CuH 4 O 2. 132 mgL CaCO3 In soil copper sulfate is partly washed down to lower levels partly bound by soil components and partly oxidatively transformed.
How Do You Know When Copper Ii Sulfate Hydrated Water Evaporates Socratic
Many compounds exist as hydrates.

Copper sulfate hydrate formula. More often than not x is a whole number but keep in mind that there are some exceptions. Put about ½ inch of hydrated copper sulfate crystals in a dry test tube. This represents the formula SnF 2 which is more properly named tinII fluoride.
1 Cu6355 gmol 1 S3207 gmol 4 O1600 gmol 15962 gmol Formula mass 15962 gmol 5 H 2 0 1802 g H 2 0mol 24972 gmol 2. For a general form copperII sulfate hydrate formula CuSO_4 xH_2O we use x to represent the number of water molecules present as water of crystallization per formula unit of anhydrous copperII sulfate CuSO_4. The empirical formula of a copper chloride hydrate is determined.
Each copper sulphate unit can attach to five water molecules so its sometimes called copper sulphate pentahydrate when its hydrated. It has also been used as a fungicide and herbicide. The systematic name is sodium.
Ammonium Sulfate Ammonium ions and Sulfate ions NH 4 and SO 4 2 NH 4 2SO 4 Calcium Phosphate Calcium ions and Phosphate ions Ca2 and PO 4 3 Ca 3PO 2 Waters of hydration use table to left copperII sulfate pentahydrate CuSO 45H 2O 11 Undeca. When copper sulfate CuSO4 reacts with water H2O the product is still copper sulfate. This is accomplished.
In our example 05 moles of water 01 moles copper sulfate 51 ratio. Page 1 of 1. Suspended in the water column in the bed.
CopperII sulfate also known as copper sulphate are the inorganic compounds with the chemical formula Cu SO 4 H 2 O x where x can range from 0 to 5The pentahydrate x 5 is the most common form. 3 Chemical and Physical Properties. In this case heating the hydroxide is able to drive off the water from the solid creating the oxide.
Sorption to these materials. 1 Structures Expand this section. Determining the Formula of a Hydrate.
Since the only component other than H2O and Cu 2 is the sulfate ion SO 4 2 we can now determine the complete formula of the hydrated copperII sulfate. A Calculate the percentage purity of the salt. Copper Sulfate Fine Crystal.
When the chemical formula for a hydrated ionic compound is written the formula for the ionic compound is separated from the waters of hydration by a centered dot. The formula of the hydrated form is CuSO 4. Allow the test tube and its contents to cool.
The most common form includes water of crystallization as the heptahydrate with the formula Zn SO 4 7H 2 O. If you consider the formula of copperII hydroxide and remove from it one O atom and two H atoms one water molecule you will see that what remains is the formula for copperII oxide. What is the empirical formula of this compound.
The latexcdotlatex indicates that the water is loosely bonded to the ionic compound. Zinc sulfate is an inorganic compoundIt is used as a dietary supplement to treat zinc deficiency and to prevent the condition in those at high risk. Copper 2 sulfate pentahydrate.
Calcium Carbide - CaC 2. A hydrate is an ionic compound that has a definite amount of water molecules attached to its crystalline structure. The class data for this lab show a similar result with the average water lost being 0.
In hydrated CuSO4 the water molecules surrounding the Central metal Cu function as ligands which bring d-d transition and hence emits blue colour in visible. TIP THE TEST TUBE SLIGHTLY DOWNWARD 2. Molecular FormulaCuO4S5H2O Molecular Weight24968.
CID 962 Water CID 23978 Copper Dates. Page 1 of 1. Melting Point C Physical Form.
Divide the number of moles of water lost by the number of moles of anhydrous salt to get the ratio of water molecules to formula units. Support Customer Support Contact Us FAQ Safety Data Sheets SDS Certificates COACOO Quality Regulatory Calculators Apps Webinars. Copper sulfate - CuSO 4.
Side effects of excess supplementation may include abdominal pain vomiting headache and tiredness. Mannitol - C 6 H 14 O 6. Monosodium Glutamate - C 5 H 8 NO 4 Na.
CopperII sulfate forms a hydrate which contains 361 by mass water. Label the bottles with the normality of EDTA. In its hydrate form copper sulfate turns a vivid blue color and has been used as a coloring ingredient in paintings and pottery.
Copper has a strong affinity for hydrous iron and manganese oxides clays carbonate minerals and organic matter. However it is the hydrated form of the salt and has 5 water molecules. 2 Names and Identifiers Expand this section.
Greek prefixes are attached to the word hydrate to indicate the number of water molecules per formula unit for the compound eg BaOH 2 8H 2 O. Calcium Chloride - CaCl 2. Washing soda is the common name for a hydrate of sodium carbonate containing 10 water molecules.
An anhydrate is the substance that remains after the water from a hydrate has been removed through heating. The dot after the formula for copper sulphate indicates bonds with water molecules. For example CuSO 4 5 H 2 O is copperII sulfate pentahydrate The notation of hydrous compound nH 2 O where n is the number of water molecules per formula unit of the salt is commonly used to show that a salt is hydrated.
