Studies involving predominantly high-humidity 50 wetsteam sauna or immersion hydrotherapy were excluded for the potential confounding mechanisms of differential sweating rates and explicit focus of this review limited to dry sauna interventions. Avoid Hot Baths and Showers.
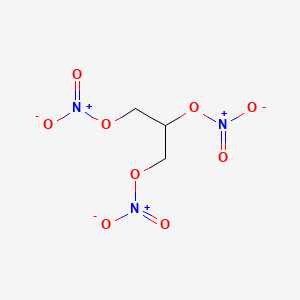
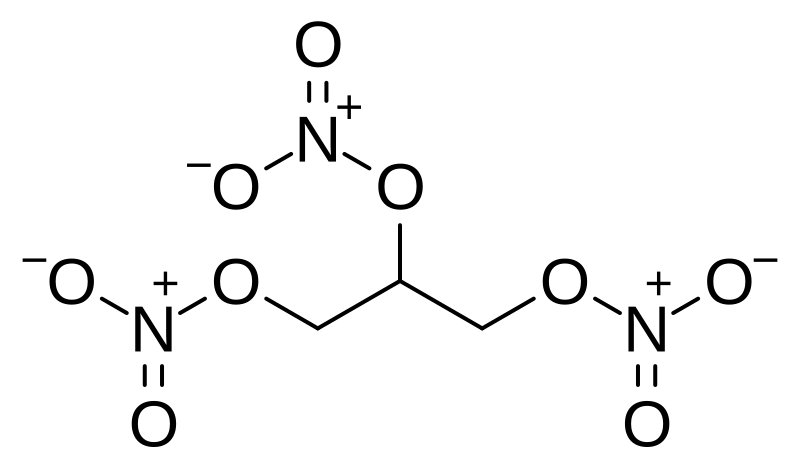
Nitroglycerin C3h5 No3 3 Pubchem
Increase by one-half inch to achieve desired results.
Nitroglycerin high pressure burn skin. By IV for 10 days reduces chest pain compared to intravenous nitroglycerin. This usually happens as a result of high blood pressure and blood pressure management is an important part of managing type 2 diabetesBlood sugar should be regularly monitored so that any problems can be detected and treated early. This medicine can ease spasms and help you heal.
Administer another nitroglycerin tablet. Histopathology confirms for example that extravasation of 10 calcium chloride which has an osmolarity of 2050 mOsmL is a true subcutaneous burn and as such involves both the overlying skin and the underlying fascia and skeletal muscle 17 p155 High osmolarity vesicants including phosphate and calcium also present a precipitation risk. Patient with ST elevation on ECG patients should receive immediate reperfusion therapy either pharmacologic thrombolytics or transfer to the catheterization.
20 Burn patients with burns less than _____ can be treated with moistened dressings for comfort. 1 inch 15 mg nitroglycerin. Jan 06 2010 The initial IMR powder was IMR4198 produced in the early 1930s.
Also called endothelium-derived relaxing factor. Give 04 mg sublingual tab wait 5 minutes if the chest pain is not relieved administer another dose. Teachertrainer will walk around room listening participating in discussion when.
How does it. Nitroglycerine is 60 bound to plasma protein at plasma concentrations between 50 and 500 ngmL. They can interact with some of the medications prescribed for treating hypertension high blood pressure pulmonary arterial hypertension HIV and other health conditions.
Nitroglycerin NG also known as nitroglycerine trinitroglycerin TNG nitro glyceryl trinitrate GTN or 123-trinitroxypropane is a dense colorless oily explosive liquid most commonly produced by nitrating glycerol with white fuming nitric acid under conditions appropriate to the formation of the nitric acid esterChemically the substance is an organic nitrate compound rather than. This is especially true if you have cuts or fissures. Gloves goggles Secondary EMT should state to ask PTs friend Vitals 1 2 P.
Have class members get into groups of 2-4 and share how each of the factors from slide 6 are important or affect exposure. This medication dilates the blood vessels to help allow any blood flow that might be impeded. Flushing can be a side effect of many drugs.
Discovery Inclusivity Innovation Integrity and Quality. 32 gr 48 gr. Hot baths and showers can dry your skin and make your condition worse.
Other injections may occur from the use of high pressure air or liquid such as in spray painting or from the rupture of high-pressure lines Discuss PowerPoint slides 5 and 6. The Association for Academic Surgery is widely recognized as an inclusive surgical organization. Ask your gastroenterologist about prescription medications like topical nitroglycerin.
Suddenly started having trouble speaking E. A drug that relaxes blood vessels and increases the supply of blood and oxygen to the heart while reducing its. Doses as high as 26 mg given qid have been used.
