Reactivity Chemical Stability.
8 water molecules octahydrate. CopperII sulfate also known as copper sulphate are the inorganic compounds with the chemical formula Cu SO 4 H 2 O x where x can range from 0 to 5The pentahydrate x 5 is the most common form. Copper sulfate so that the titration requires 200 mL of the 06 standard dextrose solution.
Common Covalent Binary Inorganic Compounds. A strong base is a base that is completely dissociated in an aqueous solutionThese compounds ionize in water to yield one or more hydroxide ion OH- per molecule of base. Blue crystals granules or powder.
However it is the hydrated form of the salt and has 5 water molecules. What is the percent water in copperII sulfate pentahydrate CuSO 4 5 H 2 O. A tolerance of 1 ppm is established in potable water for residues of copper resulting from the use of the algicides or herbicides basic copper carbonate malachite copper sulfate copper monoethanolamine and copper triethanolamine to control aquatic plants in reservoirs lakes ponds irrigation ditches and other potential sources of potable water.
This form is characterized by its bright blue colour. When copper sulphate is heated it looses water molecules and hence it looses blue colour. Write hydrate after the prefix 1 mono 2 di 3 tri 4 tetra 5 penta 6 hexa 7 hepta 8 octa 9 nona 10 deca Example.
CuSO 4 5H 2 O. Write the name of the ionic compound 2. 3CuOH 2 CuSO 4.
Transfer the sample. Why is CuSO4 blue. Archaic Name An archaic name is an older name for a chemical that predates the modern naming conventions.
Use a prefix for the number of water molecules see below 3. Washing soda is the common name for a hydrate of sodium carbonate containing 10 water molecules. The CuSO 4 molecule consists of an ionic bond between the copper cation Cu 2 and the sulfate anion SO 4 2-.
O will be studied. H315 Eye Damage 2. This dark blue to purple solid is a salt of the metal complex CuNH 3 4 H 2 O 2.
CuSO4 5H 2 O Naming a Hydrate 1. In this experiment the hydrates of copperII sulfate CuSO. It is closely related to Schweizers reagent which is used for the production of cellulose fibers in the production of rayon.
It is found in foods such as organ meats seafood nuts seeds wheat bran cereals grain products and cocoa products. 2O copper II sulfate pentahydrate. When all the water has been removed the ionic compound is said to be anyhydrous.
Greek prefixes are attached to the word hydrate to indicate the number of water molecules per formula unit for the compound eg BaOH 2 8H 2 O. High temperatures dust generation exposure to moist air or water. H400 Aquatic C 1.
Specific gravity density 67. Calculate the formula mass. The name of the anhydrous salt is followed by a prefix indicating the number of water molecules followed by the word hydrate.
Copper sulfate pentahydrate. It is produced industrially by treating copper metal with hot concentrated sulfuric. Write the formula of the anhydrous followed by a dot then the of moles of water and H 2O Example.
2286 SG 156 C 4 C 148gkg 0 C 736gkg 100 C Basic copper sulfate. Dissolve 3464 g of reagent grade crystalline copper sulfate pentahydrate CuSO 45H 2O in. When copper sulphate pentahydrate is heated it loses water of crystallization as a result of evaporation.
Older names for this compound include blue vitriol bluestone vitriol of copper and Roman vitriol. Divide the mass of water in one mole. An illustration describing the.
The water molecules are loosely. For example AquaVet is a commercial product consisting of copper sulfate pentahydrate crystals the most common form and the recommended dosage is 1 tablespoon per 1000 gallons of pool water to achieve the ideal concentration of 1 ppm. TetraamminecopperII sulfate is the salt with the formula CuNH 3 4SO 4 H 2 O.
When copper sulfate CuSO4 reacts with water H2O the product is still copper sulfate. This compound can be prepared by adding concentrated solution of ammonia to a. Copper sulfate also known as blue vitriol Salzburg vitriol Roman vitriol blue copperas or bluestone is a chemical compound comprised of Copper Sulphur and Oxygen whose formula is CuSO4.
H319 Aquatic A 1. The systematic name is. Vapor pressure mmHg at 25 C 1.