Divide the mass of water in one mole of the hydrate by the molar mass of the hydrate. A copper acetate hydrate CuxC2H3O2y zH2O compound is found to contain 3182 copper 5914 acetate and the remainder is water. The calculated mass of water lost from the compound is 0.
And ends with hydrate For example the anhydrous compound copperII sulfate also exists as a hydrate containing five water molecules and named copperII sulfate pentahydrate. 0083 mol MgSO4 120. The ratio of water mol ecules to the anhydrous salt is constant.
The pentahydrate CuSO 4 5H 2 O the most commonly encountered salt is bright blue. Analytical 63 ACS reagent 44 Puriss 35 Cell Culture 21 Reagent 18 Purum 17 Anhydrous 13 BioXtra 13 ReagentPlus 11 Plant 7 Show More. Some examples are copper II sulfate sodium sulfate nickel II sulfate iron II sulfate cobalt II chloride magnesium sulfate sodium.
The steps you need to take in order to. CopperII hydroxide can therefore be called a hydrate of copperII oxide. Thus the product is called copper sulfate pentahydrate.
Why is CuSO4 blue. You can obtain your crystals while your crucible is cooling. These compounds often come in the form of a crystal which can then be heated in order to remove the water in the form of steam.
Hydrocyanic acid - HCN. In the case of copperII sulfate hydrates you only get x as a whole. CH 3 2 CC 6 H 4 OH 2.
Tetrachloroethylene - C 2 Cl 4. Hydrates are compounds that contain water with a definite mass in the form of H_2O in their molecular formula. How can find the relative number of moles of salt and water in the hydrate.
Older names for this compound include blue vitriol bluestone vitriol of copper and Roman vitriol. Have students graph their results. When determining the formula mass for a hydrate the waters of hydration must be included.
In its hydrate form cobalt chloride is purple while its. Cobalt ll chloride hexahydrate. 8 water molecules octahydrate.
What is the percent water in copperII sulfate pentahydrate. Hold it over the flame with a test tube holder and heat it for a few minutes with the mouth of the tube slightly below the base of the tube. Polystyrene - C 8 H 8 n.
Research suggests that the nature of these bonds is not as simple as. Sulfuric acid copperII salt 11 pentahydrate. Either weigh the EDTA analytically or standardize the solution.
Mg Immune Support Blend from ferment media European Elder. In aqueous solution an octahedral complex ZnH 2 O 6 2 is the predominant species.

Zinc Sulfate Chemistry Project Home
Contains sodium lauryl sulfate which may cause skin reactions particularly in children with eczema.

Is zinc sulfate aqueous. This is a single displacement reaction of a non-metal by a metalThe zinc will react with sulphuric acid forming zinc sulphate dissolved in solution and releasing hydrogen gas. However the conventional aqueous electrolytes are not capable of working at low temperature. NH 4 OH was added to the emulsion in order to obtain the zinc cation.
With a mild-acidic triflate electrolyte here the authors show a high. Ionic bonds also melt at high temperatures. Lactic acid Potassium sulfate Urea Lactose 1-Propanol Zinc sulfate REFERENCES 1.
Benefiting from high safety low cost and competitive energy density aqueous zinc-ion batteries AZIBs have become a very promising technique for grid-scale energy storage. However they often require the use of a surfactant to ensure ease of handling and suspension in aqueous solutions. The Journal of Endodontics the official journal of the American Association of Endodontists publishes scientific articles case reports and comparison studies evaluating materials and methods of pulp conservation and endodontic treatmentEndodontists and general dentists can learn about new concepts in root canal treatment and the latest advances in techniques and instrumentation in the one.
Here we report a frigostable cost-effective safe and eco-friendly hybrid electrolyte with high. The resulting ZnO calcinates were analyzed by XRD IR and. Zinc as zinc oxide from ferment media 15.
Villalpando S Rivera JA King JC. Zinc administered in aqueous solutions to fasting subjects is absorbed quite efficiently at a rate of 60-70. The component that does the dissolving Generally the component present in the greatest quantity is considered to be the solvent.
Ionic bonds are atomic bonds created by the attraction of two differently charged ionsThe bond is typically between a metal and a non-metal. Genetically modified plants for improved trace element nutrition. Skin barrier disruption by sodium lauryl sulfate-exposure alters the expressions of involucrin transglutaminase 1 profilaggrin and kallikreins during the repair phase in human skin in vivo.
Aqueous solution of ammonium hydroxide. 3 When dissolved sodium hydroxide reacts with sulfuric acid aqueous sodium sulfate water and heat are formed. V Aqueous Solutions and Body Fluids Hoeber 1966.
The structure of the bond is rigid strong and often crystalline and solid. Sıhnel O and Novotny P Densities of Aqueous Solutions of Inorganic Substances Elsevier Amsterdam 1985. In aqueous solution all forms of zinc sulfate behave identically.
Aqueous Solutions Solution - a homogeneous mixture Solute. A white coating appears on the copper electrode almost immediately and after a. Refer to sizes for USA standard sieves ASTM E11 for ranges of particle sizes.
Mineral form of basic copper carbonate. The absorption of zinc from soluble zinc acetate zinc sulfate. The development of rechargeable aqueous zinc batteries are challenging but promising for energy storage applications.
The component that is dissolved Solvent. Bioavailability of zinc from the various forms was compared on. The volatilization of zinc in combination with zinc chloride at temperatures above 285 C indicates the formation of Zn 2 Cl 2 a zinc compound with a 1 oxidation state.
