Lithium has been associated with rare instances of mild serum aminotransferase elevations but has not been convincingly linked to clinically apparent acute liver injury. Cash Operating Costs per tonne of lithium carbonate US2914.
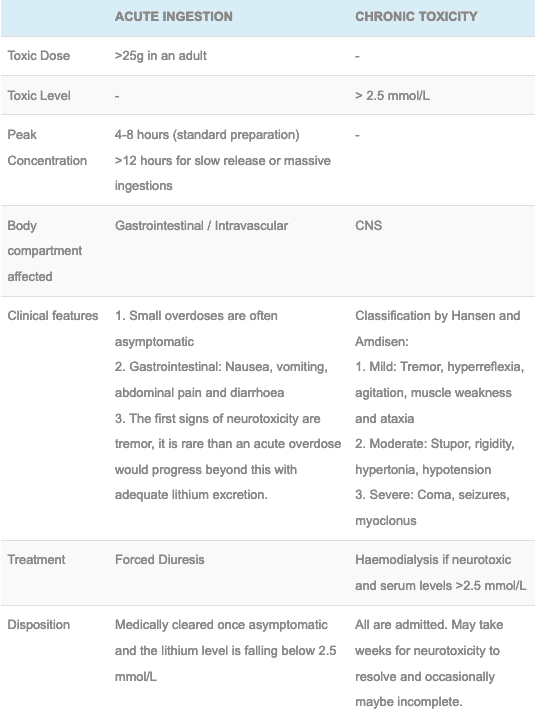
Lithium Toxicity Litfl Toxicology Library Toxicants
One of the issues is the deposition of metallic lithium on the anode graphite surface under fast charging or low-temperature conditions.
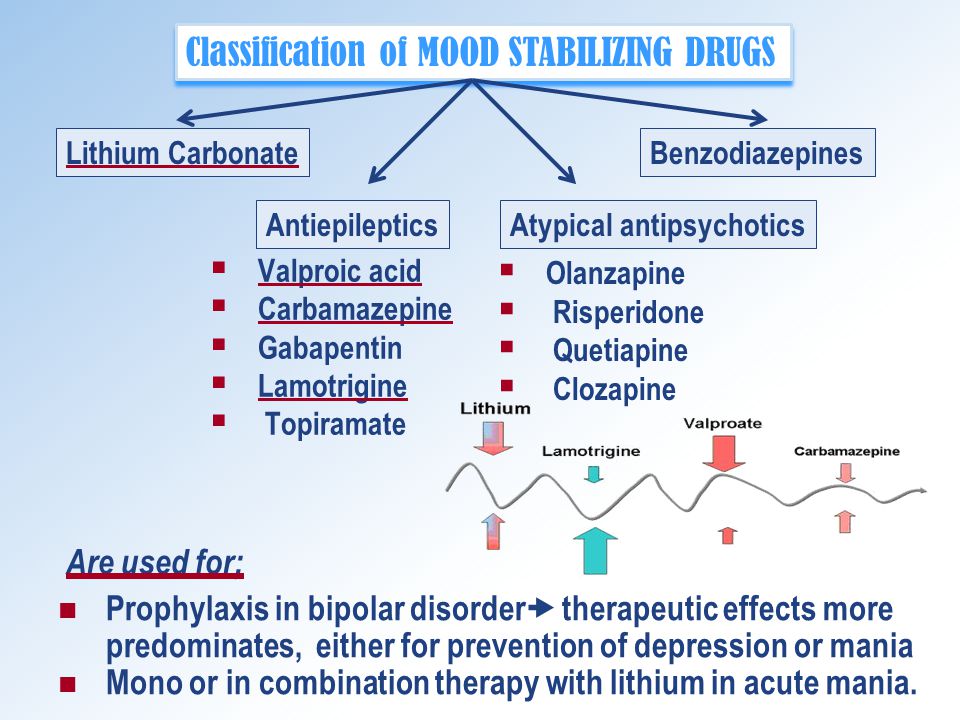
Lithium carbonate classification. 11 Although utilization of lithium is not as widespread as in the past it is commonly prescribed. In this piece we highlight four companies that represent key. 26 2021 CNW - Neo Lithium Corp.