O is named sodium thiosulfate pentahydrate. Name Formula Systematic Name Common Name Formula Name Formula Methane CH 4 Methanoic acid Formic acid HCO 2H 12-Dichloroethane C 2H 4Cl 2 Ethane C 2H 6 Ethanoic acid Acetic acid CH 3CO 2H Methylamine CH 3NH 2 Propane C 3H 8 Propanoic acid Propionic acid C 2H 5CO 2H. The formula of copper sulphate is CuSO4.
The body stores copper mostly in the bones and muscles. Once a true hydrate had been heated. The water in these compounds can be removed quantitatively by heating the compound with a bunsen burner.
O and magnesium sulfate MgSO. When the chemical formula for a hydrated ionic compound is written the formula for the ionic compound is separated from the waters of hydration by a centered dot. Formula K sp 25 C Aluminium hydroxide.
The pentahydrate CuSO 4 5H 2 O the most commonly encountered salt is bright blue. However it can be noted that the anhydrous form of this salt is a powder that is white. Thus the product is called copper sulfate pentahydrate.
Hold eyelids apart and flush eyes with plenty of water for at least 15 minutes. BaIO 3 2. When determining the formula mass for a hydrate the waters of hydration must be included.
Copper Sulphate is an odorless crystalline substance electric blue in color highly toxic and not safe to work with. Incompatibilities with Other Materials. The loss in mass of the compound after losing the water can be used to calculate the amount of water originally in the hydrated sample.
For example copper sulfate is a vernacular name which may refer to copperI sulfate or copperII sulfate. Copper is a mineral. 1 aqueous solution PROCEDURE Weigh accurately an amount of sample such that after dilution the solution contains about 06 reducing sugars Note 4.
Copper II sulfate pentahydrateA few things to consider when finding the molar mass for CuSO4. Hold eyelids apart and flush eyes with plenty of water for at least 15 minutes.

Copper Ii Sulfate Pentahydrate Cas 7758 99 8 Glentham Life Sciences
But approved pesticide uses may change periodically and so federal state and local authorities must be consulted for currently approved uses US.
Copper ii sulfate pentahydrate. Get it as soon as Wed Dec 1. Other names include copper 2 tretraoxidosulfate or copper II. Hydrated ionic compounds ie hydrates have a specfic number of water molecules in their chemical formulas.
CuSO45H2Os heat CuSO4s 5H2OgIf 363g CuSO45H2O is decomposed to CuSO4 predict. H410 7732-18-5 ZC0110000 Water 750 -900 231-791-2 NA No data available. Pure copperII sulfate is white.
And copperII sulfate pentahydrate - CuSO 4 5 H 2 O. When copperII sulfate pentahydrate CuSO45H2O is heated it decomposes to the dehydrated form. Copper II sulfate pentahydrate 100 -250 231-847-6 029-023-00-4 Acute ToxO 3.
2O copper II sulfate pentahydrate. Copper sulfate is an algaecide bactericide and fungicide. This dark blue to purple solid is a salt of the metal complex CuNH 3 4 H 2 O 2.
Other examples of hydrates are. None listed Cupric sulfate anhydrous none listed 1 mgm3 TWA as Cu except Copper fume listed under Copper compounds nos100 mgm3 IDLH dust and mist as Cu listed under Copper. More Buying Choices 1799 2 new offers Laboratory-Grade Copper II Sulfate 5-Hydrate Powder 500g - The Curated Chemical Collection.
4 Spectral Information Expand this section. CopperII sulfate pentahydrate CuSO 45H 2O 11 Undeca. Using the procedure below it should take no more than 30 minutes to produce the filtered salt solution.
2 Names and Identifiers Expand this section. It has a role as a sensitiser a fertilizer and an emetic. For CopperII sulfate pentahydrate USEPAOPP Pesticide Code.
For example copper sulfate is a vernacular name which may refer to copperI sulfate or copperII sulfate. Explanation of how to find the molar mass of CuSO4 5H2O. When all the water has been removed the ionic compound is said to be anyhydrous.
Once a true hydrate had been heated. When water is present in a sample of copperII sulfate it turns blue. 1 Structures Expand this section.
FREE Shipping on orders over 25 shipped by Amazon. CopperII sulfate pentahydrate none listed 1 mgm3 TWA as Cu except Copper fume listed under Copper compounds nos100 mgm3 IDLH dust and mist as Cu listed under Copper compounds nos. Environmental Protection AgencyOffice of Pesticide Programs Chemical Ingredients Database on Copper.