Aromatic spirit of ammonia. Cardio Support Blend Organic Fenugreek seed from ferment media Organic Oregano leaf from ferment media Grape Seed extract Organic Hawthorn berry leaf and flower from ferment media. With a reduction potential of 076 V zincII reduces only with difficulty.
2-88 2-25 Partial Pressures of H 2 O over Aqueous Solutions of Sodium Carbonate. When heated above 680 C zinc. And insoluble zinc oxide was compared in ten human volunteers who were dosed orally with 50 mg Zn in various forms separated by two weeks intervals.
Similar to other Zn 2 H synergistic intercalative batteries zinc hydroxide sulfate ZHS was also found to be formed in the Znpoly44-TDP system caused by the precipitation of residual OH with Zn 2. Copper as copper sulfate anhydrous from ferment media 15. Solid polymer spheres in dry powder form.
Aqueous solutions are those in which water is the solvent. Reported a novel aqueous Zndiquinoxalino 23-a23-c phenazine ZnHATN battery which was enabled by the reversible H uptakeremoval behavior via. Solute Mass mmol kg-1 cmol L-1 ρg cm-3 n C ηmPa s Acetic acid 05 0084 0083 09989 13334 016 1012 CH3COOH 10 0168 0.
ZnSO 4 BaCl 2 BaSO 4 ZnCl 2. Zinc sulfate is also obtained as a hexahydrate ZnSO46H2O and as a heptahydrate ZnSO47H2O. Barium sulfate forms when these solutions are treated with solutions of barium ions.
To address this issue this work employs cotton-derived cellulose film prepared by a facile. 2 NaOH aq H 2 SO 4l Na 2 SO 4 2 H 2 O l Double Replacement 4 When fluorine gas is put into contact with calcium metal at high temperatures calcium fluoride powder is created in an exothermic reaction. A dark coating of copper metal appears on the zinc within two minutes and when 45 minutes have elapsed there is a thick coat of copper metal powder on the zinc strip and the blue color of the solution has lightened considerably.
Aqueous zinc-based energy storage ZES devices are promising candidates for portable and grid-scale applications owing to their intrinsically high safety low cost and high theoretical energy density. These aqueous solutions consist of the metal aquo complex ZnH 2 O 6 2 and SO 2 4 ions. 2-24 Total Vapor Pressures of Aqueous Solutions of NH 3.
No compounds of zinc in oxidation states other than 1 or 2 are known. Banana oil artificial isoamyl acetate. Custom color and density polyethylene microspheres can be manufactured by request.
To stabilize the water-in-oil emulsion the surfactant Span 80 was added to the heptane. When zinc metal is immersed in a solution of 01 M aqueous copperII sulfate solution c opper metal plates out on the zincThe solution is initially blue in color. Free radicals are highly reactive and unstable chemicals generated during normal body activities that require oxygen eg respiration digestion blood circulation immune system response increased physical activity etc and after exposure to UV.
Zinc administered in aqueous solutions to fasting subjects is absorbed efficiently. However the life span of AZIBs is severely influenced by the uncontrolled zinc dendritic growth and undesirable side reactions. All forms are soluble in water.
Recently Niu et al. Immediate steps should be taken to limit its spread to the environment. The precipitate was dried and then calcined at 7001000 C.
Vitamin C and zinc together help the bodys natural defense against damaging free radicals antioxidant effect and help boost immune function. A zinc electrode and a copper electrode are placed in a beaker containing an aqueous solution of zinc sulfate ammonium citrate and ammonium chloride. The aqueous phase of the system was zinc acetate dissolved in de-ionized water and the organic phase was heptane.
The primary hazard is the threat posed to the environment. Calculations indicate that a zinc compound with the oxidation. Zinc reacts with dilute sulphuric acid to form zinc sulphate and hydrogen gas is evolved.
Zinc absorption from zinc oxide zinc sulfate zinc oxideEDTA or sodium-zinc EDTA does not differ when added as fortificants to maize tortillas. Anhydrous zinc sulfate is a colorless crystalline solid. The electrodes are connected to a DC power supply and a voltage is applied - the negative lead on the copper electrode and the positive lead on the zinc electrode.
Rechargeable zincmanganese dioxide batteries that use mild aqueous electrolytes are attracting extensive attention due to high energy density and environmental friendliness.
The risk or severity of neuromuscular blockade can be increased when Magnesium sulfate is combined with Gentamicin. Atropine gr 1100 IM on call preoperatively Supply.
63323017302 Henry Schein Medical
When gentamicin is administered by intravenous IV infusion over a two-hour period the serum concentrations are similar to those obtained by IM administration.

Gentamicin sulfate im. Gentamicin may be administered IVIM. There is limited data regarding the safety of taking. Highly nephrotoxic - give with SCIC fluids.
Der Arzneistoff setzt sich aus mehreren Einzelverbindungen der Stoffgruppe der Gentamicine zusammen. To ensure that the collagenase enzyme in SANTYL maintains activity in vitro testing was performed across a wide variety of commonly used products to determine compatibility. May cause pain on IM injection.
