Hydrochloric acid is prepared by dissolving gaseous hydrogen chloride in water. Boric acid is determined by ashing tissues in an alkaline medium at 600 C dissolving in hydrochloric acid centrifuging mixing part of the supernatant with carminic acid in sulfuric acid and measuring the color at 575 nm after 1 hr.

Coke Cans In Acid And Base Periodic Table Of Videos Youtube
John Wiley and Sons 1991-Present p.
Sulfuric acid dissolving metal. How can sodium hypochlorite be produced. Citric acid is a weak acid found in citrus fruits and used as a natural preservative and to impart a sour flavoring. Precipitation of AgCl.
At 145C the stream of steam is increased and during 25-3 hours the phenylacetone is steam. Purpose of Electroplating. Because of the corrosive nature of the acid ceramic glass or sometimes tantalum apparatus is commonly used.
The solution is electrolyzed and forms a sodium hypochlorite. Filter with vacuum assist and let vacuo suck as dry as possible. The dissolved metal ions are reduced at the cathode plating the metal onto the item.
Hydrochloric acid is usually marketed as a solution containing 2835 percent by weight hydrogen chloride commonly known as concentrated hydrochloric acid. Lysergic acid and potassium sulphate will be seen to precipitate. Reacting copperII oxide with sulfuric acid.
1025g 75 sulfuric acid was mixed with 1g ZnCl 2 and 192g 116 mol Ephedrine or Pseudoephedrine freebase was dissolved at a temperature of 50-100C and the reaction mixture was heated further to 145-150C. The acid creates a layer on the steel and this layer prevents the acid from dissolving the metal. Less harmful gasses are produced when sulfuric acid is used.
To the filtrate add a solution of 25 g 12-tungstosilicic acid in 20 cm 3 water slowly with stirring. Nitric acid HNO 3 is a strong acid because when it is dissolved in an aqueous solution it completely dissociates into H and NO 3 ions in the solution. Carbonic acid is a weak acid.
HNO3 H2O -. Sodium hypochlorite can be produced in two ways. At 125C steam is passed through the solution to facilitate mixing of the contents.
Includes kit list and safety instructions. As on dissolving HNO 3 in an aqueous solution no hydrogen remains bound all are dissociated and converted into H ions which means the concentration of hydrogen ion increased in the solution. In association with Nuffield Foundation.
Hydrofluoric acid is actually a weak non-oxidising acid and in the good old days it was often used to dissolve mud and sand that had clogged-up the cooling water passages of marine. 93 wt sulfuric acid has a freezing point below zero at -21ᵒF so many industrial applications can utilize carbon steel tanks with 93 wt H 2 SO 4 uninsulated but 98 wt freezes at 30ᵒF and so much more care is needed for successful storage. Atoms are represented as spheres and are colour-coded.
Although nearly 100 sulfuric acid solutions can be made the subsequent loss of SO 3 at the boiling point brings the concentration to 983 acid. Stir the solution for a few minutes and filter off any precipitate. Laboratory chemicals Synthesis of substances 13 Details of the supplier of the safety data sheet Company.
For example concentrated sulfuric acid often referred to as Oleum is normally transported in steel tanker wagons. Let stand for 2-3 hours in the 5-10 cooling mixture. It is used to.
The remaining liquid is known as raffinate a waste product. LOUIS MO 63103 UNITED STATES. Simply the acids concentration becomes diluted.
Make a solution from 150 ml of liquid ammonia and 25 liters. In soils these acids can cause the release of natural but toxic elements like aluminum. It is used in the dye industry as a mordant and as a reducing agent.
Sulfuric acid is a strong acid that strongly reacts with bases and that is very corrosive. HClg in water produces hydrochloric acid which is a solution of hydronium and chloride ions. And when these compounds react with water vapor in the atmosphere they turn into sulfuric acid and nitric acid and then return to the surface as acid rain.
The freezing point of different sulfuric acid concentrations can vary markedly. FeCl 2 4H 2 O by dissolving metallic iron in hydrochloric acid. However there is an acid base reaction that Nitric acid is constantly undergoing which is.
This is the only acid excreted by the lungs as a gas. Ferric chloride is generally prepared from ferrous chloride through the action of chloride or nitric acid. B the appearance of a substance changes but its identity does not.
Dissolve 05 g of ferrocene in 10 cm 3 concentrated sulfuric acid. Now Is HNO 3 Nitric acid strong or weak. 3050 SPRUCE ST ST.
Direct current is supplied to the anode oxidizing its metal atoms and dissolving them in the electrolyte solution. - By dissolving salt in softened water which results in a concentrated brine solution. Sulfuric acid can be obtained by dissolving sulfur trioxide in water.
Allow the solution to stand for at least half an hour then pour it into 150 cm 3 distilled water. With a little dilute sulfuric acid. Answer 1 of 11.