Magnesium carbonate pentahydrate - MgCO 3 5H 2 O. Benedicts reagent also known as Benedicts solution is a chemical reagent which is made up of a complex mixture of sodium citrate sodium carbonate and the pentahydrate of copperII sulfate. H315 Eye Damage 2.
By reacting copperII oxide a black solid with colourless dilute sulfuric acid they produce copperII sulfate with a characteristic blue colour. This compound can be prepared by adding concentrated solution of ammonia to a. 1 The International Union of Pure and Applied Chemistry IUPAC name for this active ingredient is copper 2 sulfate or copper II sulfate.
47 out of 5 stars 153. Its helpful to know archaic names of chemicals because older texts may refer to chemicals by these names. First Aid Measures Description of First Aid Measures.
Solutions of copperII sulfate are also blue. It is still a dry solid because the individual water molecules are trapped within the ionic lattice surrounding the copperII ions. An illustration describing the.
Cu2 CopperII 2cupric. H319 Aquatic A 1. The most common form of copper sulfate is its pentahydrate given by the chemical formula CuSO 45H 2 O.
When all hydrating water is removed the material is said to be anhydrous and is referred to as. The water in the hydrate referred to as water of hydration can be removed by heating the hydrate. The balanced equation is.
We regularly produce chemical solutions to specifications designed by government and regulatory bodies commercial and trade associations and the specific needs of individual users and businesses. The body stores copper mostly in the bones and muscles. It is found in foods such as organ meats seafood nuts seeds wheat bran cereals grain products and cocoa products.
Copper Sulfate Pentahydrate - Crystals - 252 Cu - 5 Pounds. Students can then obtain blue copperII sulfate pentahydrate crystals. Our product line consists of chemical solutions prepared to exact quality standards and certified for use in laboratories and production processes.
Registered for use in the US. This form is characterized by its bright blue colour. 024401 ACTIVE products with label matches.
Get it as soon as Wed Dec 1. 3 Chemical and Physical Properties Expand this section. When it is mixed with calcium hydroxide it is known as Bordeaux mixture.
CopperII sulfate is a metal sulfate compound having copper2 as the metal ion. An archaic name is an older name for a chemical that predates the modern naming conventions. TetraamminecopperII sulfate is the salt with the formula CuNH 3 4SO 4 H 2 O.
A strong base is a base that is completely dissociated in an aqueous solutionThese compounds ionize in water to yield one or more hydroxide ion OH- per molecule of base. H400 Aquatic C 1. When exposed to reducing sugars and other reducing substances Benedicts reagent changes its colour from clear blue to brick red.
40 out of 5 stars 7. FePO 4 2H 2 O. The water in these compounds can be removed quantitatively by heating the compound with a bunsen burner.
Lithium perchlorate trihydrate - LiClO 4 3H 2 O. The CuSO 4 molecule consists of an ionic bond between the copper cation Cu 2 and the sulfate anion SO 4 2-. The loss in mass of the compound after losing the water can be used to calculate the amount of water originally in the hydrated sample.
Experimental work can begin without delay if the dilute sulfuric acid. Copper is a mineral. However it can be noted that the anhydrous form of this salt is a powder that is white.
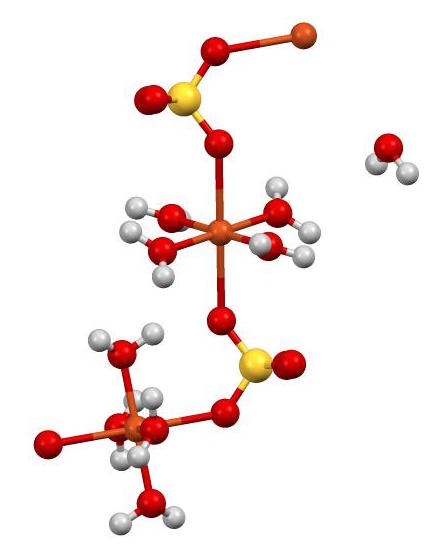
It contains a copper2. Older names for this compound include blue vitriol bluestone vitriol of copper and Roman vitriol. The waters of hydration are released from the solid crystal and form water vaporThe hydrated form is medium blue and the dehydrated solid is light blue.