Heart Infusion HI Broth and Agar HIA for Vibrio M61. Monitor peak 4-12 mgL and trough 1-2 mgL Monitor nephrotoxicity neurotoxicity and ototoxicity. The doctor prescribes 100 mg of gentamicin IM.
2 Xylocaine DENTAL with Adrenaline epinephrine 180000 Solution for injection. Hektoen Enteric HE Agar. CSF concentrations of gentamicin following intrathecal administration depend on the dose administered the site of injection the volume in which the dose is diluted and the presence or absence of obstruction to CSF flow.
The vial available contains 40 mgmL. How much gentamicin should you give. The medication is poorly absorbed.
Members Contact Us. Doxycycline is recommended as an alternative to azithromycin. Gentamicin is a bactericidal aminoglycoside that was discovered and isolated from Micromonospora purpurea in 1963.
The effect it may have on the baby is still unknown. Assess at beginning of therapy and. Obat ini bekerja dengan cara mengganggu.
7 It is one of the most frequently prescribed aminoglycosides due to its spectrum of activity low cost and availability. May need to reduce dose in renal. Die ersten Gentamicine wurden von Mitarbeitern von Schering in New Jersey im Jahr 1963 in den Produkten des Bakterienstamms Micromonospora.
Generic Name Gentamicin DrugBank Accession Number DB00798 Background. Infuse over 30-120 min when administering IV. Gentamicin equivalent IM or IV Use.
After intramuscular IM administration of Gentamicin Sulfate peak serum concentrations usually occur between 30 and 60 minutes and serum levels are measurable for six to eight hours. Gentamicin is distributed into cerebrospinal fluid CSF in low concentrations following IM or IV administration. Medicines Data Sheets 09 Sodium Chloride Solution for infusion 09 Sodium Chloride Solution for infusion.
Previous studies have shown 17 22 the positive outcomes when using SANTYL Ointment in conjunction with other wound care products. Perlu diketahui obat ini tidak bisa digunakan untuk mengobati infeksi akibat virus atau jamur. 100-200 mgkg IV as 1-3 solution.
Dosing regimens are numerous and are adjusted based on CrCl and changes in volume of distribution as well as on the body space where distribution of the agent will occur. Administer total dose within 1 hr one-half within first 15-20 minutes. IV IM Children 510 yr.
SC injection probably less painful. Paromomycin topical cream with or without gentamicin is an effective treatment for ulcerative cutaneous leishmaniasis according to the results of a phase-3 randomized double-blind parallel groupcontrolled trial. Search Children and Young Peoples Services MKN.
04 mg per mL Give. The absorption of Gemifloxacin can be decreased when combined with Magnesium sulfate. Is kept in freezer and must be brought to room temperature before injecting.
Z 18A Z Z BLACK 431C CrossTech72322Proof A2 Form M01 Fresenius Kabi USA LLC PO. 2 mL Rx only Single Dose Vial Reference ID. Aluminum sulfatecalcium acetate otc benzoin compound 30isopropyl alcohol 448 spray otc.
Gentamicin 75 mgkg IM or IV once a day for at least 5 days. Digitalizing dose 1530 mcgkg given as 50 of the dose initially and one quarter of the initial dose in each of 2 subsequent doses at 612 hr intervals. Morphine sulfate 10 mg per mL Give.
22 mgkgdose PO twice daily on day 1 then 22 mgkgdose PO once daily. Digitalizing dose 2535 mcgkg given as 50 of the dose initially and one quarter of the initial dose in each of 2 subsequent doses at 612 hr intervals. As it is more widely available it can be used in children who.
Gentamicin termasuk golongan antibiotik aminoglikosida. 100 mgkg IM q4-6hr PRN or 20-40 mgkg IM PRN. Magnesium sulfate is a drug used to treat convulsions during pregnancy nephritis in children magnesium deficiency and tetany.
Collagenase SANTYL Ointment compatibility with commonly used wound care products. 114 mg IM then 2nd dose in 24 hours o. Aluminum salts aluminum hydroxide aluminum phosphate or aluminum hydroxyphosphate sulfate AS01 B 3-O-desacyl-4-monophosphoryl lipid A MPL Quillaja saponaria Molina fraction 21 QS-21 cholesterol dioleoyl phosphatidylcholine DOPC disodium phosphate anhydrous potassium dihydrogen phosphate sodium chloride water for injection AS04 3-O-desacyl-4-monophosphoryl lipid A.
IV IM Children 25 yr. O Lincomycin OR clindamycin 600 mg IV every 8 hours and o Gentamicin 5 mgkg IV daily and o Metronidazole 500 mg IV every 12 hours If labour does not ensue and no evidence of chorioamnionitis and membranes intact then. Heart infusion agar HIA Difco M60.
For active tuberculosis it is often given together with isoniazid rifampicin and pyrazinamide. Gentamicin ist ein Aminoglycosidantibiotikum das als Arzneistoff in Form seines Sulfatsalzes bei bakteriellen Infektionen eingesetzt wird. An aminoglycoside such as gentamicin may be added if no improvement in first few days or if patient is HIV-infected.
_____ mL 16. Helvetica Neue A Helvetica A Typesmiths Pi Font m NDC 63323-010-02 1002 GENTAMICIN INJECTION USP 40 mgmL Gentamicinequivalent For. There may be considerable interpatient variation in.
