Structure of Potassium Chloride. Potassium alum is considered by the FDA as a generally recognized as safe GRAS substance.
![]()
Potassium Hydroxide Molecule Icon Stock Illustration Illustration Of Molecule Kali 168427388
And theres that little gotcha moment even knowing the right ingredients is not enough if theyve been synthetically produced using the toxic chemicals youre trying to avoid.

Potassium hydroxide molecule. The ensuing acid-base neutralization reaction will yield water and potassium chloride as the products. 47 beryllium hydroxide BeOH 2 48 zinc carbonate ZnCO 3 49 manganese VII arsenide Mn 3 As 7 50 copper II chlorate CuClO 3 2 51 cobalt III chromate Co 2 CrO 4 3 52 ammonium oxide NH 4 2 O 33 potassium hydroxide KOH 54 lead IV sulfate PbSO 4. 2KI Cl 2 2 KCl I 2.
These are all strong bases. The neutralising reaction can produce two salts - either the monosodium variety generally known simply as sodium salicylate where the proton from the carboxylic acid group is donated or the disodium one where both available protons are donated. An example of such a reaction is provided below.
Hydrogen discovery atomic structure and location information. Engineering ToolBox - Resources Tools and Basic Information for Engineering and Design of Technical Applications. Potassium nitrate is a common active ingredient in toothpaste exerting an anti-sensitivity action.
Structure of KCl Molecules. Product Number Packaging. Currently sodium carbonate or sodium hydroxide is used to neutralize the fatty acid and convert it to the salt.
Write a net ionic equation for the overall reaction that occurs when aqueous solutions of potassium hydroxide and hydrosulfuric acid are combined. Potassium iodide can be oxidized into an I 2 molecule by introducing an oxidizing agent to it. In contrast a weak base only partially dissociates into its ions in water.
This compound is used as an iodide source in several organic synthesis reactions. Other alkenes react in just the same way. Potassium hydroxide CAS 1310-58-3 pellets for analysis EMSURE - Find MSDS or SDS a COA data sheets and more information.
Soap is produced by a saponification or basic hydrolysis reaction of a fat or oil. Fortunately there arent very many strong bases. Potassium alum is formed by large transparent crystals that are used in different.
Potassium Amyl Xanthate Molecule of the Month for March 2007 PAX Xanthate. PH values for bases like sodium hydroxide ammonia and more. Calcium hydroxide SrOH 2.
16 How many moles are in 34 x 10-7 grams of silicon dioxide SiO 2. They can form ethyl butyl isobutyl normal butyl secondary butyl propyl isopropyl normal propyl and amyl isoamyl normal amyl. The acid or base molecule does not exist in aqueous solution only ions.
The reaction is reversible. Chemistry of the Reaction. Potassium hydroxide results in a soft bar and is usually used for making liquid soap.
They are hydroxides of the alkali. Documentation Sodium hydroxide SDS. Xanthates can be obtained by reacting an alcohol with carbon disulphide and an alkali such as sodium hydroxide or potassium hydroxide.
A diatomic molecule is one that is composed of two atoms of the same element and is either homonuclear or heteronuclear. The hydrogen ion or hydronium ion is a BrønstedLowry. Strong bases react with strong acids to form stable compounds.
Zn Zinc 0001. Potassium 005. One such example is the synthesis of aryl iodides from the diazonium salts of arenes.
Unsaturated fatty acids above the more efficient the stacking the harder the resulting soap. These compounds ionize in water to yield one or more hydroxide ion OH- per molecule of base. Acting as an acid acting as a base Non-aqueous solutions.
If the fatty acid salt has potassium rather than sodium a softer lather is the result. K 4FeCN 6 Answer. Strontium hydroxide BaOH 2.
Examples of Strong Bases. In the image shown at the right one molecule of H 2 O acts as a base and gains H to become H 3 O while the other acts as an acid and loses H to become OH. The potassium cation breaks away from the hydroxyl anion.