About the Societies. Coleus or forskolin are used for high blood pressure chest pain asthma dry eye and many other conditions but there is no good scientific evidence to support any of these uses. Nitroglycerin is readily absorbed through the mucous membranes lungs and intact skin.
Initial dose one-half inch q 8 hr. Some of these medications include nitrates nitroglycerin and alpha-blockers. Cancer Research UK is a registered charity in England and Wales 1089464 Scotland SC041666 the Isle of Man 1103 and Jersey 247.
Monitor a patients blood pressure hold for a systolic BP of less than 90 mmHg. Call the resuscitation team immediately. Treatment involves lifestyle changes such as eating a healthy and balanced diet and regular physical exercise.
Low blood pressure is a difficult clinical finding for a healthcare professional to address. Some heart and blood pressure medicines including calcium channel blockers and nitroglycerin and ED prescriptions like sildenafil Viagra. After checking the clients heart rate and blood pressure the nurse administers nitroglycerin 04 mg sublingually.
Early research suggests that taking a liquid garlic extract daily for one month reduces prostate mass and urinary frequency. Adjust to higher doses by using patches that deliver more drug or by applying more than one patch. While high blood pressure is known as the silent killer because it is associated with few acute symptoms hypotension hypolow tensionpressure may be normal for a patient if it is without symptoms but can be of great importance if it is associated with abnormal body function.
Our mission is to be the premier outlet for high-impact research in surgery. A compound produced by the endothelium the lining of the interior walls of arteries that helps widen blood vessels and counteract high blood pressure. These conditions include high blood pressure low blood pressure high cholesterol.
Learning curves in minimally invasive esophagectomy. Rosacea is a skin condition that may. It is a beta-blocker 3 Medical Sex.
Dos during heart attack. Up to 45 inches q 4 hr have been used. After 5 minutes the client states My chest still hurts If the vital signs have remained stable which action should the nurse perform.
The impetus of the membership remains research-based academic surgery and to promote the shared vision of research and academic pursuits through the exchange of ideas between senior surgical residents junior faculty and established academic surgical professors. A company limited by guarantee. Apply 10 L of oxygen via nasal cannula.
Apply one patch each day. An average plasma concentration of 23 ngml was observed one hour after dermal application of 45 mg of nitroglycerin as an ointment. Loosen any tight clothing.
M or F BSI. Ammunition is offered from many manufacturers which further increases its Cannons usually used lower burn-rate powders because most would burst with higher burn-rate powders. Tight space inside the resturaunt O.
Avoid in hypotensive patients and those with inferior ST elevation. This can happen 3 times total. Patient may complain of their discomfort as heart burn or nausea NTG Nitroglycerin ASA Aspirin atenolol A common blood pressure medication.
Benign prostatic hyperplasia. A systematic review and evaluation of benchmarking parameters. Patient should be made to sit down rest and try to keep calm.
Usual dose is 12 inches q 8 hr. Studies of partial body heating were excluded since proposed mechanisms of action may or may not be the same as whole-body heating. Still high blood pressure itself is not a cause of flushing according to the American Heart Association.
Keep the anus clean and apply a barrier cream to help soothe inflamed skin. Viagra and sildenafil do have some potential interactions. Ask if the patient takes any chest pain medic ation f or a known heart condition such as nitroglycerin and help him take it.
If lifestyle changes alone are not enough to. This is the initial medication given along with aspirin. But the quality of this research is questionable.
Skin exposed to chemicals should be flushed for at least ____ minutes. Feb 06 2021 Powder Burn Rate 4050 S. Nitroglycerin has shown a mortality benefit aim for 10 mean arterial pressure MAP reduction in normotensive patients and 30 MAP reduction in hypertensive patients.
If the pain does not go away with rest or within 3 minutes of taking nitroglycerin call for emergency medical help.
A much smaller amount of carbon approximately 1000 Pg is located near the ocean surface. And of course we are indirectly influencing the atmospheric water vapour levels by our CO2 methane nitrous oxide emissions.

Nitrous Oxide Gas Encyclopedia Air Liquide Air Liquide
The company Nitrous Oxide Systems NOS was born.

Vapor pressure of nitrous oxide. Huge amounts of lava and ash can spew from the volcano. 57 MPa 57 42753 20 Estimating vapor pressure from molecular structure. In ecological problems vpd is often regarded as a measure of the drying power of air because it plays an.
Ethylene oxide a monoepoxide is a gas at room temperature and normal atmospheric pressure. 2 A pressure relief device when installed must be in communication with the vapor space of a cylinder containing a Division 21 flammable gas material. Used to freeze.
