Once a true hydrate had been heated. Greek prefixes are attached to the word hydrate to indicate the number of water molecules per formula unit for the compound eg BaOH 2 8H 2 O.

When Copper Ii Sulfate Pentahydrate Cuso Clutch Prep
Thus the product is called copper sulfate pentahydrate.
Copper sulfate pentahydrate formula. Ac-CoA Synthase Inhibitor - CAS 508186-14-9 - Calbiochem. What is the percent water in copperII sulfate pentahydrate CuSO 4 5 H 2 O. BaOH 2 8H 2 O.
When the chemical formula for a hydrated ionic compound is written the formula for the ionic compound is separated from the waters of hydration by a centered dot. A tolerance of 1 ppm is established in potable water for residues of copper resulting from the use of the algicides or herbicides basic copper carbonate malachite copper sulfate copper monoethanolamine and copper triethanolamine to control aquatic plants in reservoirs lakes ponds irrigation ditches and other potential sources of potable water. Copper II sulfate pentahydrate 100 -250 231-847-6 029-023-00-4 Acute ToxO 3.
The water in these compounds can be removed quantitatively by heating the compound with a bunsen burner. Because of the presence of copper the compound has a characteristic blue. When all the water has been removed the ionic compound is said to be anyhydrous.
It is found in foods such as organ meats seafood nuts seeds wheat bran cereals grain products and cocoa products. Calculate the formula mass. 8 water molecules octahydrate.
It is produced industrially by treating copper metal with hot concentrated sulfuric. Write hydrate after the prefix 1 mono 2 di 3 tri 4 tetra 5 penta 6 hexa 7 hepta 8 octa 9 nona 10 deca Example. For example the anhydrous compound copperII sulfate also exists as a hydrate containing five water molecules and named copperII sulfate pentahydrate.
A strong base is a base that is completely dissociated in an aqueous solutionThese compounds ionize in water to yield one or more hydroxide ion OH- per molecule of base. H410 7732-18-5 ZC0110000 Water 750 -900 231-791-2 NA No data available. One litre of Benedicts reagent can be prepared by mixing 173 grams of copper sulfate pentahydrate CuSO 45H 2 O 100 grams of sodium carbonate Na 2 CO 3 and 173 grams of sodium citrate in distilled water required quantity.
The body stores copper mostly in the bones and muscles. Copper sulfate pentahydrate CuSO45H2O or CuH10O9S CID 24463 - structure chemical names physical and chemical properties classification patents literature biological activities safetyhazardstoxicity information supplier lists and more. H315 Eye Damage 2.
This dark blue to purple solid is a salt of the metal complex CuNH 3 4 H 2 O 2. BaIO 3 2. Aqueous solution of copper2 sulfate is an acid.
In hydrated CuSO4 the water molecules surrounding the Central metal Cu function as ligands which bring d-d transition and hence emits blue colour in visible. C 27 H 42 N 7 Na 2 O 20 P 3 S xH 2 O. Use a prefix for the number of water molecules see below 3.
This compound can be prepared by adding concentrated solution of ammonia to a. Write the name of the ionic compound 2. Formula K sp 25 C Aluminium hydroxide.
Copper is a mineral. Divide the mass of water in one mole. The most common form of copper sulfate is its pentahydrate given by the chemical formula CuSO 45H 2 O.
Washing soda is the common name for a hydrate of sodium carbonate containing 10 water molecules. Copper sulfate also known as blue vitriol Salzburg vitriol Roman vitriol blue copperas or bluestone is a chemical compound comprised of Copper Sulphur and Oxygen whose formula is CuSO4. 1 Cu6355 gmol 1 S3207 gmol 4 O1600 gmol 15962 gmol Formula mass 15962 gmol 5 H 2 0 1802 g H 2 0mol 24972 gmol 2.
CuSO 4 5H 2 O. Name Formula Systematic Name Common Name Formula Name Formula Methane CH 4 Methanoic acid Formic acid HCO 2H 12-Dichloroethane C 2H 4Cl 2 Ethane C 2H 6 Ethanoic acid Acetic acid CH 3CO 2H Methylamine CH 3NH 2 Propane C 3H 8 Propanoic acid Propionic acid C 2H 5CO 2H. The loss in mass of the compound after losing the water can be used to calculate the amount of water originally in the hydrated sample.
Copper sulfate CuSO 4 is an inorganic compound used in agriculture to control insects plant roots snails bacteria and fungi and its one of the best pool algaecides around. However it is the hydrated form of the salt and has 5 water molecules. It is closely related to Schweizers reagent which is used for the production of cellulose fibers in the production of rayon.
For example copper sulfate is a vernacular name which may refer to copperI sulfate or copperII sulfate. Stable at room temperature in closed containers under normal storage and handling conditions. The pentahydrate CuSO 4 5H 2 O the most commonly encountered salt is bright blue.