Another example is furnished by substances like aluminium hydroxide AlOH 3. There are also tutorials on the first thirty-six elements of the periodic table. Well look at the reaction with ethene.
Potassium Iodide Structure KI Structure A molecule of. 13 How many grams are in 33 moles of potassium sulfide K 2 S. It provides increasing.
It is a natural source of nitrate and has been used as a constituent for several different purposes including food preservatives fertilizers tree stump removal rocket propellants and fireworks. 15 How many grams are in 23 x 10-4 moles of calcium phosphate Ca 3 PO 3 2. Barium hydroxide These bases completely dissociate in solutions of 001 M or less.
It is an inorganic salt also called potassium aluminum sulfate with a formula of AlKSO42 that is predominantly produced in the dodecahydrate form AlKSO42 12H2O. Ni Nickel 000025. The other bases make solutions of 10 M and are 100 percent dissociated at that.
ManganateVII ions are a strong oxidizing agent and in the first instance oxidize ethene to ethane-12-diol old name. The alcohols employed are not unique. The reason for the difference is that the sodium ion is a bit smaller than the potassium ion it interferes less with the stacking of the soap molecules at a molecular level and just like with saturated vs.
General overall hydrolysis reaction. 12 How many moles are in 68 grams of copper II hydroxide CuOH 2. An illustration describing the structure of a KCl molecule is provided below.
BUT potassium alum can also be synthetically produced often using aluminium hydroxide a white cosmetic opacifying agent mixed with potassium or ammonium sulphate in sulphuric acid. BioReliance End-to-End Solutions Greater control increased flexibility for both small large molecule development. PH is a measure of the hydrogen ion H activity in a solution and therefore its acidity or alkalinity.
If air is present carbonates may form. The beryllium hydroxide BeOH 2 is amphoteric in nature. With alkali earth metals.
Although the reaction. Pb Lead 00005. A water molecule assists in this dissociation by forming a solvent separated ion pair with a water molecule wedged between the potassium cation K and the hydroxyl anion HO-Then things get complicated with the hydroxyl and water molecules swapping hydrogens.
Safety Data Sheet SDS Sodium hydroxide Certificates of Analysis. The type of chemical bond that. Answer 1 of 3.
Mg Magnesium 00005. Potassium nitrate is an inorganic salt with a chemical formula of KNO3. Cesium hydroxide CaOH 2.
Bases - pH Values pH values for bases like sodium hydroxide ammonia and more. Ammonia is a good example of a weak base. While the other hydroxides of this group like magnesium hydroxide MgOH 2 calcium hydroxide CaOH 2 etc.
Again remember that you never have to indicate the number of cations and anions in the name of an ionic compound. Since ethylenediamine is a neutral molecule the oxidation number of cobalt in the complex ion must be 3. Hydroxide is a diatomic anion with chemical formula OH It consists of an oxygen and hydrogen atom held together by a single covalent bond and carries a negative electric chargeIt is an important but usually minor constituent of waterIt functions as a base a ligand a nucleophile and a catalystThe hydroxide ion forms salts some of which dissociate in aqueous solution liberating.
In the laboratory KCl can be prepared by reacting bases of potassium such as potassium hydroxide with hydrochloric acid. Fat NaOH --- glycerol sodium salt of fatty acid. Most popular and most useful bases are formed with alkali metals such as sodium hydroxide NaOH potassium hydroxide KOH lithium hydroxide LiOH etc.
14 How many moles are in 12 x 103 grams of ammonia NH 3.
For the relief of tooth sensitivity and is also used as a pesticide. The name of the element is derived from the Latin words nitron and genes for nitre potassium nitrate forming Elemental nitrogen comprises about 78 of the Earths.