In the event of a leak inert gases can quickly displace air in a large area creating an oxygen-deficient atmosphere toxic gases can create poison atmospheres and flammable or reactive gases can result in fire. Gas used in health care facilities and nitrous oxide which is used frequently as an inhalation anesthetic. The system increases the engines power output by allowing fuel to be burned at a higher-than-normal rate because of the higher partial pressure of oxygen injected with the fuel mixture.
Matter changes into a gas when temperature increases and pressure decreases. Compressed gases can be toxic flammable oxidizing corrosive or inert. Oxygen O 2 316.
The greenhouse effect happens when certain gases which are known as greenhouse gases accumulate in Earths atmosphere. The protection afforded by the atmosphere is very important for life on Earth. Some examples are SIMPOL1 method the method of Moller et al and EVAPORATION Estimation of VApour Pressure of ORganics Accounting for Temperature.
These hydrocarbons are commonly known as fossil fuels. Melting point from molecular weight - Calculation of melting point of hydrocarbons from molecular weight molar mass. Greenhouse gases allow the suns light to shine onto Earths surface and then the gases such as ozone trap the heat that reflects back from the.
Nitrous oxide can increase cerebral blood flow and cause increased intracranial pressure whereas volatile anesthetics can decrease cerebral metabolic rate decreasing cerebral blood flow to a point until reaching 15 MAC and resulting vasodilation can occur increasing the intracranial pressure. The chemi-cal composition of the atmosphere is given in Table 2-1. A nitrous oxide engine sometimes referred to as NOS is an internal combustion engine in which oxygen for burning the fuel comes from the decomposition of nitrous oxide N 2 O rather than air.
Its OK to spray nitrous to Q16. When you spray multiple kits you are introducing all the oxygen ie nitrous the engine can handle. The frequency of a sound wave is called it pitch.
They can contain solitary atoms ions and compounds. Water vapor 134 C. Carbon dioxide is one of the most important gases for all life on Earth because plants need it for photosynthesis.
Types of Volcanoes We usually think of volcanoes as tall mountains in the shape of a cone however there are different types of volcanoes. It wasnt until the late 1970s when two racers perfected its use. Nitrous Oxide N 2 O A powerful greenhouse gas with a global warming potential of 298 times that of carbon dioxide CO 2.
Greenhouse gases include carbon dioxide CO2 methane CH4 nitrous oxide N2O ozone O3 and fluorinated gases. A liquid having a vapor pressure exceeding 40 psi at 100 deg. C as determined by ASTM D-323-72.
167 157. The Earths oceans contain 38000 PgC most of which is in the form of dissolved inorganic carbon stored at great depths where it resides for long periods of time. This is where you want a fuel that will help reduce detonation and where VPs C23 is perfect.
The GWP is from the IPCCs. I wouldnt recommend multiple kits but single stages up to 500hp or so. Boils to give a colorless gas that is sweet-smelling and moderately toxic.
Both situations ameliorate with the use of IV anesthetics andor. O Deuterium oxide P 0 18 2 26 6 36 3 47 6 8 H 2 S Hydrogen sulfide 14 6 20 5 26 4 32 4 4 H 3 N Ammonia 25 1 37 2 53 1 68 6 9 He Helium P 0 74 7 118 3 155 7 189 6 221 4 251 6 10 Kr Krypton P 0 6 5 9 5 12 3 14 8 17 1 11 NO Nitric oxide 17 8 25 9 33 1 39 6. This requirement does not apply to DOT Specification 39 cylinders of 12L 75 cubic inches or less in volume filled with a Liquefied petroleum gas Methyl acetylene and Propadiene mixtures stabilized Propylene Propane or Butane.
Major sources of nitrous oxide include soil cultivation practices especially the use of commercial and organic fertilizers fossil fuel combustion nitric acid production and biomass burning. Biological materials v ms soft tissues. The greenhouse effect happens when certain gases which are known as greenhouse gases accumulate in Earths atmosphereGreenhouse gases include carbon dioxide CO 2 methane CH 4 nitrous oxide N 2 O ozone O 3 and fluorinated gases.
Gases can be pure elements compounds or mixtures. Racers dabbled with nitrous oxide injection from time to time with varied results. Oxygen is one of the most important gases for humans.
This carbon is exchanged rapidly with the. The saturation vapor pressure deficit of an air sample sometimes vapor pressure deficit vpd or just saturation deficit for short is the difference between the saturation vapor pressure and the actual vapor pressure ie. Cinder cones - These are volcanoes formed from.
Water vapor H 2O 0 to 4 by volume and still smaller amounts of carbon dioxide CO 2 methane CH 4 nitrous oxide N 2O ozone O 3 and dozens of other gases at still smaller concentrations. The gas has narcotic effects when inhaled laughing gas. When the pressure gets high enough the volcano can erupt with a huge explosion.