2286 SG 156 C 4 C 148gkg 0 C 736gkg 100 C Basic copper sulfate. Copper Sulphate is an odorless crystalline substance electric blue in color highly toxic and not safe to work with. CuSO4 5H 2 O Naming a Hydrate 1.
An illustration describing the. Empirical Formula Hill Notation. Blue crystals granules or powder.
The systematic name is. 95562 anhydrous basis Compare Product No. However it can be noted that the anhydrous form of this salt is a powder that is white.
Incompatibilities with Other Materials. Here the copperII sulfate acts as a source of Cu 2 ions the sodium carbonate provides an alkaline medium and the sodium citrate forms complexes with the Cu. This form is characterized by its bright blue colour.
2O copper II sulfate pentahydrate. Common Covalent Binary Inorganic Compounds. The pentahydrate x.
Hold eyelids apart and flush eyes with plenty of water for at least 15 minutes. When determining the formula mass for a hydrate the waters of hydration must be included. Write the formula of the anhydrous followed by a dot then the of moles of water and H 2O Example.
Some examples are copper II sulfate sodium sulfate nickel II sulfate iron II sulfate cobalt II chloride magnesium sulfate sodium. CopperII sulfate also known as copper sulphate are the inorganic compounds with the chemical formula CuSO 4 H 2 O x where x can range from 0 to 5. CopperII sulfate also known as copper sulphate are the inorganic compounds with the chemical formula Cu SO 4 H 2 O x where x can range from 0 to 5The pentahydrate x 5 is the most common form.
Archaic Name An archaic name is an older name for a chemical that predates the modern naming conventions. Specific gravity density 67. Ac-CoA Synthase Inhibitor - CAS 508186-14-9 - Calbiochem.
The ratio of water mol ecules to the anhydrous salt is constant. H319 Aquatic A 1. Vapor pressure mmHg at 25 C 1.
A hydrate is an ionic compound that has a definite amount of water molecules attached to its crystalline structure. CopperII sulfate pentahydrate CuSO 45H 2O 11 Undeca. Molecular FormulaCuO4S5H2O Molecular Weight24968 Section 10 - Stability and Reactivity Chemical Stability.
High temperatures dust generation exposure to moist air or water. The CuSO 4 molecule consists of an ionic bond between the copper cation Cu 2 and the sulfate anion SO 4 2-. Many compounds exist as hydrates.
Molecular weight gmol 3. 3CuOH 2 CuSO 4. Why is CuSO4 blue.
Public health information CDC Research information NIH SARS-CoV-2 data NCBI Prevention and treatment. Copper sulfate pentahydrate. H400 Aquatic C 1.
When copper sulfate CuSO4 reacts with water H2O the product is still copper sulfate. Older names for this compound include blue vitriol bluestone vitriol of copper and Roman vitriol. Determining the Formula of a Hydrate.
TetraamminecopperII sulfate is the salt with the formula CuNH 3 4SO 4 H 2 O. First Aid Measures Description of First Aid Measures.
Densities of common products in both Imperial and SI-units. When ammonium salt is heated with conc.

3 Ways To Make Copper Acetate Wikihow
Copper plating has a variety of uses.

How to make copper acetate from copper sulphate. Many other oxyanions form complexes. These are also made of polyethylene but copper is added into these. The chocolate brown film of copper oxide advances the patination process and provides architects with a different colour option to the bright new copper.
Trade Statistics by Product HS 6-digit Quickly obtain export and imports of HS 6-digit products by clicking on the product name. Copper powder has been found to be superior to all other known fire extinguishing agents for lithium. The lippes loop is the most popular and commonly used device.
We provide solutions to students. The use of missī thought to be sanctified by Fāṭimah the Prophets daughter became deeply. Meeting the requirements of the ZDHC MRSL V20 does not.
The agent of choice for teeth blackening became known as missī a powdery mixture of 1 iron and copper sulphate 2 a plant source of tannins such as myrobalans and 3 flavouring agents. Because of this some artists even treat copper surfaces with acetic acid to form copper acetate also known as verdigris. The Commission shall without delay make the information referred to in points a to g of paragraph 1 and in paragraphs 2 and 3 available electronically to all competent authorities.
The most soluble cadmium salts are chloride acetate nitrate and sulphate while the insoluble salts are the sulphides. Since the lungs are rich in carbon dioxide a variety of cadmium sulphides can react with the carbon dioxide in the lungs. Navy for fighting lithium and lithium alloy fires.
Simply stir one tablespoon more if preferred directly into food or drink. Eventually a film of green copper salts will appear on top of the oxide layer. 01 copper-zinc malonate cream applied twice daily for 8 weeks showed significant reduction of wrinkles.