Because lithium reacts violently with water non-aqueous electrolytes must be used. Thus it is essential to determine the rapid and accurate methods for separation. From the energy security to carbon footprint from daily life to industrial growth from environmental safety to mental health the lithium is a very important commodity.
Therefore lithium orotate can be used in much lower doses eg. It is a precursor to other salts including ceramics and materials for lithium batteries. However there are still many issues facing lithium-ion batteries.
She was stable until a non-steroidal anti-inflammatory drug NSAID was added leading to fatal lithium toxicity. It is classified as the cyclic carbonate ester of ethylene glycol and carbonic acidAt room temperature 25 C ethylene carbonate is a transparent crystalline solid practically odorless and colorless and somewhat soluble in water. Lithium has the highest specific energy of all but it has only become possible since mid 1970s to manufacture practical batteries.
These raw materials then move into the chemical conversion process to produce lithium carbonate or lithium hydroxide. Average Annual Production lithium carbonate 20000. Lithium interferes with transmembrane sodium exchange in nerve cells by affecting sodium potassium-stimulated adenosine triphosphatase Na K-ATPase.
Clinical trials involving 150 mg daily doses of lithium orotate administered 4 to 5 times a week. Mineral regions were digitized as shown in the source reports and thus may be represented as points or polygons. In addition it is found dissolved in blood plasma andor in erythrocytes.
Yellow color may be removed but another acid dye or lithium carbonate or washing with changes of 50-70 alcohol. And blocks inositol. Use the Search on the right to filter publications by divisions programs or keywords.
Ethylene carbonate sometimes abbreviated EC is the organic compound with the formula CH 2 O 2 CO. The first association of lithium with psoriasis was reported in 1972 and since then there have been several reports of lithium-induced. NE2 is pleased to announce positive results of a National Instrument 43-101 Feasibility Study FS for the production of lithium carbonate from its wholly owned Tres Quebradas lithium brine project 3Q Project in Catamarca Province Argentina.
Lithium hydroxide LiOH is used to remove carbon dioxide from the atmosphere of spacecraft. It is a mood stabilizer. Lithium stearate LiC 18 H 35 O 2 is used as a general purpose and high temperature lubricant.
Lithium is mixed with other light metals such as aluminum and magnesium to form strong light-weight alloys an alloy is a mixture of metals. The Electrochemical Society was founded in 1902 to advance the theory and practice at the forefront of electrochemical and solid state science and technology and allied subjects. 1129 billion after-tax NPV with 8 discount rate and a 395 IRR at an average price 12321 t LCE with a 50-year life of mine and payback of 2.
These salts and many other lithium salts exhibit distinctively high solubility in ethers in. For example the commodity lithium carbonate Li2CO3 is reported in the Material field. Lithium Carbonate Extended-Release Tablets USP contain lithium carbonate a white odorless alkaline powder with molecular formula Li 2 CO 3 and molecular weight 7389.
Lithium carbonate tablets Priadel 200mg 400mg Lithium citrate liquid Priadel 520mg5ml 5ml equivalent of 204mg lithium carbonate Other brands of lithium carbonate tablets include Camcolit 250mg400mg and Liskonum 450mg. Another form of lithium called lithium orotate is preferred because the orotate ion crosses the blood-brain barrier more easily than the carbonate ion of lithium carbonate. Lithium- and Drug-Provoked Psoriasis.
Other brands of lithium citrate liquid include Li-Liquid 509mg5ml1018mg5ml. Lithium carbonate Li 2 CO 3 is used as a drug to treat manic depression disorder. This white solid is the principal product of beneficiation of lithium ores.
5 mg with remarkable results and no side effects 4950. Lithium-ion batteries have many important properties to meet a wide range of requirements especially for the development of electric mobility. Lithium reacts with water but not as violently as sodium.