The current through the circuit is such that the rate at which the anode is dissolved is equal to the rate at which the cathode is plated. Solutions of hydrocyanic acid and potassium nitrite are mixed. NO 2 1 nitrite ion.
Aqua regia dissolves gold although neither constituent acid will do so aloneNitric acid is a powerful oxidizer which will actually dissolve a virtually undetectable amount of gold forming gold ions Au 3The hydrochloric acid provides a ready supply of chloride ions Cl which react with the gold ions to produce tetrachloroaurateIII anions also in solution. It is responsible for dissolving limestone to produce geological features such as stalagmites and stalactites. Physical properties Grades of sulfuric acid.
H 2 SO 4aq HSO 4-aq H aq The second dissociation constant is much smaller than the first so the reaction in which HSO 4-loses a proton H does not go to completion the ions are in equilibrium with the undissociated acid molecules. Ferric sulfate is produced on a large scale by adding sulfuric acid and an oxidizing agent eg nitric acid or hydrogen peroxide. Between the sulfuric acid concentrations of 35 - 75 wt freezing is.
H ions are attracted to the cathode gain electrons. Sulfuric acid Product Number. Illustrate the reaction of an insoluble metal oxide with a dilute acid to produce crystals of a soluble salt in this class practical.
Break up the filter cake and put in a 2 liter beaker. In water they can poison aquatic wild life and on land the acidity can cause animals eggs to not hatch and plants to lose nutrients. Sulfuric acid is a polyprotic strong acid The first dissociation constant is very large so that we assume this reaction goes to completion.
We identify HCN as the reacting acid K 1 as a spectator ion and the NO 2 1 ion as. 7664-93-9 12 Relevant identified uses of the substance or mixture and uses advised against Identified uses. Neutralize the mixture by adding cold dilute sulfuric acid to a congo red end point pH 42.
Anhydrous liquid hydrogen chloride is. There are several reasons. And form hydrogen gas.
Another process called electrowinning uses electricity to extract copper out of the PLS onto thin metal sheets. V2 289 1992 Hazardous Substances Data Bank HSDB Made commercially from aluminous materials such as bauxite. Then the copper-bearing liquid is combined with another acid to precipitate the copper from the organic material.
The 983 grade is more stable in storage and is the usual form of what is described as concentrated sulfuric acid. Now things have. Nonahydrate Kirk-Othmer Encyclopedia of Chemical Technology.
If water is acidified. Metal Ions as Lewis Acids. Raffinate can contain concentrated amounts of TENORM.
Prepared by dissolving aluminum or aluminum hydroxide in dilute nitric acid and crystallizing the product from the resulting aqueous solution. OH-ions are attracted to the anode lose electrons and form. B crushing the sugar cube and dissolving it in water c dehydrating the sugar cube with sulfuric acid d chewing the sugar cube and digesting it 9 A physical change occurs when a both the appearance of a substance and its identity change.
It is corrosive to metals and tissue. Sulfuric acid is a strong dehydrating agent that can strip available water molecules and water components from sugar and organic tissue.

Danger Sulphuric Acid Signs From Key Signs Uk
Sulfuric acid has to be disposed as a hazardous waste in the appropriate acidic waste container kept closed at all times.
Is sulfuric acid flammable. Sulfuric acid is a corrosive causing burns to body tissues. Substance Substance name. Non-flammable but reacts violently with water generating large amounts of heat with potential for spattering of the acid.
Any concentration of sulfuric acid other than 100 pure H 2 SO 4 will react with certain metals to produce flammable and potentially explosive hydrogen gas. It is soluble in water with release of heat. Long term exposure to low concentrations or short term exposure to high concentrations can result in adverse health effects from inhalation.
Reacts with sulfides carbides borides and. Flammable liquids flashpoint. All alcohols acetone acetaldehyde.
Strong inorganic mists containing sulfuric acid American Conference for Governmental Industrial Hygienists ACGIH. Low maintenance batteries like AGM batteries are the exception because they have the ability to compensate for water loss. Concentrated sulfuric acid 93 is added to tricalcium phosphate rock.
Sulfuric Acid Chemical Name. 7664-93-9 Product code. Sulfuric Acid 93 CAS.
Can react with combustible materials to generate heat and ignition. Highly reactive with most substances these acids must be double contained ie the primary container must be kept inside a non-corrosive canister tray or tub. It is corrosive to metals and tissue.
Concentrated sulfuric acid is also a strong oxidizing agent incompatible with flammable organic chemicals. Section 8 - Exposure Controls Personal Protection Engineering Controls. Greater than 95 recovery of sulfuric acid and phosphoric acid.
Reacts exothermically with carbonates including limestone and building materials containing limestone and hydrogen carbonates to generate carbon dioxide. It is a colorless odorless and viscous liquid that is miscible with water. Lead is toxic and some lead compounds are listed as Carcinogenic.