TPW Vitamin Mineral Blend Tricalcium Phosphate Potassium Citrate Sodium Chloride Potassium Dihydrogen Phosphate Magnesium Oxide Heavy Vitamin C Zinc Gluconate Ferric Pyrophosphate Vitamin E Acetate Potassium Iodide Vitamin B3 Sodium Selenite Vitamin A Acetate Vitamin B5 Copper Gluconate Manganese Sulphate Monohydrate D-Biotin Vitamin B12 Vitamin D2 Vitamin B6 Vitamin. This will take up to twenty years or more. The copper compound smothers the fire and provides an excellent heat sink for dissipating heat.
Change selection to view exportsimports by country to a country. Aesthetically it can be used to create a steampunk look on otherwise ill-fitting metals. The loop is double s shaped and has an attached tail made of fine nylon threads.
Please Use Our Service If Youre. It is made of polyethylene and contains barium sulphate which makes it possible to be located when required by x-ray. Other harmful chem- icals present in the water may be formaldehyde based dye fixing agents chlorinated stain remo- vers hydro carbon based softeners non bio deg- radable dyeing chemicals.
And heavy metals like copper arsenic lead cad- mium mercury nickel and cobalt and certain auxiliary chemicals all collectively make the te- xtile effluent highly toxic. Wishing for a unique insight into a subject matter for your subsequent individual research. It is used in a fungicide called the Bordeaux mixture.
To practice you can open a chilled macro lager pour it into a glass with a small piece of cleaned iron or copper pipe. The 3000 m 2 of copper sheet on the Copper Box in Londons Olympic Park is pre-oxidised in the copper factory. Worker exposure to chemical.
Its quite an experience hearing the sound of your voice carrying out to a over 100 first year. Do this incrementally until you can perceive the off flavor. The second was a Copper extinguishing agent specially developed by the US.
Used zinc sulphate as 1 2 and 4 topically in three groups of 30 patients each with herpes genitalis for a period of 3 months and observed that. Whilst ZDHC takes every reasonable effort to make sure that the information is as accurate as possible ZDHC makes no claims promises or guarantees about the accuracy completeness or adequacy of the contents of this document. This is because the biological composition.
The main ingredient used to blacken teeth was a dark-brown solution of ferric acetate called kanemizu. A replace applicable national environmental or workplace safety restrictions. In reducing flame cupric metaborate is reduced to metallic copper which is red and opaque.
NaOH ammonia gas is evolved which gives white fumes with dilHCl due to. Shop by department purchase cars fashion apparel collectibles sporting goods cameras baby items and everything else on eBay the worlds online marketplace. Anodizing the object after plating can create brilliant multicolored objects.
Material Properties - Material properties for gases fluids and solids - densities specific heats viscosities and more. Rather than buying copper acetate pre-made we will make it ourselves. The presence of carbon dioxide can be detected by passing the gas through lime water which becomes milky and hydrogen is oxidised to water as evidenced by the blue colour of anhydrous copper sulphate.
The system was highly efficient achieving removal of the dye and COD of around 98 and 95 respectively. CopperII sulfate forms a blue crystalline pentahydrate the most familiar copper compound in the laboratory. The trace element solution comprised of zinc sulphate heptahydrate manganeseII chloride tetrahydrate cobaltII chloride hexahydrate nickelII chloride hexahydrate sodium molybdate dihydrate boric acid and copperII chloride dihydrate.
The liver and the kidneys are the organs with the highest concentration of cadmium. Carbon and hydrogen are detected by heating the compound with copper ll oxide. In vivo use of zinc acetate gel has been found effective in preventing sexual transmission of HSV-2 and HIV infections.
Complan Original should not be added to boiling food or drinks. Copper sulphate used as an algicide in water purification. This group includes ammonium ion NH 4.
Then sit back and enjoy with the knowledge you have added extra fortification. What happens when zinc granules are treated with a dilute solution of H 2 SO4 HCl HNO 3 NaCl and NaOH also write the chemical equations if a reaction occurs. The copper enhances the contraceptive effect.
In the case of everyday copper utensils copper is most likely to form a brown or black tarnish in the presence of oxygen moisture human touch dust and certain. These include copperII acetate copperII nitrate and copperII carbonate. CWhen silver metal is added to Copper Sulphate solution there will not be any reaction as silver is non-reactive metal.
Prepare the food per serving and use within 2 hours. Density - Density of different solid materials liquids and gases. Definitions and convertion calculators.
I wanted this instructable to be as easy as possible safe as possible and as cheap as possible. These organic mate- rials. Potassium sulphate reacts with copper sulphate.
Alternatively make up into a paste and add to your food or drink. Ball-and-stick model of the complex CuNH 3 4 H 2 O 2 2 illustrating the octahedral coordination geometry. Chemical reactions involved in Group Zero analysis a Sodium hydroxide test.