May cause hypersensitivity reactions diarrhea granulocytopenia thrombocytopenia mild azotemia. Be diluted for IV use. O Gentamicin 5 mgkg IV daily o Metronidazole 500 mg IV every 12 hours If penicillin hypersensitivity and chorioamnionitis.
Magnesium sulfate has a better safety profile in the management of acute severe asthma than aminophylline. Morphine sulfate gr 16 IM q3-4h prn. Amikacin amiglyde sulfate 5 mgkg IM then 25 mgkg IM q 72 hr.
Gentamicin adalah obat untuk mengatasi infeksi bakteri ringan hingga berat pada berbagai bagian tubuh mulai dari telinga luar mata kulit hingga otak. Intravenous magnesium sulfate may provide additional benefit in children with severe asthma treated with bronchodilators and corticosteroids. Amikacin sulfate gentamicin so4 neomycin sulfate streptomycin sulfate tobramycin sulfate entecavir ranibizumab tiotropium am350 grnlrcnst-oral clindamycin phosphate am500 capreomycin inj cycloserine ethambutol hcl ethionamide isoniazid isoniazidrifampin pyrazinamide rifampin am550 am600 am650.
It is administered by injection into a vein or muscle. Children 8 years and older and Adolescents weighing less than 45 kg. For pregnant and lactating patients use erythromycin or azithromycin.
58 Gentamicin is effective against both gram-positive and gram-negative organisms but. Streptomycin is an antibiotic medication used to treat a number of bacterial infections including tuberculosis Mycobacterium avium complex endocarditis brucellosis Burkholderia infection plague tularemia and rat bite fever. In patients with normal renal function peak.
WLNA and WLDA Agar plates. If you are using a video camera start recording.

6kg Potassium Sulphate Powder Fire Extinguisher Code 25064 1 233 B C Uni En 3 7
物質名ppowdered 粒子密度ρpgcm3 かさ密度gcm3 圧縮度 安息角 スパチュラ角 Carr指数流動性噴流性 疎充填 密充填 ア 亜鉛粉 zinc p.
Fire extinguisher aluminum sulfate. Never boil your solution in a kitchen outdoors or in a non-chemically designed ventilation area. 425 -45 g cm-3. Aluminum sulfate may be used as a deodorant.
How to make tannerite more powerful. Even though a small fire that has just started can sometimes be extinguished with a laboratory fire extinguisher attempt to put out such fires only if you are trained to use that type of extinguisher confident that you can do it successfully and quickly and from a position in which you are always between the fire and an exit to avoid being trapped. Sulfuric acid primarily industrial use is in the production of fertilizers ammonium sulfate and superphosphate.
It has superior hardness and has a sharp edge. Pour your NaOH solution into an appropriate container. NFPA 704 Sulfuric Acid Signs.
And 1025µm pcd inserts is suitable for machining si12 high silicone aluminum alloy and tungsten carbide. Alert regarding hydrogen balloon explosions. 1 First tear off the small lead and pull out the insurance pin.
Before people used aluminum they used alum which is an aluminum sulfate that can be derived from the same minerals in which aluminum is found. Do not attempt to extinguish fires of any. When baking soda or sodium Bicarbonate NaHCo3 reacts with water carbonic acid is formed The reaction is exothermic that is heat and Carbonic acid are produced.
Most metal hydrides react violently with water. Most fire authorities recommend that all establishments have an ABC fire readily on hand. Fission And Fusion Difference.
Metal hydrides and pyrophorics air or water reactive Examples. 714 298 362 2555 亜鉛華 zine oxid 56 0567 1263 55. Sodium Hydroxide is used in the manufacture process of soaps paper aluminum and more.
As a safety precaution make sure to have a container of water with you. NaOH is extremely basic and can cause severe chemical burns if it contacts your skin. This reaction was the basis of an early fire extinguisher invented by Aleksandr Loran in 1904.
Carbonic acid is unstable so breaks up into carbon dioxide fizzy or effervescence and water. Sodium borohydride calcium hydride lithium aluminum hydride. It is best used against copper hard rubber wood.
Manganese sulfate monohydrate MnSO4H2O or H2MnO5S CID 177577 - structure chemical names physical and chemical properties classification patents literature biological activities safetyhazardstoxicity information supplier lists and more. How to use portable dry powder fire extinguisher. Dont use aluminum to boil your solution or the dish will melt.
Barium sulfate appears as white or yellowish odorless powder or small crystals. They used alum to dye fabric. Public health information CDC Research information NIH SARS-CoV-2 data NCBI Prevention and treatment.
You may find that laying the. Hydrogen Balloons Latex 1 Responses. Have an adult use a match to light the solid fuel tablet.
Food Chemistry Impact Factor. The bigger of pcd material grain size the better of wear resistance and the cutting edge will. A fire extinguisher would usually have a fire rating sticker clearly stated on its.
Make sure a Type D fire extinguisher is available. Sun State Ford Inc. A fire extinguisher would be a great alternative.
The State of Texas Cooperative Purchasing Program available for local governments education and non-profit assistance organizations is now called the Texas SmartBuy Membership Program. NFPA diamonds are an important tool for the identification of chemicals and the hazards they may cause. Custom NFPA 704 Signs.