13 02282018 EN English Page 1 SECTION 1. The remaining injuries were mostly due to lifting or dropping batteries as they are quite heavy. Sulfuric acid dehydrates whatever it touches and the heat caused by that reaction with water can create secondary thermal damage.
This is typically found in nature as the substance fluorapatite. Reacts exothermically with organic bases amines amides and inorganic bases oxides and hydroxides of metals. Sulfuric nitric chromic perchloric Store separately from organic acids.
Sulfuric Acid ACS CAS-No. Sulfuric Acid Perchloric Acid Phosphoric Acid Hydrochloric Acid Chromic Acid Hydrofluoric Acid 68 and Sulfuric acid 93 in a secondary container Do not store acids on metal Flammable Liquids Flammable Solids Bases Organic Acids Cyanides Sulphides PoisonsToxins Heat Gas Generation Violent Reaction DO NOT POUR WATER INTO ACID Perchloric acid vapor can form explosive compounds. The electrolyte consists of water and sulfuric acid.
Sulfuric acid is a colorless oily liquid. Health Hazards - Acute and chronic. These functional groups typically react with H 2SO 4 to form new compounds.
Pure sulfuric acid does not exist naturally on Earth due to its strong affinity to water vapor. Ammonium Nitrate acids metal powders flammable liquids chlorates nitrates sulfur finely divided organics or combustibles Aniline nitric acid hydrogen peroxide Arsenic compounds any reducing agent Azides acids Bromine ammonia acetylene butadiene butane hydrogen sodium carbide turpentine finely divided metals Calcium water carbon dioxide carbon tetrachloride and chlorinated. Sulfuric acid 8 UN2584 II 8 B2 IB2 T8 TP2 TP12 TP13 154 202 242 1L 30 L B Alkyl sulfonic acids liquidor Aryl sulfonic acids liquid with not more than 5 percent free sulfuric acid 8 UN2586 III 8 IB3 T4 TP1 154 203 241 5L 60 L B Alkyl sulfonic acids solidor Aryl sulfonic acids solid with more than 5 percent free sulfuric acid.
The products of this reaction are phosphoric acid and calcium sulfate or gypsum along with other impurities. It is used to. Density 15 lb gal.
Ideally sulfuric acid should be stored in isolation from all other chemicals in an approved acid or corrosives safety cabinet. Flammable liquids are defined by dangerous goods regulations as liquids mixtures of liquids or liquids containing solids in solution or suspension which give off a flammable vapour have a flash point at temperatures of not more than 60-65C liquids offered for transport at temperatures at or above their flash point or substances transported at elevated temperatures in a liquid state and. Skin and other bodily burns from sulfuric acid can be more serious than burns from other strong acids.
These gasses are very flammable and the reason your RV or marine batteries must be vented outside. Concentrated Sulfuric Acid Water-insoluble compounds that are insoluble in 5 HCl are tested with concentrated sulfuric acid H 2SO 4. Acid burns to the face and eyes comprise about 50 of injuries related to the use of lead acid batteries.
Sulfuric acid can create a highly flammable hydrogen gas if it is spilled on metals. Virtually all organic compounds containing alkene functional groups or oxygen or nitrogen atoms are soluble in concentrated H 2SO 4. Greater than 95 over the range 05 to 10 mgm 3 of H 2 SO 4 on 045 µm pore size PTFE filters 4.
Contact with either is highly unlikely to occur unless the case is broken or spilled and then only contact with the acid is likely. The incompatibility of iron nickel and zinc is very important when considering storage transport and piping of sulfuric acid as. Product contains lead and sulfuric acid.
Carefully isolate it. 22 Chemical Hazards 221 Sulphuric Acid Lead acid batteries are usually filled with an electrolyte solution containing sulphuric acid. The burning of coal andor petroleum by industry and power plants generates sulfur dioxide SO 2 that reacts with atmospheric water and oxygen to produce sulfuric acid H 2 SO 4 and sulfurous acid H 2 SO 3.
Sulfuric acid A2 - Suspected human carcinogen. 58 Monday March 26 2012 Rules and Regulations Date of issue. Sulfuric acid American spelling or sulphuric acid Commonwealth spelling also known as oil of vitriol is a mineral acid composed of the elements sulfur oxygen and hydrogen with the molecular formula H 2 SO 4.
Perchloric acid presents special hazards. It is soluble in water with release of heat. Common metals known for this reactivity are sodium potassium magnesium calcium iron nickel and zinc.
This is then purified through the. It is highly toxic to aquatic organisms and plant life. This is a very corrosive chemical pH.
Paramount Chemicals Plastics Inc. It is not a hazard unless it comes contact. Gassing causes water loss which is why lead acid batteries need to have water added periodically.
It will char wood and most other organic matter on contact but is unlikely to cause a fire. Sulfuric Acid 93 Safety Data Sheet SDS Health 3 Fire 0 Reactivity 0 Personal Protection 3 0 Section 1. It is non-flammable Freeman said.