The effort paid off as NOS quickly became. The notation P 0. These gases are oxidizers that when present in sufficient quantity and concentration form one side of the fire triangle When the other two sides of the triangle heat and fuel are added fire andor explosion can result.
Probably an old version of the CRC. The vapor density is 149. Note that although mankinds water vapour emissions dont have a significnat effect on greenhouse levels of atmospheric water vapour we can probably influence the hydrological cycle by land use effects.
Ethylene oxide is very reactive with nucleophiles such as water alcohols halides amines and sulfhydryl compounds EPA. High frequency sounds are said to be high pitched or just high. A region of increased pressure on a sound wave is called a compression or condensation.
Gases have many uses. 5660 MPa 5660 42453 25 Carbon dioxide. The vapor is highly flammable at concentrations ranging from 3 to 100 and it may undergo explosive de composition WHO 1985.
An oxygenated fuel hasnt proved. Vapor pressure is at about 745 psig at 70F. During the first several years NOS spent most of its resources demonstrating that nitrous oxide was an efficient safe form of performance enhancement.
Nitrous oxide refrigerated liquid appears as a colorless liquid. 1 NTP - Normal Temperature and Pressure - is defined as 20 o C 29315 K 68 o F and 1 atm 101325 kNm2 101325 kPa 147 psia 0 psig 30 in Hg 760 torr Since specific gravity is the ratio between the density mass per unit volume of an actual gas and the density of air - specific gravity has no dimension. When this happens pressure can build up over a long period of time.
Several empirical methods exist to estimate the vapor pressure from molecular structure for organic molecules. Hydrogen - Thermophysical Properties - Chemical Physical and Thermal Properties of Hydrogen - H 2. KPa 1 bar or to the saturation vapor pressure if that is less than 100 kPa.
Hydrocarbones - Vapor Pressure - Vapor pressure versus temperature for propane n-butane n-heptane and n-pentane. Density 122 g cm3 at its boiling point of -89C.
Question from Student Questionschemistry. Vapour pressures of benzene hexafluorobenzene and naphthalene The Journal of Chemical Thermodynamics 1990 22 6.
Vapor Pressure Of Benzene From Dortmund Data Bank
Temperature K A B C Reference Comment.

Vapour pressure benzene. The solubility of naphthalene is closest to ideal for solvents that are similar to it eg. Benzol Phenyl hydride Benzene and mixtures having 10 benzene or more IBC code Name of the supplier. Solution A has vapour pressure of 7 5 4.
Phenylformic acid E210 Sodium benzoate E211 3113. 5 mm of Hg at the normal boiling point of water and solution B has the same vapour pressure at the normal boiling point of benzene. 65-85-0 200-618-2 Sodium benzoate.
However it has threetarget organs central nervous system kidney and hematopoietic system. Vapour Pressure of a Solute above a Solution Henrys Law. Heating the molecules of the liquid can help change them to the vapour phase and thus increase the vapour pressure of the liquid.
Vapour cartridge or canister may be permissible under certain circumstances where airborne concentrations are expected to exceed exposure limits. Temperature and Pressure. If the liquid is heated a little over 100 C the transition from liquid to gas will occur not only at the surface but.
So theres 566 mols of molecules for every 1L of solution one comes from glucose and 556 from water as calculated. The apparent enthalpy of fusion is different and the plot may become curved. The ebulliometric method of vapour-pressure measurement.
It has 6 carbon atoms joined in a ring and has 1 hydrogen atom attached to each of the carbon atoms. Q-Suggest two materials other than hydrogen that can be used as fuels in fuel cells. Lead is also a specific organ toxin.
Vapour may accumulate in hazardous amounts in low-lying areas especially inside confined spaces resulting in a toxicity hazard. Ammonia - Vapour Pressure at gas-liquid equilibrium. P A P A 0 x A and P B P 0 B x B as the total vapour pressure P A 0 x A P 0 B x B is greater than what it should be according to Raoults Law.
Identification SDS Record Number. Protection provided by air-purifying respirators is limited. Trade names and abbreviations 3114.
The pressure lowering of the water is PX as P stands for the pressure of pure solvent and X is the molar fraction of the solute. 1L of water has 1000g of water so there are 100018 mols of water 556 mols. Of solutions say vapour pressure with the concentration of the solution and quite useful in describing the calculations involving gas mixtures.
PCS 95004 Date of SDS. The concentration of a vapor in contact with its liquid especially at equilibrium is often expressed in terms of vapor pressure which will be a partial pressure a part of the total gas pressure if any other gases. At 100 C and atmospheric pressure equilibrium is not reached until the air is 100 water.
Online calculator with figures and tables showing density and specific weight of ammonia for temperatures ranging -50 to 425 C -50 to 800 F at atmospheric and higher pressure - Imperial. There are two effects when the difference between solute and solvent increases. It may be chemical physical or biological in.
