It reacts explosively with bromine carbon disulfide and chromyl chloride. The azide groups of BA also reacted with and crosslinked the alkyl side chains on polymer semiconductors through azideCH insertion 28 Supplementary Fig.

2 Nan3 Na 3 N2 Not Balanced Ppt Download
For Research Use Only.
Molecular mass of sodium azide. Show your calculation including proper units. If physical findings persist for 30 minutes after antidote administration sodium nitrite and sodium thiosulfate may be readministered at half their original respective doses. NaN 3 300C 2Na 3N 2 NH 4 2 Cr 2 O 7 N 2 Cr 2 O 3 4H 2 O.
For more information please contact technical support. Calibration curves were linear for 05 to 20 ugmL sodium azide and the detection limit was 005 ugmL. However methemoglobin levels should be.
Sodium is a chemical element with the symbol Na from Latin natrium and atomic number 11. A Laboratory Manual Third Edition 1982. Full PDF Package Download Full PDF Package.
Used for making gasoline additives electric power cable sodium lamps other chemicals. Western blotting is an important technique used in cell and molecular biology. Pierre Ronco Paris France KI is one of the most cited journals in nephrology and widely regarded as the worlds premier journal on the development and consequences of kidney disease.
In order to make 100 mL of a 17 sodium azide solution you would need to weigh out 17 grams of sodium azide and then add water until the final volume is 100 mL. Sodium is now produced commercially through the electrolysis of molten sodium chloride. RCOCl NaN 3 Sodium azide RCON 3 Acyl azide NaCl.
Determination was performed by isocratic ion chromatography using suppressed conductivity detection. Burns violently with explosions that may spatter the material. It forms explosive compounds with phosgene brass zinc trifluoroacrylol fluoride and nitrogen-diluted bromine vapor.
High salt ionic strength Related Products Pierce IgG Elution Buffer Pierce Gentle AgAb Elution Buffer pH 66. We next used 96-well microtiter plates to prepared an azide library from 1224 different amines most of which have a molecular mass of less than 300 Da. Not for use.
A Laboratory Manual Third Edition. They should be used with standard staining buffers which contain PBS or HBSS protein such as BSA or FBS and sodium azide. Determine the mass of Tris base to weigh by multiplying the number of moles by the molecular weight 12114 gmol of Tris.
It may decompose explosively with shock concussion or friction. A Laboratory Manual Third Edition. We are a leading supplier to the global Life Science industry with solutions and services for research biotechnology development and production and pharmaceutical drug therapy development and production.
RCOOC 2 H 5 RCONHNH 2 Acyl hydrazide RCON 3 2H 2 O. Moles needed gmol g 3 Dissolve Tris Base in Water. Sodium is a chemical element with.
Relative atomic mass A r. Both reactions must only be carried out under controlled conditions by a professional. Meta-Analysis Addressing the Effect of Sodium-Glucose Cotransporter 2 Inhibitors on Flow-Mediated Dilation in Patients With Type 2 Diabetes Mellitus.
Employed only in rather specialized applications only about 100000 tonnes of metallic sodium are produced annually. 1400643 1400728 see notes g r. A short summary of this paper.
Contains 002 sodium azide IgG Elution Buffer. Shipped as a solid or molten liquid. No other additives should be used.
We would like to show you a description here but the site wont allow us. Amine-based Gentle AgAb Binding Buffer. Its only stable isotope is 23 Na.
The classical claisen rearrangement is the first and slow step of the isomerization of allyl aryl ethers to ortho allylated phenols. The C 2 N 14 molecule is a monocyclic tetrazole with three azide groups with a molecular weight of 22016 gmol 1. Sodium is the sixth most abundant element in the Earths crust and exists.
Acyl azide required for the reaction is obtained as follows. The ideal gas equation uses absolute pressure. The technique uses three elements to accomplish this task.
Assume that the temperature of the gas is 250 o C. KI is peer-reviewed and publishes original research in both basic science and. Contains 005 sodium azide Gentle AgAb Elution Buffer.
The free metal does not occur in nature and must be prepared from compounds. You can make use of this equation in another way. The normalized intensity of the deglycosylated peptide resulting from on-chip deglycosylation with the PNGase H and PNGase Dj IMERs is shown after 1 30 and 50 days of storage in 20 mM glycine 01 sodium azide and 10 glycerol pH 25 pH 35 for PNGase Dj IMERs at 4 C.