At this stage water is added so that gypsum and other insoluble materials are removed through filtration. Laboratory testing with generated atmospheres of sulfuric acid mist yielded a collection efficiency of. Diluted solutions of sulfuric acid can be handled out of the ventilated fume hood.
HYDROCHLORIC ACID is an aqueous solution of hydrogen chloride an acidic gas. A hazmat crew was called to a scene in Oakland where several train cars reportedly carrying sulfuric acid derailed. It will char wood and most other organic matter on contact but is unlikely to cause a fire.
Chemical Product and Company Identification Product Name. Sulfuric Acid ACS Safety Data Sheet according to Federal Register Vol. Sulfuric acid is a colorless oily liquid.
Hydrogen sulfate Chemical Formula. Only alkanes alkyl halides and some aromatic. Reacts with most metals particularly when diluted with water to form flammable hydrogen gas which may create an explosion hazard.
Facilities storing or utilizing this material should be equipped with. Sulfuric acid Group 1 - Carcinogenic to humans.
Sulfuric acid Sulfate SO42- H2SO4 Nitrous acid Nitrite NO2- HNO2 Chromic acid Chromate CrO42- H2CrO4 Hydrosulfuric acid Sulfide S-2 H2S Name the following acids. 93 wt sulfuric acid has a freezing point below zero at -21ᵒF so many industrial applications can utilize carbon steel tanks with 93 wt H 2 SO 4 uninsulated but 98 wt freezes at 30ᵒF and so much more care is needed for successful storage.

Type Of Reaction For Koh H2so4 K2so4 H2o Youtube
Shandong Sulfuric Acid Quotation Temporarily Stable on September 22.

Potassium sulfate sulfuric acid. Concentrated sulfuric acid acts as both an oxidizing and dehydrating agent. Dilute sulfuric acid is used as the electrolyte in this investigation. Check our FAQ section for more details.
The grade most often shipped is 93 weight percent. Chemical Product and Company Identification Product Name. Sulfates occur widely in everyday life.
The primary aim of the cards is to promote the safe use of chemicals in the workplace. The price of sulfuric acid in Shandong is temporarily stable this week 96-910 Quotation of Shandong sulfuric acid rose by 127 on September 6. Acetic acid acetic aldehyde acetone.
We are happy to answer any question you have. The number in parentheses is the metric equivalent in kilograms. 7 HClO4 q perchloric acid.
Our product line consists of chemical solutions prepared to exact quality standards and certified for use in laboratories and production processes. Other sources of sulfate which can cause sulfate attack include seawater oxidation of sulfide minerals in clay adjacent to the concrete sulfuric acid is produced which reacts with the concrete bacterial action in sewers anaerobic bacterial produce sulfur dioxide which dissolves in water and then oxidizes to form sulfuric acid. You can also cause a double replacement chemical reaction when you combine an acid and a.
Shandong sulfuric acid price rose by 1056 in August. Hydrogen sulfate Chemical Formula. Synonyms Dipotassium sulfate.
It contains an aluminium3. Paramount Chemicals Plastics Inc. Sulfuric Acid 93 Safety Data Sheet SDS Health 3 Fire 0 Reactivity 0 Personal Protection 3 0 Section 1.
In the environment. CID 1118 Sulfuric acid CID 5462222 Potassium Dates. The freezing point of different sulfuric acid concentrations can vary markedly.
32 14 57 52 11 Emergency Number US001-201-796-7100. Sulfuric acid dipotassium salt. The mixture is refluxed 24 h.
Different values of the pure water 0 concentration density reflect the fact that the measurements were done in different temperatures. The ICSC project is a common undertaking between the World Health Organization WHO and. Electrolysis of dilute sulfuric acid The products of electrolysing water acidified with sulfuric acid are hydrogen gas and oxygen gas Two experimental setups are described the Hofmann voltameter demonstration left diagram and a simple cell right diagram for use in schools and colleges for pupils to use.
SNI Pupuk Kalium Sulfat. Safety Data Sheet SD-318. An explosion occurred when concentrated sulfuric acid was mixed with crystalline potassium permanganate in a vessel containing moisture.
Sulfuric acid dipotassium salt. Potassium sulfate can also be used in pyrotechnics in combination with potassium nitrate to generate a purple flame. Manganese heptoxide was formed which explodes at 70C Delhez 1967.
The first number under the column headed RQ is the reportable quantity in pounds. C533 describe neutralisation as acid reacting with alkali to form a salt plus water including the common laboratory acids hydrochloric acid nitric acid and sulfuric acid and the common alkalis the hydroxides of sodium potassium and calcium. HNO3aq nitric acid.
Sulfates are salts of sulfuric acid and many are prepared from that acid. Sulfuric acid is available in many grades ranging from electrolyte grade 33 weight percent for batteries to 93 weight percent 66 deg Baume 98 weight percent and 20-22 weight percent fuming oleum containing excess dissolved sulfur trioxide. Between the sulfuric acid concentrations of 35 - 75 wt freezing is.