CAS EINECS ELINECS number CAS EINECS Benzoic acid. The vapour pressure of the liquid increases with an increase in its temperature. Its quite an experience hearing the sound of your voice carrying out to a over 100 first year.
While naming the substituted benzene compounds we prefix the name of the substituent to the word benzene. 50 g of urea NH 2 CONH 2 is dissolved in 850 g of water. Structural formula Benzoic acid Sodium benzoate.
Benzene Product Description. Benzoic acid Benzene carboxylic acid. Glucose or sucrose are soluble in water but cyclohexane or benzene simple six membered ring compounds are insoluble in water.
The former results from. New Movies to Watch with Your Family this Thanksgiving. Temperature and Pressure - Online calculator figures and table showing density and specific weight of benzene C 6 H 6 at temperatures ranging from 5 to 325 C 42 to 620 F at atmospheric and higher pressure - Imperial and SI Units.
Figures and table showing ammonia saturation pressure at boiling point SI and Imperial units. 5 Toxicology Toxic agent or substance Toxic agent is anything that can produce an adverse biological effect. Common VOCs include ethanol formaldehyde benzene toluene and xylene.
Q-Vapour pressure of pure water at 298 K is 238 mm Hg. This percentage increases as the temperature goes up. What is the mole ratio of benzenepB150 torr and toluenepT50 torrin vapour phase if the given solution has a vapour pressure of 120 torr.
A solution of liquid toluene dissolved in liquid benzene has a benzene mole fraction of 850. Assume that we have 100 g of solution one can start with any amount of solution because the results obtained will be the same. Outdoor VOCs are usually emitted from industrial facilities and vehicles and.
Benzene is a hydrocarbon with the chemical formula C 6 H 6. Calculate the vapor pressure of the solution given that the vapor pressures of pure benzene and toulene are 183 mmHg and 592 mmHg respectively. 11 February 2015 Identity of the substance.
For example Acetone and benzene have higher vapour pressure than water at a particular temperature. Coefficents calculated by NIST from authors data. Benzene is a specific organ toxin in that it is primarily toxic to the blood-forming tissues.
In thermodynamics and chemical engineering the vaporliquid equilibrium VLE describes the distribution of a chemical species between the vapor phase and a liquid phase. VOCs are of concern to both indoor air quality and outdoor quality. Ammonia Gas - Density vs.
Chemistry 38 Calculate the mole fraction of ethylene glycol C 2 H 6 O 2 in a solution containing 20 of C 2H6O2 by mass. In this article we. Assuming X undergoes partial dimerization in benzene what is the percentage of X dimerized in benzene solution.
These liquids will show positive deviation when Raoults Law when. Petrochemical Corporation of Singapore. Use a positive-pressure air-supplied respirator if there is any potential for uncontrolled.
By replacing one or more of the hydrogen atoms with some functional group we get several benzene compounds. Liquid can accumulate static charge by flow splashing or agitation. The slope of the plot changes ie.
Take A Sneak Peak At The Movies Coming Out This Week 812 New Movie Releases This Weekend. Benzene SDS Page 1 of 11 SAFETY DATA SHEET SDS BENZENE 1. Besley and Bottomley 1974.
Closed containers may rupture. Benzene - Density and Specific Weight vs. So the solute molar fraction is 1566 176810-2.
Vapour may travel a considerable distance to a source of ignition and flash back to a leak or open container. Let the vapour pressure pure vapour pressure and mole fraction of component A be P A P A 0 and x A respectively and that of component B be P B P B 0 and x B respectively. At room temperature and pressure the water jar reaches equilibrium when the air over the water has a humidity of about 3.
Indoor VOCs are usually emitted from consumer products and building materials such as paints and carpets and may adversely impact the health of people that are exposed.
Acetic acid anhydride CH 3. The total gas flow across the bubbler should be as low as possible pressure and concentration of the absorbing substance.
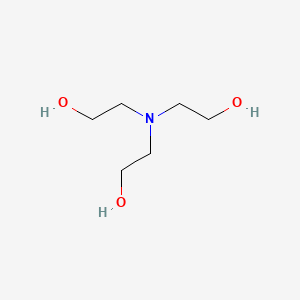
Triethanolamine C6h15no3 Pubchem
Roshan Shahid Zubair ZM.

Triethanolamine vapor pressure. Therefore the plasticizer should have low vapor pressure and a low rate of diffusion in the polymer. The boiling point is defined as the temperature at which the saturated vapor pressure of a liquid is equal to the surrounding atmospheric pressure. Production of Dimethyl Carbonate by Liquid Phase Oxidative Carbonylation of Methanol.