Sign in to respond to this question. We recently had an accident with a hydrogen balloon catching fire and melting rather than popping. Some pyrophoric chemicals such as the gaseous silane SiH.
Contact EHS Fire Life Safety 858 822-5706 or 534-3531 if you have questions about fire extinguishers. Thermite is an oxidation of the aluminum powder to aluminum oxide and a concurrent reduction of the iron oxide powder to iron. Which of the following chemicals was not used to stain this tissue labster.
3 Hold the nozzle with the other hand and spray the dry powder jet to the flame root of. Use chemical gloves not regular kitchen gloves while handling the solution. Insoluble in water dilute acids alcoholSoluble in hot concentrated sulfuric acidUsed as a weighting mud in oil-drilling in.
A number of ancient historians made mention of alum including Herodotus a Greek historian who lived in the 5th century BC and Pliny the Elder a Roman historian who lived in the 1st century AD. The aluminum and iron oxidation and reduction respectively form the two half cells of the thermite redox reaction. 2 Use one hand to press the pressure handle and then lift the fire extinguisher.
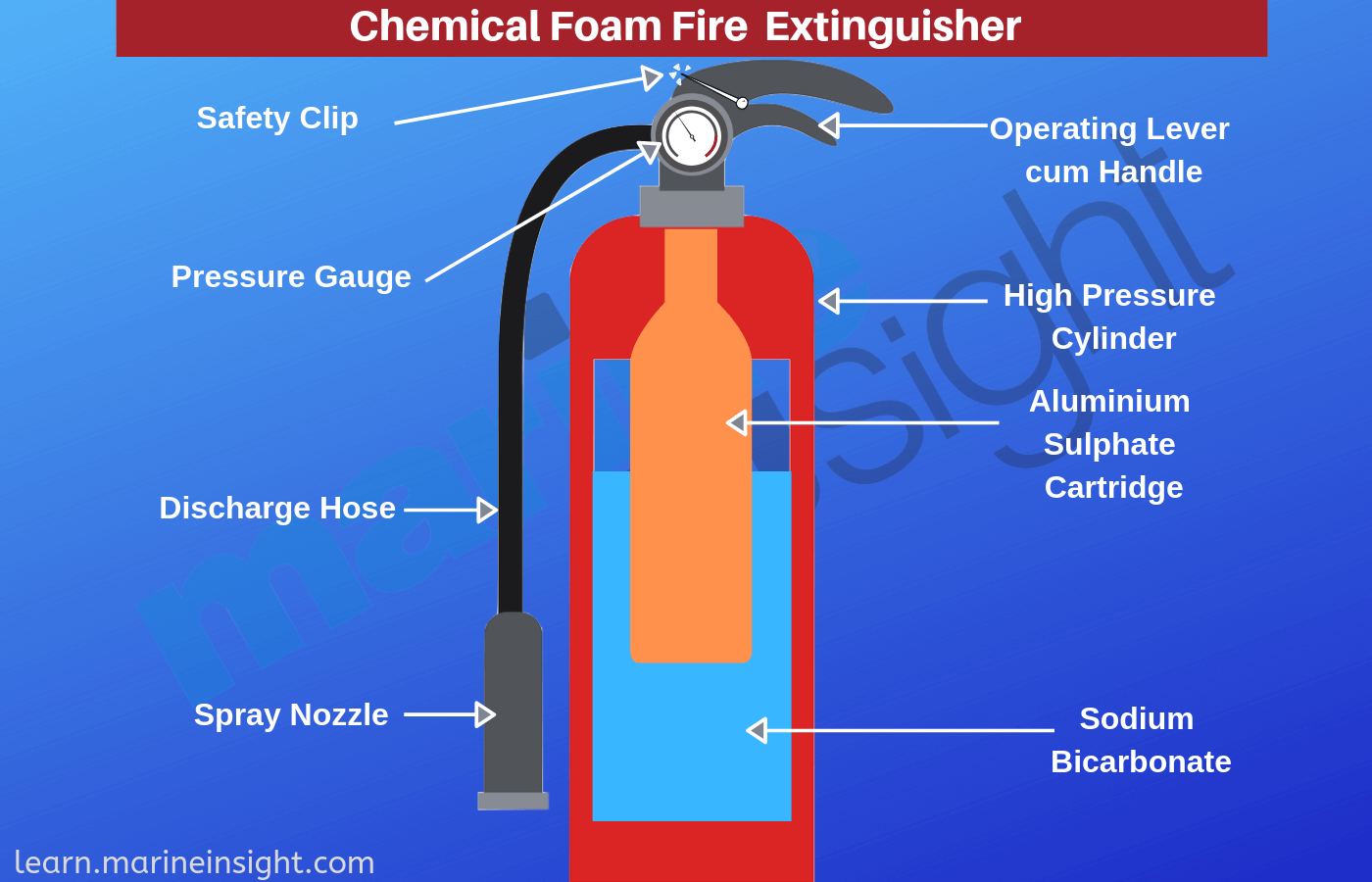
One of our Science teachers has asked for these specific agar plates that are used for culturing microorganisms associated with brewing and. A pyrophoric chemical is a chemical with the ability to spontaneously ignite without the influence of heat or fire in air at temperatures of 54C 130F or below can be in the gas phase the liquid phase or the solid phase. The reaction of aluminium sulfate and sodium bicarbonate forms carbon dioxide and aluminium hydroxide which stabilises the formation of a foam.