Dosage Route ADULTS. Lithiums toxic effects limited its clinical use until appropriate serum monitoring became more readily available. Lithium salts particularly lithium carbonate Li 2 CO 3 and lithium citrate are used in the treatment of mania and bipolar depression although lately its use has been extended to unipolar depression.
Its effects are thought to be based on its active effects on human serotonin function. Some lithium in the form of lithium carbonate or lithium citrate is used as medicine to treat gout an inflammation of joints and to treat serious mental illness. Lithium is a simple alkali metal the salt of which acts as a mood stabilizing agent which has been extensively used for the treatment of mania for more than 50 years.
600 mg PO tid or 900 mg slow-release form PO bid to produce effective serum levels between 1 and 15. Organic solvents such as acetonitrile and propylene carbonate plus inorganic solvents such as thionyl chloride SOCl2 are typical with a compatible solute to provide conductivity. Publications are listed in the order they are published.
A 72-year-old woman with impaired renal function was prescribed lithium and cilazapril as well as other medicines. Lithium is an element of the alkali-metal group with atomic number 3 atomic weight 694 and an emission line at 671 nm on the flame photometer. Thousands of cells may be combined to create a battery pack for an EV.
It was suspected that the NSAID triggered lithium toxicity due to a pharmacokinetic interaction. Proteins comprise more than 50 of. Alters the release of neurotransmitters.
The compounds LiBH 4 and LiAlH 4 are useful reagents. The USGS authors convert the reported quantity of Li2CO3 to the elemental form Li in SI units and populate the fields CntSIComAm and CntSIComUt accordingly. Affects cyclic adenosine monophosphate concentrations.
Lithium lithium carbonate lithium citrate. Lithium is industrially produced mainly as lithium carbonate lithium hydroxide lithium chloride lithium bromide and butyl lithium. Carbolith CAN Duralith CAN Eskalith Eskalith CR Lithane CAN Lithobid Lithonate Lithotabs PMS-Lithium Carbonate CAN Classification.
Lithium carbonate has been described as the most important compound of lithium. Lithium plating reduces the battery life. Reaction of fixatives of proteins.
In the 1970s Lithium carbonate was approved in the United States for the treatment of acute mania and bipolar disorder and it has been in use ever since. In fixation of tissues for routine histopathology the most important reactions are those which stabilize proteins. Battery producers combine carbonate or hydroxide with materials to form a cathode and an anode together forming an individual battery cell.
Lithium Carbonate is the carbonate salt of lithium a soft alkali metal with antimanic and hematopoietic activities. Antimanic drug Pregnancy Category D. Neo Lithium or the Company TSXV.
Lithium is a metal ion that has been used extensively in the treatment of manic-depressive disorder since the 1970s.
Right To Know Hazardous Material Information Labeling System Labels. Whitacre says that Lithium-ions efficiency comes at a price.

First Nonflammable Lithium Ion Battery Will Stop Your Smartphone Car And Plane From Exploding Extremetech
Stone is a chemical element with the symbol Li and atomic number 3.
How flammable is lithium. Gives off irritating or toxic fumes or gases in a fire. I am needing a storage container for used lithium batteries while they wait to be shipped out for recycling. March 8 2021 at 1020 am.
In the wrong setting lithium batteries can overheat and burst into flames. Volume 2A 2B 2C. Zinc-air batteries also contain no toxic compounds and are neither highly reactive nor flammable allowing them to be recycled and safely disposed of.
And if they touch for any reason. The onset of chemical breakdown is at a relatively low temperature and when the cathode breaks down prodigious amounts of energy are released. The difficulty with posting or shipping lithium batteries and their associated devices is that in certain conditions lithium batteries are extremely flammable.
Why would I. Trucking Transportation Supplies. The contents of the battery are under pressure so if a metal fragment punctures a partition that keeps the components separate or the battery is punctured the lithium reacts with water in the air vigorously generating high.
