Potassium hydroxide residue in a catalyst pot reacted violently when acetic acid was added MCA Case History 920. It is abundant multivalent and nonmetallicUnder normal conditions sulfur atoms form cyclic octatomic molecules with a chemical formula S 8Elemental sulfur is a bright yellow crystalline solid at room temperature.
Peracetic acid is a colorless liquid with a strong pungent acrid odor.
Molecular weight of glacial acetic acid. Ethyl acetate ethyl ethanoate is an ester. Glacial acetate acidaniline 731 at room temperature at dark for 2 hours to induce the site-specific cleavage. Mobile phase acetonitrile and buffer solution.
2 Qualitative and Quantitative Composition. Results show a higher recovery for pure glacial acetic acid samples 1143 than its corresponding 50 glacial acetic acid samples 9601. In simple words 1 mole is equal to the atomic weight of the substance.
Apart from the number of double bonds present in the molecule the IV is also influenced by the molecular weight thus IV oleic acid 2 127282 100 901 IV glyceryl 6 12741 3 281 100 862 triolein similarly IV linoleic acid 181 trilinolein 1732 IV linolenic acid 2735 trilinolenin 2616. Sulfur is the tenth most common element by mass in the universe. 1 filter to remove any particulate matter and store in appropriate bottles Destaining solution.
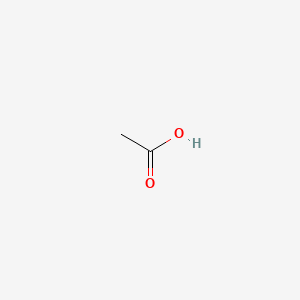
This structure is also available as a 2d Mol file or as a computed 3d SD file The 3d structure may be viewed using Java or Javascript. Acetic Acid CH3COOH- Acetic Acid is an organic compound with formula CH3COOHVinegar is a water solution of acetic acid containing 5-8 of acetic acid by volume. During these processes detonating mixtures may be produced.
The recovery of the method was higher than other spectrophotometric methods reported previously 15 18. And as a polymerization catalyst for. 4 wv Paraformaldehyde-1 vv glutaraldehyde.
After precipitation the NaBH 4-treated RNAs were subsequently treated with aniline-acetate solution H 2 O. APO-Tranexamic Acid tablets contain the active ingredient tranexamic acid. The standard composition is 01 of dye and 5 of acetic acid Biotium Novus biologicals and Sigma-Aldrich.
For example 1 mole of acetic acid is equivalent to 6005 g of acetic acid molecular weight. Molecular weight 1X molarity Add for 500ml Add for 1L. Molecular weight of H2SO4.
LIQUID AND MIST CAUSE SEVERE BURNS TO ALL BODY TISSUE. 0874 M 5 25 ml. Hazards Identification Emergency Overview----- POISON.
Tranexamic acid is a white crystalline powder that is odourless or almost odourless. Concentration of Sulfuric acid. Addition of a small amount of water.
Pure compound is a solid below 62F. Add glacial acetic acid to pH 55. When properly mixed filter the solution through a Whatman No.
Molarity refers to the number of moles of the solute present in 1 liter of solution. 95 by mass wtwt Step 1. The difference between them is the melting point of their.
For example 1 mole of H2SO4 is equal to 9808 grams of H2SO4 molecular weight 9808. Visit BYJUS for more content. Density weight volume or Volume weight density or The.
As an oxidant for preparing epoxy compounds. It has a pungent smell and a sour taste. Calculate the volume of 100 grams of Sulfuric acid.
Where several fatty acids are present the IV. More information on the manner in which spectra in this collection were collected can be found here. Other examples of esters include ethyl propanoate propyl methanoate propyl ethanoate and methyl butanoate.
Methacrylic acid appears as a clear colorless liquid or low-melting solid with a pungent odor. Acetic acid glacial 6005. Glacial acetic acid reagent grade.
Often used in an aqueous solution. Add 90 ml methanolwater 11 vv and 10ml of Glacial acetic acid mix properly using a magnetic stirrer. QTBSBXVTEAMEQO-UHFFFAOYSA-N Copy CAS Registry Number.