Potassium aluminium sulfate is a metal sulfate composed of potassium aluminium and sulfate ions in the ration 112. The cards are data sheets intended to provide essential safety and health information on chemicals in a clear and concise way. 001-800-ACROS-01 Europe call.
Uses advised against Food drug pesticide or biocidal product use. Sulfuric Acid 93 CAS. We regularly produce chemical solutions to specifications designed by government and regulatory bodies commercial and trade associations and the specific needs of individual users and businesses.
API Monogram 13A Certificate. Silver nitrate sodium chloride --- silver chloride and sodium nitrate. H2Teaq hydrotelluric acid.
Sulfuric acid barium hydroxide --- barium sulfate and water. It has a role as a flame retardant a mordant and an astringent. In masonry sulfates are present in bricks and can be.
Details of the supplier of the safety data sheet Emergency Telephone Number For information US call. The main target users are workers and those responsible for occupational safety and health. After cooling 500 ml of solvent CCl 4 CHCl 3 CH 2 Cl 2 is added and the mixture is poured to a flask containing 2000 ml ice.
Lead nitrate potassium iodide --- lead iodide and potassium nitrate saltpeter sodium bicarbonate baking soda vinegar --- carbonic acid and sodium acetate. 9 H2SO4aq sulfuric acid. It forms rhombic pyramids which melt at 197 C 387 F.
Potassium sulfate 21 Recommended Use Laboratory chemicals. 8 HClaq hydrochloric acid. Potassium AcetateCopper Sulfate 3c.
Sulfuric Acid Chemical Name. Potassium persulfate saturated S S Sulfuric acid 70 S O Potassium sulfate concentrated S S Sulfuric acid 80 S U Potassium sulfide concentrated S S Sulfuric acid 96 O U Potassium sulfite concentrated S S Sulfuric acid 98 concentrated O U Propane gas S S Sulfuric acid fuming U U Propargyl alcohol S S Sulfurous acid S S Propyl alcohol S S T Propylene glycol S S Tannic acid 10 S. Potassium hydrogen sulfate also known as potassium bisulfate KHSO 4 is readily produced by reacting K 2 SO 4 with sulfuric acid.
The sulfate or sulphate ion is a polyatomic anion with the empirical formula SO 2 4Salts acid derivatives and peroxides of sulfate are widely used in industry. HIaq hydroiodic acid. The fact that the use of modern highly concentrated fertilizers leads to unbalanced plant nutrition suggests that sulfate-based fertilizers which have up to now only been used for certain special crops may have a wider application.
A mixture of acrylonitrile with concentrated sulfuric acid must be kept well chilled otherwise a vigorous exothermic reaction occurs Chem. Potassium sulfate is a potassium salt. Potassium sulfate K 2 SO 4 is usually manufactured by reacting the chloride with sulfuric acidNormally K 2 SO 4 contains 43 K.
To a mixture of 136 g 10 mol phenylacetic acid 70 g sodium or potassium acetate and 16 g 01 mol anhydrous cupric sulphate is introduced 2000 ml anhydrous acetic anhydride in 4000 ml flask. This week the price of sulfuric acid in Shandong increased by 065 89. The latest news from Timuraya.
It is a metal sulfate an aluminium salt and a potassium salt. 3 Chemical and Physical Properties Expand this section. 1 Structures Expand this section.
This is the list of all compounds with density tables present in the full database - it was actual on May 4 th 2005 as the list is growing it may be already incomplete. H3PO4aq phosphoric acid. 2 Names and Identifiers Expand this section.
Battery acid Hydrogen sulfate Oil of vitriol Sulfuric acid aqueous Colorless to dark-brown oily odorless liquid. Keep reading to learn more about sulfuric acid its properties hazards and how to protect yourself against contamination.

Sulfuric Acid Safety Tips Sulfuric Acid Msds Information Velocityehs
2 - Battery Hazards.
Hazards of sulfuric acid. Very hazardous in case of skin contact corrosive irritant permeator of eye contact irritant corrosive of ingestion of inhalation. Sulfuric acid is a colorless oily liquid. 3050 SPRUCE ST ST.
Pure sulfuric acid does not exist naturally on Earth due to its strong affinity to water vapor. The main hazards associated with lead acid batteries are. Severe exposure can result in death.
Oil of Vitriol Chemical Name. In an experiment studying the clearance via the blood of radiolabeled sulfuric acid aerosol in different species the authors have observed that sulfur from sulfuric acid was rapidly cleared from 2 to 9 minutes from the lungs of animals into the blood following inhalation exposure. It is wise to treat unknown materials as very hazardous until they are.
Density 15 lb gal. Substance Substance name. Ideally sulfuric acid should be stored in isolation from all other chemicals in an approved acid or.