Sodium appears as a silvery soft metal that becomes grayish white upon exposure to air. 4 Adjust the pH Using a pH meter titrate the solution of Tris with 1M hydrochloric. Sodium azide in a sample was acidified and the azide was converted to the volatile hydrazoic acid which was trapped in 25 mM sodium hydroxide solution.
However the decomposition of sodium azide is one route to N 2 and decomposition is ammonium dichromate is another. Calculate the mass in grams of sodium azide required to generate enough nitrogen gas to fill the airbag at the pressure you calculated in part b. UltraComp eBeads are compatible with all fluorochromes excited by an ultraviolet 355 nm or violet 405 nm laser.
Under the editorial leadership of Dr. Protons and Neutrons in Sodium. The created covalent bonding.
By using a western blot researchers are able to identify specific proteins from a complex mixture of proteins extracted from cells. It is a soft silvery-white highly reactive metal. Recoveries of sodium azide from.
Reacts with copper lead silver mercury. Gauge pressure atmospheric. Metallic sodium is used mainly for the production of sodium borohydride sodium azide indigo and triphenylphosphine.
All measurements were performed in duplicate and the standard. Protein G IgG Binding Buffer. 36 Full PDFs related to this paper.
The answer you determined in part b is gauge pressure. For example a small mass 100 g of NaN 3 will generate approximately 50 L of N 2. Kidney International KI is the official journal of the International Society of Nephrology.
For example when each sodium atom in a sample of sodium metal group 1 gives up one electron to form a sodium cation Na and each chlorine atom in a sample of chlorine gas group 17 accepts one electron to form a chloride anion Cl the resulting compound NaCl is composed of sodium ions and chloride ions in the ratio of one Na ion for each Cl ion. 1 separation by size 2 transfer to a solid support and 3 marking target protein using a proper primary and secondary. Sodium azide may explode when heated above its melting point especially if heated rapidly.
Among many engineering considerations the amount of sodium azide used must be appropriate for generating enough nitrogen gas to fully inflate the air bag and ensure its proper function. Next administer 075 mL per pound body weight of 25 sodium thiosulfate 165 mL per kilogram body weight of 25 sodium thiosulfate intravenously over a period of 10 minutes. It has a molecular equilibrium between a closed and an open form isocyanogen tetraazide which has been known since 1961 the latter being quickly cyclized to the cyclic tetrazole form C 2 N 14 at room temperature.
Sodium is an alkali metal being in group 1 of the periodic table. Dissolve the required mass of Tris into a volume of deionized water approximately 13 of the desired volume of buffer to be made. Nitrogen is made on massive scale.
Incidence of Myopericarditis and Myocardial Injury in Coronavirus Disease 2019 Vaccinated Subjects.