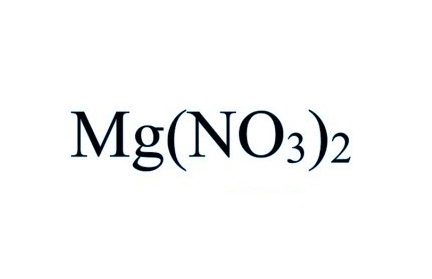
Magnesium Nitrate Facts Formula Properties Uses
The nitrate ion with the partial charges shown.

Magnesium nitrate molecule. Small Molecule Groups Approved Structure. Salts containing this ion are called nitrates. What Is Reduction.
Positive charges indicated by Roman numeral. In the textile. What is the relative mass formula of sodium chloride.
The aldehyde version of this nonanal also causes undergrads some amusement especially when a hyphen is. All group 2 elements have the same electron configuration in the outer electron shell and a similar crystal structure. The ammonia can also be in the form of ammonium salts such as ammonium nitrate NH 4 NO 3 ammonium sulfate NH 4 2 SO 4 and various ammonium phosphates.
Its official name is magnesium iron silicate hydroxide and it has the formula. The molecule shown is 2-nonanone but 5-nonanone with the CO group in the middle would be the same forward as well as backwards thus being palindromic in spelling and in structure. The molar mass of magnesium nitrate equals 14833 gmol.
Recently a novel vertical cutoff wall backfill comprised of reactive magnesium-activated slag bentonite and sand referred to as MSBS backfill has been developed by Wu et al. C 6 H 14. Nitrate is a polyatomic ion with the chemical formula NO 3.
An illustration describing the structure of the silver nitrate molecule is provided below. Magnesium deficient sweetpotato leaves become reddish-purple. In the trial subjects took a daily dose of ZMA which included 30 mg zinc monomethionine 450 mg magnesium aspartate and 105 mg vitamin B-6 at night during 7 weeks.
The ion is the conjugate base of nitric acid. Cytokine immune system signaling molecule. Fe 2 ironII ion Fe 3 ironIII ion Cu copperI ion Cu 2 copperII ion b Older but still used rule.
Magnesium is a chemical element with the symbol Mg and atomic number 12. The MSBS backfill is shown to possess excellent unconfined compressive strength hydraulic performance and durability. Nitrogen is a colorless odorless gas.
The chemical formula of table salt or sodium chloride is. NaCl There are one sodium atom and one chlorine atom in each molecule. In one molecule of the compound determine how many atoms of every element are present for each one of these chemical formulas.
3 c magnesium nitrate MgNO. The number of positive charges is not indicated in the name because it is not necessary Rule. Sodium Chloride is an ionic solid with the formula Na Cl-.
Plasma nitrate Ø. The relative mass formula of sodium chloride is M r NaCl A. In its elemental form nitrogen is found as the diatomic molecule N 2 in which the two nitrogen atoms are held together by a triple bond.
A silver metal Ag b ammonia gas NH. Balance oxygen last since it is present in more than one molecule on the right side of the equation Show Answer. Consider as an example the reaction between one methane molecule CH 4 and two diatomic oxygen molecules.
Latin stem for. A 5494 g. Magnesium is a cofactor in protein synthesis muscle relaxation and energy production.
Calculate the molar mass for each of the following substances. Fifty subjects were evaluated using 2 tactile methods and cold air sensitivity dental air syringe along with subjective. Quercetin hesperetin and caffeic acid 5-S-cysteinyl-dopamine and its oxidation product dihydrobenzothiazine.
While any expression that cites the number and kind of atoms is a. Creating firework colors is a complex endeavor requiring considerable art and application of physical science. Carbon dioxide is a one-carbon compound with formula CO2 in which the carbon is attached to each oxygen atom by a double bondA colourless odourless gas under normal conditions it is produced during respiration by all animals fungi and microorganisms that depend directly or indirectly on living or decaying plants for food.
Now the equation is balanced with 2 Chlorides Cl with total charge -2 and 3 Chromiums with total charge 3 on both sides. Write a balanced equation for the decomposition of ammonium nitrate to form molecular nitrogen molecular oxygen and water. Structure for Magnesium hydroxide DB09104.