Identification of Cations Basic Radicals Analysis of Group Zero. The carbon in the compound is oxidised to carbon dioxide. If not drop it back in for an additional time period.
Let it sit for about 30 seconds then pull it out and see if you can smell and taste metallic notes. That information may be used by competent authorities only for the purposes of market surveillance market analysis evaluation and consumer information in the context of Articles 25 26 and 27. Verdigris gives copper a green color but it is not as durable as natural patinas and it is soluble.
Cu2CO3 copper I carbonate 3. KHCO3 potassium bicarbonate 7.

How May Copper Acetate Ligands Be Manipulated To Change Colors Chemistry Stack Exchange
EleCare Jr Amino acid-based nutrition powder for ages 1 and up ELECARE JR is a 30 Calfl oz nutritionally complete amino acid-based medical food for ages 1 and older who cannot tolerate intact or hydrolyzed protein.

Copper acetate from copper carbonate. Ionic bonds are atomic bonds created by the attraction of two differently charged ionsThe bond is typically between a metal and a non-metal. Our product line consists of chemical solutions prepared to exact quality standards and certified for use in laboratories and production processes. Palm sells various forms of crystals and powders including.
15 mL 10 M Copper II sulfate 06 g Magnesium powder 30 mL 10 M Sulfuric acid 15 mL 5 Sodium acetate solution CYCLE B 15 mL 05 M Copper II chloride 15 mL 05 M Sodium carbonate 15 mL 10 M Hydrochloric acid Equipment needed per group to run both cycles 50 mL Erlenmeyer flask Glass stirring rod. Ionic bonds also melt at high temperatures. 28 zinc carbonate ZnCO3 29 manganese VII arsenide Mn3As7 30 copper II chlorate CuClO32 31 cobalt III chromate Co2CrO43 32 ammonium oxide NH42O 33 potassium hydroxide KOH 34 lead IV sulfate PbSO42 35 silver cyanide AgCN 36 vanadium.
Amyl acetate 100 O U Butyric acid 100 S S Amyl alcohol 100 S S Amyl. However the pH of the ileal digesta was increased by Cu supplementation P 00002. Draganjacs Introduction to Chemistry CHEM1003 General Chemistry I CHEM1013 and General Chemistry II CHEM1023 classes are responsible for learning the names and formulae for the common acids and common reagents and for learning the names formulae and the charges for the common cations and anions listed below.
IronII carbonate FeCO 3 3210 11. Copper acetate CuC 2 H 3 O 2 2 weighs 1 880 kgm³ 11736457 lbft³ weight to volume volume to weight price mole to volume and weight mass and molar concentration density Copper carbonate hydroxide azurite CuCO 3 CuOH 2 weighs 3 880 kgm³ 24222049 lbft³ weight to volume volume to weight price mole to volume and weight mass and molar concentration. 8 CuOH copperI hydroxide.
AgNO3 silver nitrate 10. Copper chloride saturated S S Fuel oil S U Cooper cyanide saturated S S Furfural 100 O U Copper fluoride 2 S S Furfuryl alcohol S O Copper nitrate saturated S S Copper sulfate dilute S S G Copper sulfate saturated S S Gallic acid saturated S S Corn oil S S Gasoline S U Cottonseed oil S S Glucose S S. Cu2CO3 copper I carbonate 8.
CopperII chromate CuCrO 4 3610 6. 9 ZnNO 2 2 zinc nitrite. Many other oxyanions form complexes.
CopperII sulfate forms a blue crystalline pentahydrate the most familiar copper compound in the laboratory. In the case of cupric carbonate copper was much more distributed in the tissues especially in the liver than for copper sulfate. IronII sulfide FeS 610 19.
6 K 3N potassium nitride. Write the formulas for the following chemical compounds. ZnHCO32 zinc bicarbonate 15.
Acetate CH3COO-C2H3O2-bromate BrO3-carbonate CO3 2-chlorate ClO3-chlorite ClO2-chromate CrO4 2-cyanide CN-dichromate Cr2O7 2-hydrogen carbonate bicarbonate HCO3-hydrogen sulfate bisulfate HSO4-hydrogen phosphate biphosphate HPO4 2-hydroxide OH-hypochlorite ClO-iodate IO3-nitrate NO3-nitrite NO2-oxalate C2O4 2-perchlorate ClO4-periodate IO4-permanganate MnO4-peroxide O2 2-phosphate. Additionally whereas the classic Huisgen 13-dipolar cycloaddition often gives mixtures of regioisomers the copper-catalyzed reaction allows the synthesis of the 14-disubstituted regioisomers specifically. A copper-catalyzed variant that follows a different mechanism can be conducted under aqueous conditions even at room temperature.