M Thermal decomposition of the fluorocarbon chain in air leads to the formation of oxidized products containing carbon fluorine and oxygen. Discard the sample and start over if any are seen.
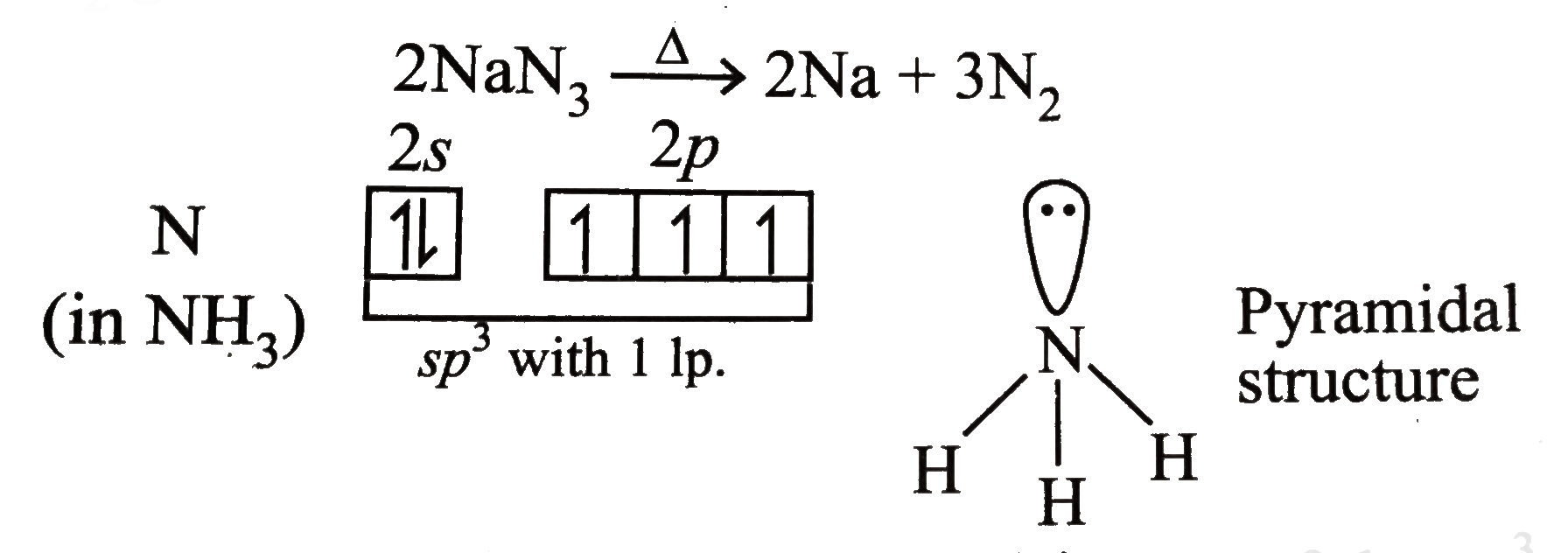
A Write The Reaction Of The Thermal Decomposition Of Sodium
Conditions to Avoid Incompatible products.

Decomposition of sodium azide. How many grams of sodium azide are required to produce 370 L of nitrogen gas at 105 atm and 270 degrees Celsius. Sodium azide Revision Date 25-Apr-2019 Reactive Hazard Yes Stability Risk of explosion by shock friction fire or other sources of ignition. Calibration curves were linear for 05 to 20 ugmL sodium azide and the detection limit was 005 ugmL.
Check for air bubbles. NaN 3 300C 2Na 3N 2 NH 4 2 Cr 2 O 7 N 2 Cr 2 O 3 4H 2 O. Both reactions must only be carried out under controlled conditions by a professional.
Sodium appears as a silvery soft metal that becomes grayish white upon exposure to air. It may also be produced by the thermal decomposition of sodium azide. Sodium azide in a sample was acidified and the azide was converted to the volatile hydrazoic acid which was trapped in 25 mM sodium hydroxide solution.
An index of exposure to these products is possible through their alkaline hydrolysis followed by a quantitative determination of fluoride content. This requirement is satisfied in many automotive airbag systems through use of explosive chemical reactions one common choice being the decomposition of sodium azide NaN 3. May dissolve in water but it.
Google Scholar Hamza RZ. Iodobenzene Dichloride in Combination with Sodium Azide for the Effective Synthesis of Carbamoyl Azides from Aldehydes X-Q. When sensors in the vehicle detect a collision an electrical current is passed through a carefully measured amount of NaN 3 to initiate its decomposition.
Sodium azide released during this process may contaminate the steel iron and nonferrous metals recovered at this stage. Zhang Synthesis 2008 2589-2593. Sodium azide is the inorganic compound with the formula NaN 3This colorless salt is the gas-forming component in legacy citation needed car airbag systems.
The thermal behavior observed for the decomposition of SeChitosan confirmed the products stability up to temperatures of. Used for making gasoline additives electric power cable sodium lamps other chemicals. Protein concentration 110 mgml.
Finally inorganic chemistry is a subject that should not be approached with any degree. Alkane sulfonate X OSO2R alkyl azide X N3 hemiketal hemiacetal ketal acetal dithiane hydrazone oxime geminal dihalide enol ether enamine ester orthoester nitrile ketene trihalomethyl hydroxamic acid carbamate alkyl haloformate xanthate isocyanate carbodiimide aminal thioester amide urea Summary of Reagents for Oxidative Functional Group Interconversions. Heat flames and sparks.
However the decomposition of sodium azide is one route to N 2 and decomposition is ammonium dichromate is another. Hence there is a risk of ignition when airbags pass through the automobile shredder. Substance that dissociates into ions when dissolved in water.