MAY BE FATAL IF SWALLOWED. 571 ml Glacial Acetic Acid 1861g Na2EDTA2H2O QS to 10 liter with water - do not adjust pH but check. The recovery studies were carried out in both pure glacial acetic acid and 50 glacial acetic acid.
Sampling medium 450 µgmL a For sampling in ambient temperatures 60 F. May polymerize exothermically if heated or contaminated. As a reagent in making caprolactam and glycerol.
To Learn about the structure of Acetic acid its preparations chemical physical properties uses and FAQs. 76057 4 Na salt 13 mM 01 05 g. Acetic acid ə ˈ s iː t ɪ k systematically named ethanoic acid ˌ ɛ θ ə ˈ n oʊ ɪ k is an acidic colourless liquid and organic compound with the chemical formula CH 3 COOH also written as CH 3 CO 2 H C 2 H 4 O 2 or HC 2 H 3 O 2.
Chat now for more business. Sulfur in nontechnical British English. The density of glacial acetic acid is 1049 gml at 25C which means that the weight of the 1 ml of glacial acetic acid is 1049 grams at 25C.
Sulphur is a chemical element with the symbol S and atomic number 16. If the polymerization takes place inside a container the container may rupture violently. Contact China Manufactory Zouping TaiZi Welfare Vinegar Company for the product Food grade glacial acid acetic 997 200KG Shunxin brand.
There are several formulations available with a difference in the percentage of acetic acid and dye. Small molecules such as amino acids. Except where noted spectra from this collection were measured on dispersive instruments often in carefully selected solvents and hence may differ in detail from measurements on FTIR instruments or in other chemical environments.
Corrosive to metals and tissue. CH 3 COOH Glacial 100 175 Molar Strength 100 Density 105 Molecular Weight 6005 1 liter 1050 gm CH 3 COOH 1748 moles 175M. During the production of terephthalic acid n-xylene is oxidized in the presence of acetic acid.
Acetic acid aqueous Ethanoic acid Glacial acetic acid Methanecarboxylic acid Note. The hydrogen on the carboxyl group of acetic acid is replaced with an ethyl group. Most proteins peptides and enzymes of low molecular weight.
Vinegar is no less than 4 acetic acid by volume making acetic acid the main component of vinegar apart from water and other trace elements. 4 wv Paraformaldehyde 4 wv Paraformaldehyde-1 vv glutaraldehyde 10 Neutral-buffered formalin NBF Delicate tissue. Zenkers solution Helly.
Acetic acid or acetic anhydride can explode with nitric acid if not kept cold. Concentration information is not available for. Glycerides are fatty acid esters of glycerol.
After cleavage the RNA samples were purified by ethanol precipitation and used for cDNA library construction using NEBNext Small RNA Library Prep Set New England Biolabs. Can be found in concentrations of 5-8 in vinegar Colorless liquid or crystals with a sour vinegar-like odor. Can use stock EDTA to make buffer will need to check and adjust pH depending on stock 100 ml of 05 M EDTA at ph 80 1X40 mM Tris pH 76-80 20 mM acetic acid 1 mM EDTA 10X TBE Stock 1080 g Tris Base.
It is freely soluble in water. GAA - Glacial acetic acid GFP - Green fluorescent protein GUS - -Glucuronidase HEPA - High Efficiency Particulate Air HEPES - N-2-hydroxyethylpiperazine-N-2-ethanesulphonic acid H2O2 - Hydrogen peroxide HCl - Hydrochloric acid HPLC - High performance liquid chromatography h - Hours IR - Infra red IPTG - Isopropyl-thiogalactoside kDa - kilodaltons kbp - kilobase pairs Km - Michaelis-Menton. As a bleaching agent.
Each APO-Tranexamic Acid tablet contained 500 mg of tranexamic acid. Acetic Acid 64-19-7 995 - 100 Yes 3. Dissolve 204 g sodium acetate trihydrate in 2 L distilled-deionized water.
Weigh 025g of Coomassie Brilliant Blue R250 in a beaker. Used as a bactericide and fungicide especially in food processing. Liver spleen bone marrow.
Density of Sulfuric acid. Fats and oils are examples of esters.