Toxicological Data on Ingredients. The Extreme Health Hazards Associated with Sulfuric Acid. 7664-93-9 RTK Substance Number.
International Agency for Research on Cancer IARC. It is formed through a natural process. Sulfate is a normal constituent of the blood and is a normal metabolite of sulfur-containing amino acids and.
Sulfuric acid Group 1 - Carcinogenic to humans. Cancer of the larynx lung cancer. Do not store near combustible materials.
Sulfuric acid Product Number. Exposure to moist air or water. The remaining injuries were mostly due to lifting or dropping batteries.
This chemical is unique because it not only causes chemical burns but also secondary thermal burns as a result of dehydration. Nitric acid concentration processes use a dehydrating agent such as sulfuric acid or magnesium nitrate to enhance the volatility of HNO3 so that distillation methods can surpass the azeotropic concentration of nitric acid. Sulphuric acid is highly acidic.
What Is Sulfuric Acid and Where Is It Used. Keep container closed when not in use. This safety session should teach you.
LOUIS MO 63103 UNITED STATES. Common bases are ammonium hydroxide potassium hydroxide caustic potash and sodium hydroxide caustic soda. Air mixtures with as little as 0000001 H 2 S are associated with a rotten egg smell.
178g 1910 305j7 917157 and 192644. Examples include the commonly used pharmaceutical antidepressant medication fluoxetine Prozac and the material PTFE Teflon. Page 3 of 8 Section 3.
It is used to make storage batteries fertilizers paper products textiles explosives. Store in a cool dry well-ventilated area away from incompatible substances. Oil of vitriol electrolyte acid battery acid matting acid H 2SO 4.
Do not store near alkaline substances. Elemental fluorine is produced from it. March 2016 CAS Number.
Sulfuric acid is a colorless and odorless substance also known as oil of vitriol. Unfortunately however our sense of smell is not a reliable alarm - at mixing. It is used to.
Concentrated sulfuric acid is extremely corrosive and can cause serious burns when not handled properly. Sulphuric Acid 93 percent. Pure compound is a solid below 51F.
Incompatible Materials Water Organic materials Strong acids Strong bases Metals Alcohols Cyanides Sulfides Hazardous Decomposition ProductsSulfur oxides Hydrogen Hazardous Polymerization Hazardous polymerization does not occur. Store protected from moisture. Trade Names and Synonyms.
Sulfuric Acid Process 3 Physical Properties 4 Thermal Properties 5 Personal Safety and First Aid 9 Health Hazards 9 Safety Precautions 9 Personal Protective Equipment 10 Site Facilities 12 First Aid 12 Handling Precautions 13 Spills or Leaks 13 Hazardous Chemical Reactions 13 Corrosion Hazards 14 Fire and Explosion Hazards 14 Fire Fighting 14 Engineering Control of Hazards 15 Transportation. Sulfuric acid American spelling or sulphuric acid Commonwealth spelling also known as oil of vitriol is a mineral acid composed of the elements sulfur oxygen and hydrogen with the molecular formula H 2 SO 4It is a colorless odorless and viscous liquid that is miscible with water. May 22 2015 Sulphuric Acid Page 1 of 6 SULPHURIC ACID SAFETY DATA SHEET SECTION 1.
Long term exposure to low concentrations or short term exposure to high concentrations can result in adverse health effects from inhalation. Sulfuric acid as a chemical compound is very stable. Common acids include hydrochloric acid sulfuric acid nitric acid chromic acid acetic acid and hydrofluoric acid.
Hydrofluoric acid is a solution of hydrogen fluoride HF in waterSolutions of HF are colourless acidic and highly corrosiveIt is used to make most fluorine-containing compounds. The chemical is not considered to be. Sulfuric Acid ACS CAS-No.
58 Monday March 26 2012 Rules and Regulations Date of issue. H 2 SO 4 is considered to last indefinitely without degrading breaking down and without losing its strength or chemical characteristics when stored by itself. 320 mgm 2 hours Mouse.
When sulphide mineral in the rock oxidizes sulphuric acid is formed. A few other uses of sulphuric acid are provided in this article. Hydrogen sulfide is a colorless flammable gas with a strong offensive odor.
510 mgm 2 hours Rat. It will char wood and most other organic matter on contact but is unlikely to cause a fire. Strong inorganic mists containing sulfuric acid are carcinogenic to humans.
7664-93-9 12 Relevant identified uses of the substance or mixture and uses advised against Identified uses. 13 02282018 EN English Page 1 SECTION 1. That batteries have many hazards including acid fire or explosion electrical shock and heavy.
Hazards Identification Potential Acute Health Effects. Therefore it is used in the. Sulfuric acids ability to burnchar is in addition to the corrosive damage it can cause contributing to the acids safety hazards.