Of greater concern however is the large amounts of heat and friction generated by the shredder. 아자이드화 나트륨은 중요한 의약 중간체로 많은 약품 합성에 사용되며 또한 자동혈구계산기계 방부 살균 자동차 에어백 및. Reaction with water to the unstable carbamic acid derivative which will undergo spontaneous decarboxylation.
If oxygen is present a brownish-orange cloud of precipitate or floc will appear. Recall that NaN 3 reacts explosively at high temperatures. Sodium azide is made industrially by the reaction of nitrous oxide N 2 O with sodium amide in liquid ammonia as solvent.
Nitrogen is made on massive scale by liquefaction of air and fractional distillation of the resulting liquid air to. Determination was performed by isocratic ion chromatography using suppressed conductivity detection. What is particularly amazing is.
Stopper the bottle with care to be sure no air is introduced. Mix the sample by inverting several times. For example lead azide used in detonators may be prepared from the metathesis reaction between lead nitrate and sodium azide.
Sodium was first commercially produced by thermal reduction of sodium carbonate with carbon at 1100 degrees Celcius in the Deville process. The molecular weight of this compound is _____ amu. Radical Azidonation of Aldehydes L.
The heat from this ignition starts the decomposition of the sodium azide and the generation of nitrogen gas to fill the air bag. No particular concentration limit is specified pending evaluation of the toxicity of the products but. We would like to show you a description here but the site wont allow us.
Dissolve 10 g sodium azide NaN3 in 40 ml water and add to alkali- iodide solution The resultant reagent should not contain free iodine check through diluting and acidifying and adding starch indicator and observe for blue color Concentrated sulfuric acid Aqueous solution of starch indicator. Salt concentration 100200 mM. Many inorganic azides can be prepared directly or indirectly from sodium azide.
The formula weight of ammonium sulfate NH42SO4 rounded to the nearest integer is _____ amu. 50 glycerol does not freeze at 20C Alternatively store at 70C. Imine organoboranes RCH2BR2.
For instance if you had a 800 g sample of a compound that was 200 g element X and 600 g element y then the percent composition of each element would be. 아자이드화 나트륨은 화학식이 NaN 3 인 무기화합물이다. Incompatible Materials Acids Oxidizing agents Peroxides Acid chlorides Metals Hazardous Decomposition ProductsNitrogen oxides NOx.
2017 91 602 610. Cite this Article Format. Burns violently with explosions that may spatter the material.
Percent composition in chemistry typically refers to the percent each element is of the compounds total mass. Recoveries of sodium azide from. Pure sodium may be obtained by electrolysis of molten sodium chloride.
When this floc has settle. Avoid shock and friction. An alternative is to use 03 hydrogen peroxide in methanol for 20-30 minutes since methanol accelerates the destruction of the heme groups so a lower concentration of hydrogen peroxide can be.
Ameliorative effect of vitamin E and selenium against oxidative stress induced by sodium azide in liver kidney testis and heart of male mice. Add 2 mL of alkali-iodide-azide reagent in the same manner. It is an ionic substance is highly soluble in water and is very acutely poisonous.
The balanced equation for the decomposition of sodium azide is _____ 12311. Shipped as a solid or molten liquid. Chlorine is produced at the positive electrode anode while hydrogen H 2 and sodium hydroxide are produced at the negative electrode cathodeThese three materials are feedstocks for the production of bleach sodium hypochlorite NaOCl and a variety of other products including soda ash Na 2 CO 3.
Storage at 20C in PBS in 50 glycerol is usually safe over long periods. Storage at 4C with 10 mM sodium azide or 0005 merthiolate is usually safe over long periods but occasionally proteolytic breakdown occurs. NaN3 arrow Na.
This danger is amplified if. Sodium azide decomposes into solid sodium metal and nitrogen gas. The basic equation mass of element mass of compound X 100.
Dissolve 2 grams of soluble starch and 02 grams of salicylic acid in 100 ml of hot. It is used for the preparation of other azide compounds. Immerse slides in fresh made 03 hydrogen peroxide in 01 sodium azide for 10-15 minutes to make the blocking solution add 5ml of 3 hydrogen peroxide to 45 ml of 01 sodium azide and mix well.
N 2 O 2 NaNH 2 NaN 3 NaOH NH 3. The formula of nitrobenzene is C6H5NO2.