Ball-and-stick model of the complex CuNH 3 4 H 2 O 2 2 illustrating the octahedral coordination geometry. Ammonium chloride granular boric acid granular chromic acid crystal chromic acid flake cobalt carbonate powder cobalt sulfate crystal copper sulfate crystal malic acid powder nickel acetate powder nickel carbonate powder48 nickel carbonate granules40 nickel carbonate paste-38 nickel chloride crystal nickel. By contrast a later developed ruthenium-catalyzed.
Polyatomic ions are ions which consist of more than one atom. IronIII arsenate FeAsO 4 5710 21. SnSO4 tin II sulfate 14.
IronII hydroxide FeOH 2 8010 16. Barium carbonate BaCO 3 258 109 Barium chromate BaCrO 4 117 1010 Barium fluoride BaF 2 184 107 Barium hydroxide octahydrate BaOH 2 8H 2 O 255 104 Barium iodate BaIO 3 2 401 109 Barium iodate monohydrate BaIO 3 2 H 2 O 167 109 Barium molybdate BaMoO 4 354 108 Barium nitrate BaNO 3 2 464 103 Barium selenate BaSeO 4 3. CaSO4 calcium sulfate 5.
7 SO 2 sulfur dioxide. We regularly produce chemical solutions to specifications designed by government and regulatory bodies commercial and trade associations and the specific needs of individual users and businesses. These include copperII acetate copperII nitrate and copperII carbonate.
The chocolate brown film of copper oxide advances the patination process and provides architects with a different colour option to the bright new copper. 5 FePO 4 ironIII phosphate. 11 silicon dioxide SiO 2.
PbCO32 lead II carbonate 12. The structure of the bond is rigid strong and often crystalline and solid. CopperII acetate also referred to as cupric acetate is the chemical compound with the formula CuOAc 2 where AcO is acetate CH 3 CO 2The hydrated derivative Cu 2 OAc 4 H 2 O 2 which contains one molecule of water for each copper atom is available commercially.
For example nitrate ion NO 3- contains one nitrogen atom and three oxygen atomsThe atoms in a polyatomic ion are usually covalently bonded to one another and therefore stay together as a single charged unit. It is used in a fungicide called the Bordeaux mixture. FeC2H3O22 iron II acetate 4.
12 nickel III sulfide Ni 2S 3. CopperII carbonate CuCO 3 1410 10. This will take up to twenty years or more.
10 V 2S 3 vanadiumIII sulfide. We sell Borax Boric acid Sodium Bicarbonate Epsom Salt Copper Sulphate Calcium Carbonate Trisodium Phosphate Sodium Thiosulphate Ammonium Sulphate. CdOH2 cadmium hydroxide 13.
Eventually a film of green copper salts will appear on top of the oxide layer. CopperII sulfide CuS 610 37. BaClO42 barium perchlorate 11.
K2Cr2O7 potassium dichromate 6. FeNO22 iron II nitrite 9. Mean copper absorption of 57 percent range 40 to 70 per cent following oral administration of 04 - 45 mg copper as copper acetate to four volunteers.
Students enrolled in Dr. The copper level increased progresively in mitochondria lysosomal fractions of the liver in proportion to the period of administration. In the 105000 g supernatant fraction copper was distributed in the metallothionein fraction rather than in the superoxide.
Copper source had no effect on the pH of digesta in the proximal jejunum P 050 or the ileum P 096. 4 TiSO 4 2 titaniumIV sulfate. The 3000 m 2 of copper sheet on the Copper Box in Londons Olympic Park is pre-oxidised in the copper factory.
Copper II ferrocyanide CuFeCN 6 1310 16. This activity includes every compound formula and name that can be formed from the list 44 Ions provided in Chemistry A at Pickerington High School Central. CopperII hydroxide CuOH 2 2210 20.
Anhydrous copperII acetate is a dark green crystalline solid whereas Cu 2 OAc 4 H 2 O 2 is more bluish-green. An early human study suggested a maximum blood copper concentration was reached some two hours after oral copper chloride administration 15 -. 3 P 2O 5 diphosphorus pentoxide.
Copper addition decreased P 004 the pH of digesta in the proximal jejunum.
Customers who viewed this item also viewed. Roebic FRK-1LB Foaming Root Killer Clears.

Hydrated And Anhydrous Copper Sulphate Youtube
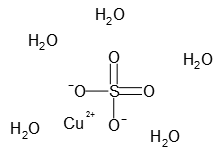
Since the only component other than H2O and Cu 2 is the sulfate ion SO 4 2 we can now determine the complete formula of the hydrated copperII sulfate.
Copper 2 sulfate hydrate. The ICSC project is a common undertaking between the World Health Organization WHO and. What color is copperII sulfate. Weiterhin existieren auch ein Trihydrat CuSO 4 3 H 2 O und KupferII-sulfat-Monohydrat CuSO 4 H 2 O.