Custom Package Testing and Design DGOf. According to Merriam-Websters Dictionary of English Usage back in the 1920s the National Fire Protection Association urged people to start using the word flammable rather than inflammable which was the original word because they were concerned some people might think inflammable meant not-flammable or nonflammable. However lithium batteries also contain a flammable electrolyte that can cause small scale battery fires.
Nanotech Energy is another company pursuing lithium across Northern Nevada. Risk of fire and explosion on contact with combustible substances and water. These electrolytes also mustnt be exposed to air.
Oil Only Absorbent Mats and Pads. Class D fires involve combustible metals - especially alkali metals like lithium and potassium alkaline earth metals such as magnesium and group 4 elements such as titanium and zirconium. The lithium-water reaction at normal temperatures is brisk but nonviolent because the hydrogen produced does not ignite on its own.
Effects of exposure to Lithium. 343 Flammable and Combustible Liquids Hazard Class 3 3431 Definition. NFPA National Fire Protection association Labels.
Unlike standard alkaline batteries most lithium batteries manufactured today contain a flammable electrolyte and have an incredibly high energy density. Combustible Liquid means any liquid that does not meet the. The colourless solution is highly alkalic.
GHS Globally Harmonized System of Classification and Labelling of Chemicals. Tiny metal fragments float in the liquid. They can overheat and ignite under certain conditions such as a short circuit or improper design or assembly.
Lithium is highly flammable and will even react spontaneously with water. Metal fires represent a unique hazard because people are often. The exothermal reactions lasts longer than the reaction of sodium and water which is directly below lithium in the periodic chart.
This high reactivity is why some lithium-ion batteries ignite or explode when exposed to high temperatures Instead lithium is usually extracted from lithium minerals that can be found in igneous rocks chiefly spodumene and from. For that reason alternative cathode materials have been developed. Lithium-ion battery fire hazards are associated with the high energy densities coupled with the flammable organic electrolyte.
A new massive lithium ion battery plant is coming to the Reno-Tahoe area. At the Tahoe-Reno Industrial Center enormous bull. Symptoms may be delayed.
However there is always on-going research into developing Li-ion batteries that are less hazardous or that meet the requirements for new applications. Lithium cells and batteries can present both chemical eg corrosive or flammable electrolytes and electrical hazards. Lithium is never found in its elemental metallic form because it is highly reactive.
The short version is that the lithium-ion batteries used in mobile phones contain flammable chemicals that are usually separated within the battery structure. Many reactions may cause fire or explosion. Lithium-ion batteries are also more expensive to produce as.
If a lithium fire broke out in a truck or even a plane it could be a terrible disaster. Lithium Cobalt Oxide was the first material used for the cathodes in Lithium secondary cells but safety concerns were raised for two reasons. Battery and Lithium Battery Related Labels and Placards.
It should be noted that this has not happened to large scale lithium batteries. Its the only mainstream battery chemistry that uses a flammable substance as an electrolyte so while its more efficient than. However while zinc is one of the most abundant metals on Earth using it at scale as an alternative to lithium could pose problems in the future.
Pattys Industrial Hygiene and Toxicology. The diagram below shows the breakdown. Are flammable there can be some safety issues with this battery.
The specifics depend on the battery but lithium-ion batteries usually contain a metal coil and a flammable lithium-ion fluid. Lithium reacts intensely with water forming lithium hydroxide and highly flammable hydrogen. The US system includes flammable gases in their Class B.
They will be place in the container on pallets. Lithium-ion batteries power most portable electronic devices today. 2 thoughts on Lithium Ion Battery Storage Requirements Holly says.
Liquid electrolytes found in lithium-ion batteries are extremely volatile and highly flammable. Lithium Battery Shipping Kits. Additionally these protection circuits can sometimes be used to prevent over-charging.
So while lithium-ion batteries are subject to events like thermal runaway leading to explosion and fire the solid electrolytes used in solid-state batteries. Batteries commonly contain materials such as lithium cobalt nickel manganese and titanium as well as graphite and a flammable electrolyte. Class B fires are those where the fuel is flammable or combustible liquid.