The last grade is 2050µm it is a high density material made out of micro diamond pieces. Gain access to purchase from State of Texas contracts and procurement resources by becoming a purchasing memberPut the states multi-billion dollar purchasing power to work for you. Custom NFPA 704 Diamond.
070-57 Trucks Fire Protection and Crash Rescue Effective 1-1-06 this item has been inactivated refer to Class 071 072 and 073 070-59 Trailers Custom. Prevention and Treatment of Explosion Accidents 1 Certain compounds are prone to explosion. It became so popular in.
Pyrophoric gases pyrophoric liquids and pyrophoric solids all share the property of spontaneous ignition. An ABC fire extinguisher is the most common type of fire extinguisher on the market. Rush Truck Centers of Florida Inc.
If you start with aluminum oxide and iron you have the stable products after a thermite reaction not the unstable reagents needed to make a. Answer 1 of 11. Chemical foam is considered obsolete in the United States and has been replaced by synthetic mechanical foams such as AFFF which have a longer shelf life are more effective and more.
Place a solid fuel tablet on a square of aluminum foil on your nonflammable surface. Aluminum Extrusions For Fabricating Window and Door Screens 150-05 Brick Fire Plastic 150-06 Builders Paper Kraft Types Not Felt and Tar Paper 150-07 Boxes Shoring For Construction Worker Protection 150-08 Cabinets Counters Shelves etc Ready-Made 150-09. It is also the most versatile and best extinguisher for home use in most circumstances.
Chem Trade Chemicals 704 904-3003. The carbon dioxide generated also served to propel the foam out of the container be it a portable fire extinguisher or fixed installation using hoselines. Personnel Food Service Equipment Refrigerated etc Effective 1-1-06 this item has been inactivated refer to Class 071 072 and 073.
Customers who viewed this item also viewed. Roebic FRK-1LB Foaming Root Killer Clears.

Hydrated And Anhydrous Copper Sulphate Youtube
Since the only component other than H2O and Cu 2 is the sulfate ion SO 4 2 we can now determine the complete formula of the hydrated copperII sulfate.
Copper 2 sulfate hydrate. The ICSC project is a common undertaking between the World Health Organization WHO and. What color is copperII sulfate. Weiterhin existieren auch ein Trihydrat CuSO 4 3 H 2 O und KupferII-sulfat-Monohydrat CuSO 4 H 2 O.
This material contains CopperII sulfate pentahydrate listed as Copper compounds nos 100 CAS 7758-99-8 which is subject to the reporting requirements of Section 313 of. Kimberly Graziano Hyunjae Kim. Am geläufigsten ist das Pentahydrat CuSO 4 5 H 2 O.
Did the copper change its charge in this reaction. FREE Shipping by Amazon. The primary aim of the cards is to promote the safe use of chemicals in the workplace.
For a general form copperII sulfate hydrate formula CuSO_4 xH_2O we use x to represent the number of water molecules present as water of crystallization per formula unit of anhydrous copperII sulfate CuSO_4. REWEIGH the dish and hydrate. A Case Study Journal of Science Education and Technology 17 466-482.
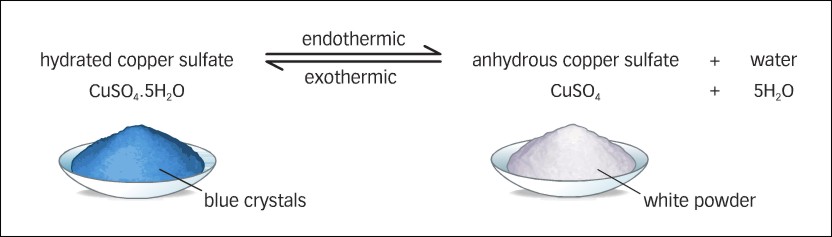
Heat strongly for 5 minutes or longer until the blue. People get magnesium from their diet but sometimes magnesium supplements are needed if magnesium levels are too low. For example blue vitriol or copper sulfate pentahydrate CuSO 4 5H 2 O is blue copper sulfate trihydrate CuSO 4 3H 2 O is blue and anhydrous copper sulfate CuSO 4 is.
Copper Sulfate Pentahydrarte. Ammonium Sulfate Ammonium ions and Sulfate ions NH 4 and SO 4 2 NH 4 2SO 4 Calcium Phosphate Calcium ions and Phosphate ions Ca2 and PO 4 3 Ca 3PO 2 Waters of hydration use table to left copperII sulfate pentahydrate CuSO 45H 2O 11 Undeca. Add about 2000 grams copperII sulfate hydrate to the dish.
Get it Thu Dec 2 - Tue Dec 7. It is a metal sulfate an aluminium salt and a potassium salt. Research suggests that the nature of these bonds is not as simple as.
The pentahydrate CuSO 4 5H 2 O the most commonly encountered salt is bright blue. Magnesium is a mineral that is important for normal bone structure in the body. Ready to use.
The dot after the formula for copper sulphate indicates bonds with water molecules. 40 out of 5 stars 7. The main target users are workers and those responsible for occupational safety and health.
Get it as soon as Wed Dec 1. MM-CoA α-Methylmalonyl coenzyme A tetralithium salt Empirical Formula Hill Notation. Potassium aluminium sulfate is a metal sulfate composed of potassium aluminium and sulfate ions in the ration 112.