This material contains CopperII sulfate pentahydrate listed as Copper compounds nos 100 CAS 7758-99-8 which is subject to the reporting requirements of Section 313 of. Kimberly Graziano Hyunjae Kim. Am geläufigsten ist das Pentahydrat CuSO 4 5 H 2 O.
Did the copper change its charge in this reaction. FREE Shipping by Amazon. The primary aim of the cards is to promote the safe use of chemicals in the workplace.
For a general form copperII sulfate hydrate formula CuSO_4 xH_2O we use x to represent the number of water molecules present as water of crystallization per formula unit of anhydrous copperII sulfate CuSO_4. REWEIGH the dish and hydrate. A Case Study Journal of Science Education and Technology 17 466-482.
Heat strongly for 5 minutes or longer until the blue. People get magnesium from their diet but sometimes magnesium supplements are needed if magnesium levels are too low. For example blue vitriol or copper sulfate pentahydrate CuSO 4 5H 2 O is blue copper sulfate trihydrate CuSO 4 3H 2 O is blue and anhydrous copper sulfate CuSO 4 is.
Copper Sulfate Pentahydrarte. Ammonium Sulfate Ammonium ions and Sulfate ions NH 4 and SO 4 2 NH 4 2SO 4 Calcium Phosphate Calcium ions and Phosphate ions Ca2 and PO 4 3 Ca 3PO 2 Waters of hydration use table to left copperII sulfate pentahydrate CuSO 45H 2O 11 Undeca. Add about 2000 grams copperII sulfate hydrate to the dish.
Get it Thu Dec 2 - Tue Dec 7. It is a metal sulfate an aluminium salt and a potassium salt. Research suggests that the nature of these bonds is not as simple as.
The pentahydrate CuSO 4 5H 2 O the most commonly encountered salt is bright blue. Magnesium is a mineral that is important for normal bone structure in the body. Ready to use.
The dot after the formula for copper sulphate indicates bonds with water molecules. 40 out of 5 stars 7. The main target users are workers and those responsible for occupational safety and health.
Get it as soon as Wed Dec 1. MM-CoA α-Methylmalonyl coenzyme A tetralithium salt Empirical Formula Hill Notation. Potassium aluminium sulfate is a metal sulfate composed of potassium aluminium and sulfate ions in the ration 112.
The formula of the hydrated form is CuSO 4. For example the anhydrous compound copperII sulfate also exists as a hydrate containing five water molecules and named copperII sulfate pentahydrate. Copper sulfate injected ip at 2 mg copperkg into vitamin e selenium deficient rats caused a 6 fold increase in the formation of the lipid peroxidation product ethane caused acute mortality in 45 rats.
TIP THE TEST TUBE SLIGHTLY DOWNWARD 2. SAFETY EXPERIMENTAL PROCEDURE. WRITE ALL of the digits from the balance reading.
Allow the test tube and its contents to cool. When copperII sulfate pentahydrate CuSO 4 5H 2 O is heated it decomposes to the dehydrated form. Washing soda is the common name for a hydrate of sodium carbonate containing 10 water molecules.
The cards are data sheets intended to provide essential safety and health information on chemicals in a clear and concise way. Kristallwasserverlust bei CuSO 4 5 H 2 O TGADTA Kupfersulfat-Pentahydrat CuSO 4. Write hydrate after the prefix 1 mono 2 di 3 tri 4 tetra 5 penta 6 hexa 7 hepta 8 octa 9 nona 10 deca Example.
Schroeder JD and Greenbowe TJ. Implementing POGIL in the Lecture and the Science. Zinc sulfate is an inorganic compoundIt is used as a dietary supplement to treat zinc deficiency and to prevent the condition in those at high risk.
Copper sulfate 2 lb. It contains an aluminium3. The balanced equation is.
Our unknown hydrate may be a hydrate of copperII sulfate magnesium sulfate ironIII chloride or ironIII nitrate. Hold it over the flame with a test tube holder and heat it for a few minutes with the mouth of the tube slightly below the base of the tube. Only 4 left in stock - order soon.
Kurzbeschreibung weißes bis graues geruchloses Pulver wasserfrei. CopperII sulfate forms a hydrate which contains 361 by mass water. Clean Finish Disinfectant Cleaner Case of 2 564 oz.
The most common form includes water of crystallization as the heptahydrate with the formula Zn SO 4 7H 2 O. Page 1 of 1 Start over Page 1 of 1. Put about ½ inch of hydrated copper sulfate crystals in a dry test tube.
Older names for this compound include blue vitriol bluestone vitriol of copper and Roman vitriol. 99 Pure Copper Sulfate Pentahydrate Crystals. The hydrated form is medium blue and the dehydrated solid is light blue.