It is closely related to Schweizers reagent which is used for the production of cellulose fibers in the production of rayon. CopperII sulfate also known as copper sulphate are the inorganic compounds with the chemical formula Cu SO 4 H 2 O x where x can range from 0 to 5The pentahydrate x 5 is the most common form. 5 Related Records Expand this.
Some chemicals are sold under archaic names or may be found in. The pentahydrate CuSO 4 5H 2 O the most commonly encountered salt is bright blue. Common Covalent Binary Inorganic Compounds of atoms Prefix element closest to fluorine goes on rightCommon Examples 1 Mono H 2 Hydrogen N 2 Nitrogen 2 Di O 2 Oxygen NH 3 Ammonia 3 Tri O 3 Ozone NO Nitrogen monoxide Nitric Oxide 4 Tetra H 2O Water Dihydrogen Monoxide NO 2 Nitrogen.
In the solid these water molecules also called waters of hydration are part of the structure of the compound. Cu 3 PO 4 2.
Benedicts solution is made by dissolving 173 g monosodium citrate 173 g cupric sulphate pentahydrate and 100 g anhydrous sodium carbonate in distilled water. Pesticides containing copper sulfate monohydrate andor copper sulfate anhydrous have been cancelled by the United States Environmental.

Copper Ii Sulfate Pentahydrate 99 Ph Eur Grade Cas 7758 99 8 Glentham Life Sciences
The pentahydrate CuSO 4 5H 2 O the most commonly encountered salt is bright blue.

Cupric sulfate pentahydrate. Cupric sulfate anhydrous none listed 1 mgm3 TWA as Cu except Copper fume listed under Copper compounds nos100 mgm3 IDLH dust and mist as Cu listed under Copper compounds nos. Cupric sulphate Blue vitriol pentahydrate Bluestone pentahydrate Bonattite trihydrate mineral Boothite heptahydrate mineral Chalcanthite pentahydrate mineral Chalcocyanite mineral Copper Sulphate pentahydrate. Aluminum nitrite AlNO 23 64.
Cupric sulfate may also have a role in iron turnover ascorbic acid metabolism phospholipid metabolism myelin formation glucose homeostasis and cellular immune defense. No OSHA Vacated PELs are listed for this chemical. Agriculture soil additive pesticides Bordeaux mixture water treating applications animal feed additive germicides textile mordant leather industry pigments electric batteries electroplated coatings copper salts reagent in analytical chemistry medicine wood preservative preservation of pulp wood and ground pulp.
Formulations include basic copper sulfate copper sulfate monohydrate copper sulfate pentahydrate and copper sulfate anhydrous. Cupric bromate CuBrO 22 67. 002 hydrate Analysis Pre Lab v1 Docx Name Date H.
Copper sulfate pentahydrate appears as blue crystalline granules or powder. After the metal passes through the basolateral membrane it is transported to the liver attached to serum albumin. Ammonium Sulfate Ammonium ions and Sulfate ions NH 4 and SO 4 2 NH 4 2SO 4 Calcium Phosphate Calcium ions and Phosphate ions Ca2 and PO 4 3 Ca 3PO 2 Waters of hydration use table to left copperII sulfate pentahydrate CuSO 45H 2O 11 Undeca.
The systematic name also called the IUPAC name is the preferred way to name a chemical because each systematic name identifies exactly one chemical. No OSHA Vacated PELs are listed for this chemical. Sodium carbonate is used to maintain an alkaline medium required for the redox reaction during the test.
Sodium carbonate renders alkaline conditions which are required for the redox reaction while sodium citrate is a. Beryllium chloride BeCl2 91. However the latter is the preferred compound described by the term copper sulfate.
The final product can be employed to check for the presence of reducing sugars. This dark blue to purple solid is a salt of the metal complex CuNH 3 4 H 2 O 2It is closely related to Schweizers reagent which is used for the production of cellulose fibers in the production of rayon. Silver nitrate is an inorganic compound discovered in the 13 th century by Albertus Magnus and has since been used in the medical industry for various issues including wound care.
Composition and Preparation of Benedicts. Silver oxide Ag 2O 65. The final volume of the solution is one litre.
When the conditions are carefully controlled the colouration developed and the amount of precipitate formed Cuprous oxide depends upon the amount of reducing sugars present. CID 1118 Sulfuric acid CID 23978 Copper CID 962 Water Dates. Copper II sulfate exothermically dissolves in water to give the aquo complex CuH2O62 which has octahedral molecular geometry and is paramagnetic.