Manganese and potassium both ignite in the vapor Ann. Personal protection equipment should be chosen according to the ANSI standards and in discussion with the supplier of the personal protective equipment. HCFC means a hydrochlorofluorocarbon mentioned in Part 5 of Schedule 10.
Avoid breathing vapor or mist. - the Auto-Ignition Temperature is not the same as Flash Point - The Auto-Ignition Temperature indicates the minimum temperature required to ignite a gas or vapor in air without a spark or flame being present. Pressure Swing Distillation of Benzene and Ethanol.
Basic O-Ring chemical resistance compatibility information is based on isolated generic O-Ring material testing in optimal conditions at room temperature and pressure. MEA has the lowest boiling point and the highest vapor pressure of the amines. Then the amines are separated using a three-step.
Eye contact with concentrated solutions may cause severe eye damage followed by loss of sight. Of positions across the film. Draw vapor-liquid equilibrium VLE phase diagrams for single compounds or binary systems.
The front and back tubes contain molecular sieves impregnated with triethanolamine and the middle or oxidizer tube contains an inert support impregnated with a chromate salt. Handgun has the meaning given in subregulation 4F4. Fight fire from a safe distance with adequate cover.
102-71-6 Triethanolamine - 5 mgm3 TWA - ACGIH Exposure controls Engineering measures. Advice for firefighters. Repeated or prolonged exposures may cause darkening of the skin erosion of exposed front teeth.
A transition from predominantly Ns-doped to NsNi mixts to purely Ni-doped titania was obsd. Direct Insertion Probe. Government Engineering College Valsad.
The majority of plastic products are prepared by so-called hot compounding techniques where the formulated ingredients are combined under heat and shearing forces that bring about a state of molten plastic fluxing which is shaped into the desired product cooled and allowed to. It is important to use a water vapor trap before measuring the outlet gas flow to prevent measurement distortions due to the presence of water in the gas stream after the bubbling process. In accordance with the theory of maturing and aggregation the resulting clusters are susceptible to rapid collision with other clusters of zinc oxide leading to an appropriate concentration of the compound.
After the reaction the distillation of the resulting mixture is carried out first to remove excess ammonia and water. This results in MEA losses of 1-3. Diethanolamine often abbreviated as DEA or DEOA is an organic compound with the formula HNCH 2 CH 2 OH 2Pure diethanolamine is a white solid at room temperature but its tendencies to absorb water and to supercool meaning that it is often encountered as a colorless viscous liquid.
Across an anatase TiO2 thin-film. Siddaganga Institute of Technology Tumkuru. Large Protective Tube Cover.
Bhargav K V And Srivathsa M M. Kamani Parth Bhanushali Miral and Khan Aarif. Specific material compound formulations can.
Production of Methyl Isobutyl Ketone. Exposing rubber O-Ring materials to multiple chemicals and compounding application factors like temperature pressure and gland design can result in significantly different performance. High vapor concentrations may cause skin sensitization.
The boiling point at atmospheric pressure 147 psia 1 bar absolute for some common fluids and gases can be found from the table below. Recycling of flexible PUF from dumped car seating was scrutinized by DEG and potassium acetate KAc under atmospheric pressure at a constant temperature 215225 C. Flash point means the temperature at which petroleum and shale products including kerosene give off an inflammable vapor upon being tested by the Abel Pensky closed test apparatus.
As in any fire wear self -contained breathing apparatus MSHANIOSH approved or equivalent and full protective gear. Residue curves for ternaries. Wear appropriate respirator when ventilation is inadequate.
Cyclohexanone-formaldehyde resin produced when 1 mole of cyclohexanone is made to react with 165 moles of formaldehyde such that the finished resin has an average molecular weight of 600-610 as determined by ASTM method D2503-82 Standard Test Method for Molecular Weight Relative Molecular Mass of Hydrocarbons by Thermoelectric Measurement of Vapor Pressure. An important role in the proposed modification technique is played by TEOH which. Triethanolamine - 5-.
Diethanolamine is polyfunctional being a secondary amine and a diolLike other organic amines. 1atm 1 atm 760 mmHg. Development of hazardous combustion gases or vapor possible in the event of fire.
HANDLING AND STORAGE Handling Put on appropriate personal protective equipment see Section 8. UV-vis λ max 280 nm Refractive index n D 1485 Thermochemistry Heat capacity C 389 J K 1 mol 1. Triethanolamine Uranyl Acetate Dihydrate.
Skin absorption rates as blood concentration-time curves after dermal application of aqueous and neat 14Ctriethanolamine to mouse skin 2000 mgkg bw enclosed by a glass ring showed no significant change with the use of water as the vehicle. Vapor deposition cAPCVD was used to synthesize a film with gradating substitutional Ns and interstitial Ni nitrogen dopant concns. Put on appropriate personal protective equipment.