Canister Heavy-Duty Multi-Purpose Cleaning Wipes 25-Count 2-Pack 2 lbs. Stoichiometry Interpreting Chemical Equations ANY TIME YOU HAVE MOLE AND MOLE IN THE SAME FRACTION ALWAYS LOOK AT THE BALANCED CHEMICAL EQUATION Stoichiometry is used to convert from moles of one substance. RECORD the mass of the dish and hydrate to - 00010 grams.
The waters of hydration are released from the solid crystal and form water vapor. In many cases the uptake and loss of water by heating decreasing pressure or other means are reversible processes sometimes accompanied by changes in colour. LEAVE TUBE TIPPED DOWNWARD.
More often than not x is a whole number but keep in mind that there are some exceptions. Copper II sulfate pentahydrate. Formulas for ionic hydrates are written by appending a vertically centered dot a.
Copper Sulfate Large Crystals 50lb Bag - EPA 99 Pure. C 25 H 36 Li 4 N 7 O 19 P 3 S xH 2 O. Label the bottles with the normality of EDTA.
Upper Saddle River NJ. Observing our nitrate it has a white crystalline structure representing that similar to table salt. Gently heat the dish by moving the burner back and forth around the base.
CuSO 4 5H 2 Os heat CuSO 4 s 5H 2 Og If 363g CuSO 4 5H 2 O is decomposed to CuSO 4. Place the dish containing the hydrate on the wire gauze. Home and Office Detailer New Cup 3-Pack Price 10 22 19 99 20 48 25 21.
Each copper sulphate unit can attach to five water molecules so its sometimes called copper sulphate pentahydrate when its hydrated. Side effects of excess supplementation may include abdominal pain vomiting headache and tiredness. Ratings 24 17 2 3Concentrated or Ready to Use.
It has a role as a flame retardant a mordant and an astringent. Impact of Learners Prior Knowledge on Their Use of Chemistry Computer Simulations. Either weigh the EDTA analytically or standardize the solution.
CopperII sulfate also known as copper sulphate are the inorganic compounds with the chemical formula Cu SO 4 H 2 O x where x can range from 0 to 5. Record n your observations. In the case of copperII sulfate hydrates you only get x as a whole.
How can we experimentally determine the formula of an unknown hydrate A. Methylmalonyl coenzyme A tetralithium salt hydrate Synonyms. Formation of metallic copper In the fume hood add 20 g of powdered zinc metal stirring until any reaction is apparently complete.
The pentahydrate x 5 is the most common form. 47 out of 5 stars 45. Rewrite the chemical equation for this process including phase designations.
Laboratory-Grade Copper II Sulfate 5-Hydrate Powder 500g - The Curated Chemical Collection. The systematic name is sodium carbonate decahydrate.
Copper cupric oxide 16 mg. Go ahead and enjoy some dirty incest kinky fun with desi aunty sex.

Differences Between Cuprous Oxide Cu2o And Copper Cupric Oxide Cuo Nanoparticles By Nanografi Blografi Medium
Initial exposure to clean seawater is crucial to the long-term performance of copper-nickel alloys.

Cupric oxide is synthetic source of copper. Pomegranate is a tree. CopperII sulfate CuSO 4 is white in is anhyrous form and deep blue when it is complexed with water in the pentahydrate form CuSO 4 5H 2 O. It is also used in the copper vapor laser a class of laser where the medium is copper bromide vapour formed in-situ from hydrogen bromide reacting with the copper discharge tube.
3 General Industry Safety Order Section 5155. Many brands substitute or confuse the synthetic pigment for the natural one giving both the same Common names and CI. Approximately 5 10 of dietary iron is absorbed and this absorption rate increases to up to 30 in iron deficiency states.
This material sometimes called blue vitrol is used in fungicides and. CID 1118 Sulfuric acid CID 23978 Copper Dates. Polytetrafluoroethylene is very non-reactive and ideal for use with most chemicals.
Although treatment response is best assessed by repeat biopsy and Cu quantification serial monitoring of serum ALT is a useful surrogate monitoring strategy. Few examples of the non-enzymatic antioxidants are vitamin C vitamin E plant polyphenol carotenoids and. Oral iron supplements are absorbed up to 60 via active and passive transport processes.
Vitamin A β-carotene 28640 IU 17 mg 572. The rapid development of nanozymes for ultrasensitive detection of contaminate has resulted in considerable attention. 1 International Agency for Research on Cancer IARC.
The purpose of this paper is to describe the nutrients of interest for age-related vision health and to identify rich dietary sources of these nutrients. Uses of Copper Sulphate. Copper sulphate blue stone blue vitriol are all common names for pentahydrated cupric sulphate CuSO 4 5H 2 O which is the best known and the most widely used of the copper salts.
The film is complex and mainly composed of cuprous oxide often containing nickel and iron oxide cuprous hydroxychloride and cupric oxide. The time depends on the temperature. The Centers for Disease Control and Prevention CDC cannot attest to the accuracy of a non-federal website.