Benedicts solution or Benedicts reagent can be prepared by complexing cupric ions Cu2 cations from the copper sulfate pentahydrate with citric acid molecules in a basic environment provided by sodium carbonate. Older names for this compound include blue vitriol bluestone vitriol of copper and Roman vitriol. Magnesium sulfate is gaining popularity as an initial.
CopperII sulfate also known as copper sulphate are the inorganic compounds with the chemical formula Cu SO 4 H 2 O x where x can range from 0 to 5The pentahydrate x 5 is the most common form. Hydrated ionic compounds ie hydrates have a specfic number of water molecules in their chemical formulas. Calcium bicarbonate CaHCO 3 92.
As silver salts have antiseptic properties they have been used as a treatment for gonorrhea prevention a cauterizing agent for the healing of oral ulcers among others. When hydrogen sulphide gas is passed through a blue solution of copper sulphate a black precipitate of copper sulphide is obtained and the sulphuric acid so formed remains in the solution. In the solid these water molecules also called waters of hydration are part of the structure of the compound.
식별자 CAS 번호. In this reaction cupric ions from copper sulphate combine with sulphide ions from hydrogen sulfide to. Hydrobromic acid HBr 89.
Magnesium chloride pentahydrate 86. TetraamminecopperII sulfate is the salt with the formula CuNH 3 4SO 4 H 2 O. Magnesium is reacted with oxygen from the air in a crucible and the masses before and after the oxidation are measured.
Systematic or IUPAC Name. 61 gmol Q3 - 5 Q4 - CuSO 4 5H 2 O Q5 - copperII sulfate pentahydrate Q6 - Turned back to blue hydrateComposition of Hydrates. It causes direct inhibition of action potentials in myometrial muscle cells.
So sodium citrate is used to stabilize the copper ions by complexing with them. Benedicts solution is a deep-blue alkaline chemical reagent used to test for the presence of the aldehyde functional group -CHO which consists of copper sulfate pentahydrate CuSO 4. 5H 2 O sodium carbonate Na 2 CO 3 sodium citrate Na 3 C 6 H 5 O 7 and distilled water.
Sulfur trioxide SO 3 63. The chemical formula of Benedicts solution is not present since the benedicts solution is a mixture and not a chemical compound. Melting point 110C with decomposition.
Hydrogen fluoride HF 88. The liver is the critical organ for the homeostasis of copper. TinII sulfide SnS 90.
The CuSO 45H 2 Ocopper sulfate pentahydrate salt is the source for the blue color due to the presence of copperII ions. Copper sulfate is a term that can refer to either of the following chemical compounds cuprous sulfate Cu 2 SO 4 or cupric sulfate CuSO 4. Ammonium sulfate NH 42SO 4 87.
Excitation and contraction are uncoupled which decreases the frequency and force of contractions. The copper is then prepared for excretion. They can reduce cupric ions Cu 2 to cuprous form Cu which is responsible for the change in color of the reaction mixture.
Calcium nitride Ca 3N2 MgCl25H 20 62. Zinc Sulfate Monohydrate Our Associate Companies A progressive and dynamic company Buradon is one of India s leading manufacture and exporter of fine chemicals to the rigid standard of GR AR EL ACS and LR grades due to its industrial exposure commitment and market fixing capabilities from India. Copper II can sometimes degrade to copperI.
Water is just added as a solvent for. IP BP EP Ph Eur USP NF JP Analytical Reagent FCC Food Grade Chemicals Excipients Shale Gas Fracturing Chemicals Manufacturers. None listed OSHA Vacated PELs.
The systematic name is determined by guidelines set forth by the International Union of Pure and Applied Chemistry IUPAC. Magnesium sulfate is a small colorless crystal used as an anticonvulsant a cathartic and an electrolyte replenisher in the treatment of pre-eclampsia and eclampsia. The anhydrous form is a pale green or gray-white powder whereas the pentahydrate the most commonly encountered salt is bright blue.
Ammonium phosphate NH 43PO 4 66. The mass of the hydrated salt obtained was 7. Other names for copperII sulfate are blue vitriol and.