Article Corrosive Storage Flammable Storage Lithium Battery Storage Safety Tags Lithium Battery Storage. LiHe a very weakly interacting van der Waals compound has been detected at very low temperatures. This creates new challenges for use storage and handling.
Solid-state batteries contain no liquid parts and will not have this volatile component. Lithium aluminum hydride has the additional fire hazard in that it is used in the flammable solvents diethyl ether and tetrahydrofuran. Tank Markings - Signs.
Whats more lithium fires are difficult to extinguish. It is a soft silvery-white alkali metalUnder standard conditions it is the lightest metal and the lightest solid elementLike all alkali metals lithium is highly reactive and flammable and must be stored in vacuum inert atmosphere or inert liquid such as. Services Custom Labeling Services.
Lithium-ion batteries have in-built protections to prevent overheating and to prevent the complete discharge of the battery which can also be damaging. Lithium is flammable and it is potentially explosive when exposed to air and especially to water though less so than the other alkali metals. Pallet Protection Labels Cones.
Professor of chemistry at the University of Southern California Sri Narayan told the New York. The terms used in the standards that apply to Hazard Class 3 are defined as follows. Flammable Liquid means a liquid that has a flashpoint of not more than 140 F 60 C or any material in a liquid phase that has a flashpoint at or above 100 F 38 C.
Most Common Chemical. It was this that caused the infamous Samsung Note 7 smartphone combustions which forced Samsung to scrap production and lose 26bn in market value. View All Packaging.
Shipping Document Labels.
Under standard conditions it is the lightest metal and the lightest solid element. In locations such as Pastos Chicas near the ArgentinaChile.
/2-12033975b6304be093692315b1ca340a.jpg)
Why Lithium Batteries Catch Fire
Tesla has bet big on them and built a.

Lithium burning from contact with water. In Salar de Atacama mining activities consumed up to 65 of the regions water causing havoc for local farmers. The fear of accident hinders the fully acceptance of. Water may not extinguish burning batteries but will cool the adjacent batteries and control the spread of fire.
Lithium hydride is an inorganic compound with the formula Li HThis alkali metal hydride is a colorless solid although commercial samples are grey. Whats more lithium can be extracted from ocean water where reserves are practically unlimited enough to fulfill the worlds energy needs for 6 million years. Stone is a chemical element with the symbol Li and atomic number 3.
Tim Crowley a vice president at Lithium Americas said the company would operate responsibly planning for example to use the steam from. Risk of fire and explosion on contact with combustible substances and water. But theres one stumbling block and its huge.
Lithium-ion batteries have taken over the world. Global inventory for tritium. Lithium from proven easily extractable land-based resources would provide a stock sufficient to operate fusion power plants for more than 1000 years.
The TR and TR-induced smoke fire and even explosion are the most common features during the accidents of lithium ion battery. The Lithium content is combined into other compounds which do not react with water. Elsewhere in South America Argentinians in the Salar de Hombre Muerto natural salt pan have expressed concerns over the lithium mining in the region citing contamination to streams and the irrigation of crops.
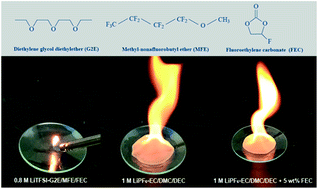
The burning reaction also tends to liberate the fluorine from the lithium salt typically LiPF 6 dissolved in the electrolyte. ADS CAS Article Google Scholar. Virtually all fires.
It is soluble and nonreactive with certain molten salts such as lithium fluoride lithium borohydride. Handbook of Reactive Chemical Hazards. According to the BLMs FEIS Lithium Americas plans to pump 17 million gallons of water from the aquifer each year which could leave less water to support the grasslands Bartells cattle graze.
It may be toxic by ingestion inhalation and skin absorption. Like all alkali metals lithium is highly reactive and flammable and is stored in mineral oil. It is used to make other chemicals.