Na 23 Cl 355 Solution. A manganese metal Mn b sulfur hexafluoride SF. Each molecule contains 2 hydrogen atoms and 1 oxygen atom.
It can be observed that silver has an. Almost all inorganic nitrates are soluble in water. Structure for Potassium nitrate DB11090.
It is sometimes convenient to use. Small Molecule Groups Approved Investigational Structure. Magnesium carbonate MgCO 3 Methanol CH 3 OH Argon gas Ar Dimethyl sulfoxide C 2 H 6 OS Iron iii oxide Fe 2 O 3 CHEMISTRY Related Links.
100951524732 Chemical Formula KNO 3 Synonyms. Other hormones like IGF. Excluding propellants or special effects the points of light ejected from fireworks termed stars generally require an oxygen-producer fuel binder to keep everything where it needs to be and color producerThere are two main mechanisms of color production in fireworks.
Preparation of the metathesis method potassium nitrate ammonium nitrate and potassium chloride raw materials products for potassium nitrate and ammonium chloride recycled mother liquor wherein the device is provided close to the production of a shunt purify production line production line set up in addition to sodium unit and the unit except magnesium which in addition to the sodium. We get Cr 3 2Cl-1 Cr 3 Cl-1 2. There are six C atoms and 14 H atoms in a hexane molecule which has a molecular formula of.
57990521206 Chemical Formula H 2 MgO 2 Synonyms. In a dentifrice or gel to alleviate dentinal hypersensitivity. It is a shiny gray solid which bears a close physical resemblance to the other five elements in the second column group 2 or alkaline earth metals of the periodic table.
An example of an insoluble nitrate is bismuth oxynitrate. Note there is no subscript for the number 1 Types of Chemical Formulas. When exposed to PbZn testing liquids with concentrations of 01 mgL 4.
MOL SDF PDB SMILES InChI. HCN hydrogen cyanide Its a toxic gas C 18 H 21 NO 3 codeine a painkilling drug Ca 10 PO 4 6 OH 2 Hydroxyapatite that is present in the enamel of the tooth. The relative mass formula of water is M r H 2 O 2 A r H A r O 2 1 16 18.
Potassium nitrate has been used. PPI PPI PPI Deficiency symptoms Because Mg is a mobile element and part of the chlorophyll molecule the deficiency symp-tom of interveinal chlorosis first appears in older leaves. Some research shows that this combination can help raise testosterone levels.
The aim of this study was to compare a 3 potassium nitrate02 sodium fluoride mouthwash with a 02 sodium fluoride control mouthwash in a 6-week double-blind study. Ammonia is also used in the manufacture of commercial explosives eg trinitrotoluene TNT nitroglycerin and nitrocellulose. Urea H 2 N 2 CO is the most commonly used source of nitrogen for fertilizer worldwide.
This is how the redox equations are balanced. Hno3 Molar Mass. 6 c strontium acetate SrC.
MOL SDF PDB SMILES InChI. Mg 2 magnesium ion H hydrogen ion Sr 2 strontium ion Al 3 aluminum ion Comment. When magnesium hydroxide reacts with nitric acid it produces magnesium nitrate and water MgOH2 2HNO3 MgNO32 2H2O In one to two sentences explain how this reaction demonstrates that matter is neither created nor destroyed in a chemical reaction 2 See answers Advertisement Advertisement Oseni Oseni It can be shown that matter is neither created nor destroyed.
To balance the unbalanced chloride molecule charges we add 2 in front of the chloride on LHS. Leaf tissue between the veins may be yellowish bronze or reddish while the leaf veins remain. Cocoa polyphenols unspecified prostacyclin.
This supplement is a combination of zinc magnesium and vitamin B-6. Calculate the molar mass for each of the following substances. One atom is present in each of the elements hydrogen carbon and.
Nitrates are common components of fertilizers and explosives.