Exposure to vapor may cause intense watering and irritation to eyes. Temperature classification of equipment in hazardous areas are related. Diethanolamine is formed whereas with three moles of ethylene oxide triethanolamine is formed Fig.
Observe Occupational Exposure Limits and minimize the risk of inhalation of vapors. The Direct Insertion Probe DIP is widely used to introduce low vapor pressure liquids and solids into the mass spectrometer. This is important for analyzing temperature sensitive compounds.
To C 59To F - 32 Note. Eating drinking and smoking should be prohibited in areas where this. Prevent fire extinguishing water from contaminating surface water or the ground water system.
D03AX12 Hazards Safety data sheet. These studies demonstrated that the conversion of NCOO. Vapor pressure diagram p-T bubbledew point xy-T Pressure.
Vapor reacts with barium oxide incandescently Mellor 1940 Vol. Product Boiling Point at Atmospheric Pressure o C Acetaldehyde CH 3 CHO. We believe this to be the first time that such a.
A slow reaction between the vapor and formaldehyde became explosive near 180C Trans. Although the direct insertion probe is more cumbersome than the direct vapor inlet it is useful for a wider range of samples. The Zn 2 ions in reaction with triethanolamine produce clusters of ZnO on the silica surface.
Generating ternary map May take a few minutes Phase diagram. Std enthalpy of formation Δ f H 298 6657 6627 kJ mol 1. The effects of solvent catalyst temperature and the mass ratios on the reaction as well as the properties of the glycolysis products were investigated.
Folding stock has the meaning given in subregulation 4F4. The middle tube is not submitted to the lab for analysis but may undergo a color change indicative of depletion of the oxidizer. Std enthalpy of combustion Δ c H 298 38421 38391 MJ mol 1.
Direct Ionization of Sample. The slow phase half-lives for elimination of triethanolamine in mice after dermal exposure to 1000 and 2000 mgkg bw in acetone were 97 hr and 186 hr. 1 Pa at 20 C Acidity pK a 774.
Bell Maximum temperature Maximum pressure Maximum density Minimum temperature 1-303-497-6970 TP PD PH. Combustion can vary in degree of violence.

Identifications And Fundamental Constants Of O Xylene M Xylene Download Table
A deflagration is a propagation of a combustion zone at a velocity less than the speed of sound in.
Vapor pressure of o-xylene. Highly Flammable liquid and vapor Danger Flammable liquids H304 8545. O-Xylene is a colorless slightly oily flammable liquid. Volatile organic compounds VOCs are those organic compounds with a Reid vapor pressure of over 103 Pa at normal temperature 29315 K and pressure 101325 kPa.
A5307 B1930 K B. Huber 1-303-497-5252 CO2 Nitrogen Methane Ethane Propane Isobutane Butane Isopentane Pentane Hexane Pressure Output Water methane ethane propane XLS Version Number. Electronic cigarette vapor was analyzed for nicotine and impurities by trapping the vapor on thermal desorption tubes.
Laboratory-grade xylene is composed of m-xylene 4065 p-xylene 20 o-xylene 20 and ethyl benzene 6-20 and traces of toluene trimethyl benzene phenol thiophene pyridine and hydrogen sulfide. Therefore in order to provide reproducible and. However access to such an apparatus was not available for this study 7.
Vapor Vapor Density R32 R125 Enthalpy Z E Dr. The vaporliquid equilibrium calculations for tracing isotherms and isobars. A7059 B1930 K C.
Water Vapor - Steam H 2 O. May cause drowsiness or dizziness Warning Specific target organ toxicity single exposure. The VOCs are a large group of carbon-based chemicals that easily evaporate at room temperature Li et al 2009 Ojala et al 2011 Olsen and Nielsen 2001.
O-Xylene 0162 001 gL Vapor pressure 63 x 10-12 Pa 20C 21 x 10-11 Pa 25C Dissociation constant pKa 1088 071 Octanolwater partition coefficient KOW 20C Deionized Water 589 pH. Migration of volatile chemicals from the subsurface into overlying buildings is called vapor intrusion VI. METHOD 1501 Issue 3 dated 15 M arch 2003 - page 3 of 7 NIOSH Manual of Analytical Methods NMAM Fourth Edition 9.
롯데케미칼은 합성수지 기초유분 화성제품 건축인테리어 자재 등 다양한 제품 솔루션을 제공합니다. Acetone - Thermophysical Properties - Chemical physical and thermal properties of acetone also called 2-propanone dimethyl ketone and pyroacetic acid. Classic equation for vapor pressure calculation and correlation.