Here are some lithium-ion battery safety tips to help businesses and their employees prevent workplace fires and injuries. Characteristic of a salt-like ionic hydride it has a high melting point and it is not soluble but reactive with all protic organic solvents. Wash skin with soap and water.
In the case of skin irritation or allergic reactions see a physician. Proponents of this technology note that hydrogen is more common than lithium by a long shot its literally the most abundant element in the universe and that it produces energy just as cleanly since the only end products of burning hydrogen are heat and pure water. Symptoms may be.
Lithium hydroxide solution appears as a clear to water-white liquid which may have a pungent odor. Sodium carbonate and sodium chloride are unsuitable to use as extinguishers for lithium fires since burning lithium will liberate the more reactive sodium in contact with them. After several months the water evaporates leaving a mixture of manganese potassium borax and lithium salts which is then filtered and placed into another evaporation pool.
E L Martín et al New constraints on the minimum mass for thermonuclear lithium burning in brown dwarfs Monthly Notices of the Royal Astronomical Society 2021. Lithium is a chemical element with atomic number 3 which means there are 3 protons and 3 electrons in the atomic structureThe chemical symbol for Lithium is Li. Many reactions may cause fire or explosion.
For every tonne of lithium produced 500000 gallons of water is used. Water immersion brought short circuit. If symptoms persist call a physician.
Of Power Sources 273 216222 2015. If symptoms persist call a physician. It is a soft silvery-white alkali metalUnder standard conditions it is the lightest metal and the lightest solid elementLike all alkali metals lithium is highly reactive and flammable and must be stored in vacuum inert atmosphere or inert liquid such as.
The amount of Lithium deposited during the Lithium plating when cells are damaged as described above is very small and not usually responsible for the fires which have occurred. The lithium extraction process uses a lot of waterapproximately 500000 gallons per metric ton of lithium. For most batteries the products typically consist of CO 2 and water vapor.
Lithium-Ion Battery Safety Tips for Employees. Butterworth-Heinemann Ltd 1990 p. Effects of exposure to Lithium.
The lithium plants which use vast amounts of water have exacerbated shortages. The most significant difference between lithium-ion and lithium-polymer batteries is the chemical electrolyte between their positive and negative electrodes. Furthermore many of the reported fires are due to burning electrolyte rather than the Lithium compounds.
Inhalation Move to fresh air. An experimental study on burning behaviors of 18650 lithium ion batteries using a cone calorimeter. Lithium production contributes to regional development and job creation in remote communities utilizes brackish non-potable water engages local communities with social programs generates material in-province tax revenue and facilitates the availability of electricity in remote and unconnected communities.
Contact may cause severe irritation to skin eyes and mucous membranes. An EV police car caught fire on the street. Always follow local state and federal regulations on proper battery disposal.
In case of fire where lithium batteries are present flood area with water or smother with a Class D fire extinguishant appropriate for lithium metal such as Lith-X. Eye Contact Rinse thoroughly with plenty of water also under the eyelids. The electrification of transportation is expected to cause a material reduction of.
Gives off irritating or toxic fumes or gases in a fire. To extract lithium miners drill a hole in salt flats and pump salty mineral-rich brine to the surface. Skin Contact Remove contaminated clothing and shoes.
Burning batteries will burn themselves out. Smoke fire and explosion are serious safety problems that arouse concerns from the public. Thacker Passs lithium is concentrated in a soft sedimentary clay which Lithium Americas intends to process with an experimental technique that involves burning hundreds of tons of sulfur.
Well burning hydrogen only produces water as a byproduct its exceptionally efficient and its much cleaner than lithium when it comes to producing it and recycling it at the end of the cars life. Workplace injuries from lithium-ion batteries are preventable with continual employee education. For the burning scenario the electrolyte burns efficiently producing primarily carbon dioxide CO 2 and water H 2 O as the by-products.
It is a soft silvery-white alkali metal. Right now we simply cannot manufacture enough. Most lithium-ion batteries unlike more traditional ones also include an electronic controller which regulates power and discharge flows so your battery doesnt overheat or explode.