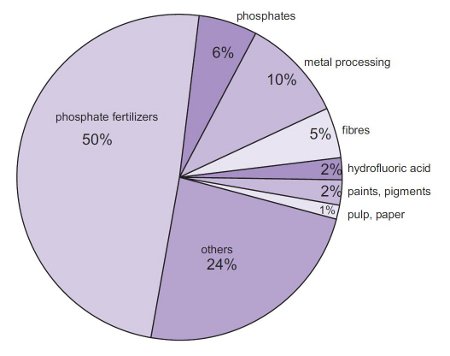
Sulfuric Acid Certified ACS Plus Revision Date 06-Jul-2018 Conditions to Avoid Incompatible products. 1 Chemical corrosive hazards 2 Risk of fire or explosion 3 Electrical shocks 4 Ergonomic hazards related to their heavy weight 5 Transportation hazards Acid burns to the face and eyes comprise about 50 of injuries related to the use of lead acid batteries. Sulphuric acid gives rise to phosphoric acid which produces phosphate fertilizers.
EHS MGA. Page 2 of 8. Sulfuric Acid ACS Safety Data Sheet according to Federal Register Vol.
Understanding volcanic hazards can save lives Not only can powerful explosive eruptions drastically alter land and water for tens of kilometers around a volcano but tiny liquid droplets of sulfuric acid erupted into the stratosphere can change our planets climate temporarily. UN 1830 Description and Use Sulfuric Acid is a clear colorless to brown odorless liquid. The level of exposure depends on dose duration and type of work being done.
Uses of Sulphuric Acid. Sulfuric acid H2S04 is a corrosive substance destructive to the skin eyes teeth and lungs. Inform laundry personnel of contaminants hazards.
Often used in an aqueous solution. It is sometimes referred to as sewer gas. Has been associated with.
Strong acid may also be made indirectly from the weak acid by using extractive distillation with a dehydrating agent. Trail Operations Trail British Columbia V1R 4L8 Emergency Telephone. Other chemicals can be corrosive too.
That following safety precautions will keep them safe when charging or changing batteries. 7664-93-9 Product code. It is corrosive to metals and tissue.
The list of sulfuric acid hazards is impressive and thats why you need to be very careful if you ever need to handle this material. It is soluble in water with release of heat. Workers may be harmed from exposure to sulfuric acid.
Interestingly the human nose is more sensitive to H 2 S than any gas monitoring instrument we have today. Laboratory chemicals Synthesis of substances 13 Details of the supplier of the safety data sheet Company. Check the supplier labels on chemical product containers.
It is commonly used to etch glass and silicon. This dangerous chemical is capable of corroding skin paper metals and even stone in some cases.
Sulfuric acid also called sulphine acid battery acid and hydrogen sulfate is a clear colorless oily liquid that is very corrosive. Generally dilute solutions are more effective than concentrated ones.
Yes you should be concerned about DHMO.
What does sulfuric acid do for food. The sulfuric acid removes water from the sugar in a highly exothermic reaction releasing heat steam and sulfur oxide fumes. These compounds interact with common atmospheric chemicals including hydrogen and oxygen to form sulfuric and nitric acids in the air. COMPOSITION INFORMATION ON INGREDIENTS HAZARDOUS COMPONENTS CAS Registry No.
To do so mix 8 mL of a sodium dichromate solution with 4 mL of sulfuric acid. Thick fuming yellow liquid. Not bioaccumulate or bioconcentrate through the food chain see Ecological Information Section 12.
Acetic acid is used in day-to-day life as vinegar. Citric acid is used as a preservative in sauces and pickles. Normal rain is slightly acidic with a pH of 56 while acid rain generally has a pH between 42 and 44.
As a bleaching agent. At this stage water is added so that gypsum and other insoluble materials are removed through filtration. Unlike sulfuric acid itself sulfonic acids can be solids.
And in lead-acid batteries used in most vehicles 25. Sulfuric acid fuming 26-29. Phosphoric acid and sulfuric acid are significant products of this normal metabolism of dietary nutrients such as proteins and phospholipids.
Density 165 lb gal. Sulfuric Acid and Sugar Chemistry Demonstration. Used as a bactericide and fungicide especially in food processing.
DANGER Variations in chemical behavior during handling due to factors such as temperature pressure and concentrations can cause equipment to fail even though it passed an initial test. Causes of acid rain. As a reagent in making caprolactam and glycerol.
Incompatible Materials Water Organic materials Strong acids Strong bases Metals Alcohols Cyanides Sulfides Hazardous Decomposition ProductsSulfur oxides Hydrogen Hazardous Polymerization Hazardous polymerization does not occur. Place this vial into the correct hole in the plastic container ie. Nonvolatile acid loads or loss of HCO 3 which is an.
Dehydrating sugar by reacting it with. And its name is hydrofluoric acid. This is typically found in nature as the substance fluorapatite.
Swirl this container gently a few times then waft the air from the mouth of the container towards your nose by fanning the air toward you with a hand with the container placed roughly 8-12. Concentrated sulfuric acid seems to have effects similar to the expected effects of sulfuric acid on sugar. Acid rain describes any form of precipitation that contains high levels of nitric and sulfuric acids.