The xylenes m-xylene o-xylene p-xylene and ethylbenzene. Assuming that the variation of vapor pressure p with temperature T maybe described by the expression 𝑝 𝐵 log 𝐴 𝑘𝑃𝑎 𝑇 What are the values of A and B. Emergency or planned entry into unknown concentrations or IDLH conditions.
The finished polymers may only be used in contact with food of the types identified in 176170c of this chapter Table 1 under Categories I II. O - Xylene xylol. C-9 Vapor Pressure C-10 Enthalpy of Vaporization C-11 Enthalpy of Formation C-12 Gibbs Energy of Formation C-13 Solubility in Salt Water C-14 Solubility of Organic Compounds in Water as a Function of Temperature C-15 Henrys Law Constant for Gases in Water as a Function of Temperature C-16 Solubility of Selected Gases in Water as a Function of Temperature C-17 Solubility of Sulfur Compounds.
View help for this app Open units converter in separate browser window Toggle keyboard type between numeric and text useful on smartphone screens Select your preferred measuring units standard. Synonyms Trade Names 12-Dimethylbenzene ortho-Xylene o-Xylol CAS No. Location of the VBA code Dr.
At levels not to exceed 01 percent by weight of olefin polymers complying with 1771520c of this chapter. Volatile organic chemicals in contaminated soils or groundwater can emit vapors which may migrate through subsurface soils and may enter the indoor air of overlying buildings. Histopathological technicians who routinely come in contact with xylene-contaminated solvents in the workplace are the population most likely to be exposed to high levels of xylene.
O-Xylene Revision Date 31-Jul-2019 pH Not applicable Melting PointRange-25 C -13 F Boiling PointRange 143 - 145 C 2894 - 293 F Flash Point 31 C 878 F Evaporation Rate 07 Flammability solidgas Not applicable Flammability or explosive limits Upper 67 vol Lower 09 vol Vapor Pressure 882 Pa 25 C Vapor Density 37. APF 10000. A7059 B2100 K D.
And other researchers have used smoking machines eg Teague TE-2 Borgwaldt RM20S to generate and collect e-cigarette aerosols. The highest concentration of a gas or vapor percentage by volume in air above which a flame will not spread in the presence of an ignition source arc flame or heat. Caustic soda can be stored in A.
Narcotic effects Precautionary Statement Codes. Table 1 lists the physicochemical and thermodynamic properties. Concentrations higher than UEL are too rich to burn.
The density of air at. The siloxanes octamethylcyclotetrasiloxane decamethylcyclopentasiloxane. Det er mine desorption efficiency DE at least once for each batch of charcoal used for sampling in the.
1 NTP - Normal Temperature and Pressure - is defined as 20 o C 29315 K 68 o F and 1 atm 101325 kNm2 101325 kPa 147 psia 0 psig 30 in Hg 760 torr Since specific gravity is the ratio between the density mass per unit volume of an actual gas and the density of air - specific gravity has no dimension. O-Xylene ortho-xylene is an aromatic hydrocarbon with the formula C 6 H 4 CH 3 2. Commercial or mixed xylene usually contains about 40-65 m-xylene and up to 20 each of o-xylene and p-xylene and ethylbenzeneXylenes are released into the atmosphere as fugitive emissions from industrial sources from auto exhaust and through volatilization from their use as solvents.
The vapor pressure of toluene is 6811 kPa at 310 K and 2415 kPa at 340 K. Building depressurization may cause these vapors to enter the home through cracks in the foundation. A5307 B2100 K 49.
Material Properties - Material properties for gases fluids and solids - densities specific heats viscosities and more. A value for K of 511. With two methyl substituents bonded to adjacent carbon atoms of a benzene ring the ortho configuration.
181314-48-7 For use only. You want to calculate. 57-Bis11-dimethylethyl-3-hydroxy-23H-benzofuranone reaction products with o-xylene CAS Reg.
To further explain the meaning of K lets look at two extremes in Table 1. May be fatal if swallowed and enters airways Danger Aspiration hazard H336 1455. O-Xylene 131 Dichloromethane 331 111-Trichloroethane 147 Tetrachloroethylene 127 n-Hexane 0043 Cyclohexane 0040 Table 1 shows values of K for a range of compounds in water-air systems at 60 C 2 3.
The FAMES fatty acid methyl esters ie biodiesel constituents methyl oleate methyl palmitate methyl stearate methyl linoleate and methyl linolenate. It is a constitutional isomer of m-xylene and p-xylene the mixture being called xylene or xylenes. 글로벌 종합화학기업 롯데케미칼 제품 사이트에 오신 것을 환영합니다.
APF 25 Any powered air-purifying respirator with organic vapor cartridges APF 10 Any supplied-air respirator APF 50 Any self-contained breathing apparatus with a full facepiece.