The formula of the hydrated form is CuSO 4. For example the anhydrous compound copperII sulfate also exists as a hydrate containing five water molecules and named copperII sulfate pentahydrate. Copper sulfate injected ip at 2 mg copperkg into vitamin e selenium deficient rats caused a 6 fold increase in the formation of the lipid peroxidation product ethane caused acute mortality in 45 rats.
TIP THE TEST TUBE SLIGHTLY DOWNWARD 2. SAFETY EXPERIMENTAL PROCEDURE. WRITE ALL of the digits from the balance reading.
Allow the test tube and its contents to cool. When copperII sulfate pentahydrate CuSO 4 5H 2 O is heated it decomposes to the dehydrated form. Washing soda is the common name for a hydrate of sodium carbonate containing 10 water molecules.
The cards are data sheets intended to provide essential safety and health information on chemicals in a clear and concise way. Kristallwasserverlust bei CuSO 4 5 H 2 O TGADTA Kupfersulfat-Pentahydrat CuSO 4. Write hydrate after the prefix 1 mono 2 di 3 tri 4 tetra 5 penta 6 hexa 7 hepta 8 octa 9 nona 10 deca Example.
Schroeder JD and Greenbowe TJ. Implementing POGIL in the Lecture and the Science. Zinc sulfate is an inorganic compoundIt is used as a dietary supplement to treat zinc deficiency and to prevent the condition in those at high risk.
Copper sulfate 2 lb. It contains an aluminium3. The balanced equation is.
Our unknown hydrate may be a hydrate of copperII sulfate magnesium sulfate ironIII chloride or ironIII nitrate. Hold it over the flame with a test tube holder and heat it for a few minutes with the mouth of the tube slightly below the base of the tube. Only 4 left in stock - order soon.
Kurzbeschreibung weißes bis graues geruchloses Pulver wasserfrei. CopperII sulfate forms a hydrate which contains 361 by mass water. Clean Finish Disinfectant Cleaner Case of 2 564 oz.
The most common form includes water of crystallization as the heptahydrate with the formula Zn SO 4 7H 2 O. Page 1 of 1 Start over Page 1 of 1. Put about ½ inch of hydrated copper sulfate crystals in a dry test tube.
Older names for this compound include blue vitriol bluestone vitriol of copper and Roman vitriol. 99 Pure Copper Sulfate Pentahydrate Crystals. The hydrated form is medium blue and the dehydrated solid is light blue.
Canister Heavy-Duty Multi-Purpose Cleaning Wipes 25-Count 2-Pack 2 lbs. Stoichiometry Interpreting Chemical Equations ANY TIME YOU HAVE MOLE AND MOLE IN THE SAME FRACTION ALWAYS LOOK AT THE BALANCED CHEMICAL EQUATION Stoichiometry is used to convert from moles of one substance. RECORD the mass of the dish and hydrate to - 00010 grams.
The waters of hydration are released from the solid crystal and form water vapor. In many cases the uptake and loss of water by heating decreasing pressure or other means are reversible processes sometimes accompanied by changes in colour. LEAVE TUBE TIPPED DOWNWARD.
More often than not x is a whole number but keep in mind that there are some exceptions. Copper II sulfate pentahydrate. Formulas for ionic hydrates are written by appending a vertically centered dot a.
Copper Sulfate Large Crystals 50lb Bag - EPA 99 Pure. C 25 H 36 Li 4 N 7 O 19 P 3 S xH 2 O. Label the bottles with the normality of EDTA.
Upper Saddle River NJ. Observing our nitrate it has a white crystalline structure representing that similar to table salt. Gently heat the dish by moving the burner back and forth around the base.
CuSO 4 5H 2 Os heat CuSO 4 s 5H 2 Og If 363g CuSO 4 5H 2 O is decomposed to CuSO 4. Place the dish containing the hydrate on the wire gauze. Home and Office Detailer New Cup 3-Pack Price 10 22 19 99 20 48 25 21.
Each copper sulphate unit can attach to five water molecules so its sometimes called copper sulphate pentahydrate when its hydrated. Side effects of excess supplementation may include abdominal pain vomiting headache and tiredness. Ratings 24 17 2 3Concentrated or Ready to Use.
It has a role as a flame retardant a mordant and an astringent. Impact of Learners Prior Knowledge on Their Use of Chemistry Computer Simulations. Either weigh the EDTA analytically or standardize the solution.
CopperII sulfate also known as copper sulphate are the inorganic compounds with the chemical formula Cu SO 4 H 2 O x where x can range from 0 to 5. Record n your observations. In the case of copperII sulfate hydrates you only get x as a whole.
How can we experimentally determine the formula of an unknown hydrate A. Methylmalonyl coenzyme A tetralithium salt hydrate Synonyms. Formation of metallic copper In the fume hood add 20 g of powdered zinc metal stirring until any reaction is apparently complete.
The pentahydrate x 5 is the most common form. 47 out of 5 stars 45. Rewrite the chemical equation for this process including phase designations.
Laboratory-Grade Copper II Sulfate 5-Hydrate Powder 500g - The Curated Chemical Collection. The systematic name is sodium carbonate decahydrate.