Iodized salt a vestige of a. But there is an acid thats really properly scary. Sulfuric Acid Certified ACS Plus Revision Date 06-Jul-2018 Conditions to Avoid Incompatible products.
Corrosive to metals and tissue quickly causing severe. These acids return. It can also occur in the form of snow fog and tiny bits of dry material that settle to Earth.
It can also be mixed with acetic acid vinegar in a 1 to 1 ratio or with hydrochloric acid. The effects of course were most severe in. The chemical reaction causes the sulfuric acid to break down into the water stored inside each cell to dilute the acid.
Many acids can be found in various kinds of food as additives as they alter their taste and serve as preservatives. That process is the storing of energy. Acid rain describes any form of precipitation that contains high levels of nitric and sulfuric acids.
Concentrated sulfuric acid 93 is added to tricalcium phosphate rock. It releases chemical sulfur from the rocks and sand creating a river rich in sulfuric acid. Aside from the sulfurous odor the reaction smells a lot like caramel.
22 This debate featured in an episode of the sitcom Malcolm in the Middle Traffic Jam in which Francis insisting the Quacks as they were called would dissolve in his stomach rather than expand takes up the dare to eat 100 of them and does so but gets very sick in the process. Remember a battery doesnt store electricity. See Section 8 for Occupational Exposure Guidelines.
Very toxic by inhalation. Cole-Parmer does not warrant neither express nor implied that the information in this chart is accurate or complete or that any material is suitable for any purpose. CID 1118 Sulfuric acid CID 24682 Sulfur trioxide Dates.
For most dilutions the water can be at room temperature. Acid rain forms because of atmospheric sulfur dioxide and nitrogen dioxide which are emitted from natural and human-made sources. To maintain acid-base homeostasis these nonvolatile acids must be excreted by the kidney.
Other nonvolatile acids such as ketoacids and lactic acids are produced in pathologic conditions. Acids generally take longer to etch steel than do chemicals that form acids in water however. These include volcanic activity and burning fossil fuels.
Because of its corrosiveness sulfuric acid at a high. And as a polymerization catalyst for polyester resinsEPA 1998. The acid is washed into streams and rivers where it reacts with rocks and sand.
Peracetic acid is a colorless liquid with a strong pungent acrid odor. CONCENTRATION wgtwgt Sulphuric Acid 7664-93-9 93 Note. FIRST AID MEASURES Eye ContactSymptoms.
In the purification of petroleum. Measure 5 cm3 of 01M sodium thiosulfate solution into a 10 cm3 measuring cylinder. The gases combined with water in the atmosphere to produce acid rain destroying crops and killing livestock.
It stores the. This is then purified through the. The products of this reaction are phosphoric acid and calcium sulfate or gypsum along with other impurities.
Swirl gently to mix then add 10 drops of the mixed solution to a test tube or other small container containing the alcohol. Cubic granular and fine enough to slip through your fingers this is typical salt shaker stuff. It is used in the manufacture of fertilizers explosives other acids and glue.
Hydrogen peroxidesulfuric acid tests are appropriate for testing resins and their potency. Place about 10 cm3 of 1M hydrochloric acid or 05M sulfuricVI acid in the acid vial. It can also occur in the form of snow fog and tiny bits of dry material that settle to Earth.
So the use of power depletes the acid. Botanical greenhouse operators cultivation facilities and commercial grow operations use hydrogen peroxide to kill mold. The white sugar turns into a black carbonized tube that pushes itself out of the beaker.
Carbonic acid is one of the most. When the battery is charging back up the process reverses and the batterys recharging builds the acid molecules back up. Theres an even stronger acid hydrocholoric acid HCl in your stomach and not only does it not burn you from the inside out usually it actually helps you to digest your food and keeps you safe from nasty bacteria.
Acid helps miners remove coal from the surrounding rocks. Government and the Centers for Disease Control CDC do not classify Dihydrogen Monoxide as a toxic or carcinogenic substance as it does with better known chemicals such as hydrochloric acid and benzene DHMO is a constituent of many known toxic substances diseases and disease-causing agents environmental hazards and can even be. As an oxidant for preparing epoxy compounds.
This step is only necessary when youll be handling extremely concentrated acid solutions such as 18M sulfuric acid or 12M hydrochloric acid. A mixture of 30 hydrogen peroxide sold by Lab Alley is commonly mixed with concentrated sulfuric acid to perform these tests. Exposure to moist air or water.
Sulfuric acid should be used only in concentrations from 10 to 25 percent. The one without the cross. Phosphoric acid for example is a component of cola drinks.
In the US its often enhanced with iodine aka. In the pickling of metal. Cool the water youll be using by keeping it in a container surrounded by ice for at least 20 minutes before the dilution begins.