Five types of cobalt oxide have been identified including CoO CoO 2 CoOOH. Folic acid 04 mg Ascorbic acid 90 mg Biotin 45 mcg Calcium 200 mg Cholecalciferol 400 unit Chromium 10 mcg Copper 2 mg Cyanocobalamin 25 mcg Magnesium 100 mg Manganese 5 mg Molybdenum 25 mcg Nicotinamide 40 mg Pantothenic acid 10 mg Potassium Iodide 015 mg Pyridoxine hydrochloride 3 mg Riboflavin 32 mg Selenium 25. Names interchangeably PBr7 is the.
Academiaedu is a platform for academics to share research papers. We are a leading supplier to the global Life Science industry with solutions and services for research biotechnology development and production and pharmaceutical drug therapy development and production. Vitamin C also known as ascorbic acid is an important water-soluble vitamin.
A large randomized placebo-controlled trial of daily supplementation with antioxidants 500 mg of vitamin C 400 IU of vitamin E and 15 mg of β-carotene and high-dose zinc 80 mg of zinc as zinc oxide and 2 mg of copper as cupric oxide the Age-Related Eye Disease Study AREDS found that administration of high-dose zinc alone or with the antioxidant combination to individuals with. The analysis of CuO after and before reactions showed that the proportions of Cu and Cu 2 were 25 and 75 in comparison with 100 proportion of Cu 2 before the reaction time. Herein a carboxyl- and aminopropyl-functionalized copper organophyllosilicate Cu-CAP was synthesized by a facile one-pot solgel method.
Evaporation is the process of conversion of a a liquid to its gaseous state below the boiling point of the substance b a liquid to its gaseous state at the boiling point of the substance c a solid to its liquid state at the melting point of the substance d a solid to its liquid state below the melting point of the substance 18. Review the chemical compatibility of Teflon and PTFE with various chemicals solvents alcohols and other products in the cart below. People use pomegranate for high blood pressure athletic performance heart disease diabetes and many other.
4 California Department of Pesticide. The antioxidant enzymes convert dangerous oxidative products to hydrogen peroxide H 2 O 2 and then to water in a multi-step process in presence of cofactors such as copper zinc manganese and iron. Links with this icon indicate that you are leaving the CDC website.
The most common oxidation states of iron are Cu the copperI or cuprous ion and Cu 2 the copperII or cupric ion. CopperII chloride is the chemical compound with the chemical formula CuCl 2The anhydrous form is yellowish brown but slowly absorbs moisture to form a blue-green dihydrate. Considering metal oxide nanoparticles as important technological materials authors provide a comprehensive review of researches on metal oxide nanoparticles their synthetic strategies and techniques nanoscale physicochemical properties defining specific industrial applications in the various fields of applied nanotechnology.
624 The hormone hepcidin. Copper is an essential trace element that is included in some over-the-counter multivitamin and mineral supplements even though copper deficiency is quite rare and. Cupric oxides CuO and Cu 2 O and zero valent copper ZVC Cu 0 were considered for activation of PMS.
Copperassociated chronic hepatitis CuCH is treated by a multimodal approach that must include dietary copper Cu restriction removal of Cu from the liver by chelation with DPen and antioxidant treatment. This work expansively reviews the recent developments of. A Calcium oxide b Common salt c Ozone d Water 17.
17 Gastrointestinal absorption of iron occurs via strict regulation by the enterocyte and duodenal cytochrome and ferric reductase enzymes. 2 Environmental Protection Agency lists pursuant to the Clean Air and Clean Water Acts. Various parts of the tree and fruit are used to make medicine.
CopperII bromide Cu Br 2 is a chemical compoundIt is used in photographic processing as an intensifier and as a brominating agent in organic synthesis. Indeed it is often the starting raw material for. Both the anhydrous and the dihydrate forms occur naturally as the very rare minerals tolbachite and eriochalcite respectively.
The sources of each hazardous substance on the list is designated by a number in the middle column of the list. We are a leading supplier to the global Life Science industry with solutions and services for research biotechnology development and production and pharmaceutical drug therapy development and production. Vitamin C is available in many forms but there is little scientific.
Natural Brown Iron Oxide andor Calcined natural Iron Oxide usually with varying ratios of hydrated Iron Oxide to anhydrous Iron Oxide along with Manganese Oxide Aluminum silica clays and other natural minerals depending on mine site. The numbers represent the following sources. Linking to a non-federal website does not constitute an endorsement by CDC or any of its employees of the sponsors or the information and products presented on the website.
Non-enzymatic antioxidants work by interrupting free radical chain reactions. Producing yellow or green light it is used in. The bifunctional groups endow it with superior catalytic activity than that of natural enzyme.
The initial film forms fairly quickly over the first couple of days but takes 23 months to fully mature. PTFE Chemical Compatibility Chart.