7758-98-7 7758-99-8 pentahydrate 16448-28-5 trihydrate 19086-18-1 heptahydrate 3D 모델. This is the basis of Benedicts test. The systematic name for CuSO 4 is copperII sulfate but it is also referred to as blue vitriol Roman vitriol the vitriol of copper and bluestone.
Product Applications for Copper Cupric Sulfate Pentahydrate. Muby Chemicals of Mubychem Group established in 1976 is the original manufacturers of Specialty Chemicals Pharmaceutical Excipient Fragrance Food Flavor chemicals Reagent Grade Chemicals Shale Gas Fracturing Chemicals in India. Their Chemical Abstracts Service CAS registry numbers are 1344-73-6 1332-14-5 7758-99-8 and 7758- 98-7 respectively.
Sodium Thiosulfate is a water soluble salt and reducing agent that reacts with oxidizing agents. For example AquaVet is a commercial product consisting of copper sulfate pentahydrate crystals the most common form and the recommended dosage is 1 tablespoon per 1000 gallons of pool water to achieve the ideal concentration of 1 ppm.

How To Write The Formula For Copper Ii Sulfate Pentahydrate Youtube
Molecular weight gmol 3.

Sulfate pentahydrate formula. Ac-CoA Synthase Inhibitor - CAS 508186-14-9 - Calbiochem. When determining the formula mass for a hydrate the waters of hydration must be included. IsoWhey Clinical Nutrition Diabetic Formula has been specially designed to support the nutritional needs of people with diabetes who cannot meet their diet needs through diet modification alone.
CopperII sulfate pentahydrate CuSO 45H 2O 11 Undeca. It is soluble in water and slightly soluble in alcohol. Write the name of the ionic compound 2.
What is the molar ratio between the anhydrous compound and. Ac-CoA Synthase Inhibitor - CAS 508186-14-9 - Calbiochem. Older names for this compound include blue vitriol bluestone vitriol of copper and Roman vitriol.
Write hydrate after the prefix 1 mono 2 di 3 tri 4 tetra 5 penta 6 hexa 7 hepta 8 octa 9 nona 10 deca. Vapor pressure mmHg at 25 C 1. IsoWhey Clinical Nutrition Diabetic Formula is designed to support sugar metabolism by providing a high protein nutritional meal replacement.
2 Note that after heating you end up with anhydrous magnesium sulfate MgSO 4 and you will have remaining the same number of moles of the anhydrous product as you had moles of the hydrated reactant. List of Sulfate Compounds Common Compounds of Sulfate SO4 Formula Molecular Weight. Remember hydrates have a fixed amount of water per mole of the non-water compound.
Copper sulfate pentahydrate CuSO45H2O or CuH10O9S CID 24463 - structure chemical names physical and chemical properties classification patents literature biological activities safetyhazardstoxicity information supplier lists and more. List of Thiosulfate Compounds Common Compounds of Thiosulfate S2O3 Formula Molecular Weight. Some examples are copper II sulfate sodium sulfate nickel II sulfate iron II sulfate cobalt II chloride magnesium sulfate sodium.
Determining the Formula of a Hydrate. II sulfate pentahydrate listed as Copper compounds nos 100 CAS 7758-99-8 which is subject to the reporting requirements of Section 313 of SARA Title III and 40 CFR Part 373. Molecular FormulaCuO4S5H2O Molecular Weight24968 Section 10 - Stability and Reactivity.
Solubility product constant K sp or the solubility product is the product of the molar concentrations of the constituent ions each raised to the power of its stoichiometric coefficient in the equilibrium equationFor instance if a compound A a B b is in equilibrium with its solution. 290 g 2464696 gmol 011766157 mol. In this example the molar ratio is 15.
It is typically found in its pentahydrate form which is either white in colour or colourless altogether. Copper sulfate so that the titration requires 200 mL of the 06 standard dextrose solution. A strong base is a base that is completely dissociated in an aqueous solutionThese compounds ionize in water to yield one or more hydroxide ion OH- per molecule of base.
What is the percent water in copperII sulfate pentahydrate CuSO 4 5 H 2 O. Many compounds exist as hydrates. Empirical Formula Hill Notation.
Although its exact mechanism of action is unknown thiosulfate likely provides an exogenous source of sulfur thereby hastening the detoxification of cyanide through the enzyme rhodanese thiosulfate cyanide sulfurtransferase which converts cyanide to the relatively nontoxic excretable. Systematic or IUPAC Name. Calculate the formula mass.
The table below gives calculated values of K. The systematic name is determined by guidelines set forth by the International Union of Pure and Applied Chemistry IUPAC. The sulfate or sulphate ion is a polyatomic anion with the empirical formula SO 2 4.
For every one mole of CuSO 4 there are five moles of water. Common Covalent Binary Inorganic Compounds. It can be consumed as a delicious shake and provides.
83762 free acid basis Compare Product No. An illustration describing the. Morphine Sulfate Tablets are an opioid agonist available in 15 mg and 30 mg for oral administration.
Specific gravity density 67. 1 Cu6355 gmol 1 S3207 gmol 4 O1600 gmol 15962 gmol Formula mass 15962 gmol 5 H 2 0 1802 g H 2 0mol 24972 gmol 2. Chemically Morphine Sulfate is 78-didehydro-45α-epoxy-17-methylmorphinan-36α-diol sulfate 21 salt pentahydrate.
Copper sulfate pentahydrate. 1 aqueous solution PROCEDURE Weigh accurately an amount of sample such that after dilution the solution contains about 06 reducing sugars Note 4. Sulfates occur widely in everyday life.
2286 SG 156 C 4 C 148gkg 0 C 736gkg 100 C Basic copper sulfate. 1 Convert the 290 g of magnesium sulfate heptahydrate to moles. 3CuOH 2 CuSO 4.
CuSO4 5H 2 O Naming a Hydrate 1. Divide the mass of water in one mole. However it can be noted that the anhydrous form of this salt is a powder that is white.
This material contains Cupric sulfate anhydrous listed as Copper compounds nos - CAS 7758-98-7 which. Salts acid derivatives and peroxides of sulfate are widely used in industry. Use a prefix for the number of water molecules see below 3.
Name Formula Systematic Name Common Name Formula Name Formula Methane CH 4 Methanoic acid Formic acid HCO 2H 12-Dichloroethane C 2H 4Cl 2 Ethane C 2H 6 Ethanoic acid Acetic acid CH 3CO 2H Methylamine CH 3NH 2 Propane C 3H 8 Propanoic acid Propionic acid C 2H 5CO 2H. Dissolve 3464 g of reagent grade crystalline copper sulfate pentahydrate CuSO 45H 2O in. The systematic name also called the IUPAC name is the preferred way to name a chemical because each systematic name identifies exactly one chemical.
Transfer the sample. Notice that this is not a chemical reaction it is simply a chemical formula of a hydrate. This pentahydrate of sodium thiosulfate is described by the following chemical formula.
Spelling Sulfate is the spelling recommended by IUPAC but sulphate was traditionally used in British English. Write the formula of the anhydrous followed by a dot then the of moles of water and H 2O Example. K sp M n m A m- n.
Na 2 S 2 O 35H 2 O. Blue crystals granules or powder. CopperII sulfate also known as copper sulphate are the inorganic compounds with the chemical formula Cu SO 4 H 2 O x where x can range from 0 to 5The pentahydrate x 5 is the most common form.
C 25 H 42 N 7 O 17 P 3 S xLi CAS No. Where M m A n is the slightly soluble substance and M n and A m-are the ions produced in solution by dissosiation of M m A n. The pentahydrate CuSO 4 5H 2 O the most commonly encountered salt is bright blue.
CuSO 4 5H 2 O. The CuSO 4 molecule consists of an ionic bond between the copper cation Cu 2 and the sulfate anion SO 4 2-. For ionic compounds with limited solubility in water an equilibrium constant K sp can be defined from the ion concentration in water from the equation.
This form is characterized by its bright blue colour. The ratio of water mol ecules to the anhydrous salt is constant. The most common form of copper sulfate is its pentahydrate given by the chemical formula CuSO 45H 2 O.
In other words there is a 11 molar. A hydrate is an ionic compound that has a definite amount of water molecules attached to its crystalline structure. Sulfates are salts of sulfuric acid and many are prepared from that acid.
Morphine Sulfate USP is a white to off-white crystalline solid. M m A n s mM n aq nA m-aq. Sodium thiosulfate which is also referred to as sodium sulphate is a chemical compound that has the formula Na 2 S 2 O 3.
Public health information CDC Research information NIH SARS-CoV-2 data NCBI Prevention and treatment.