Moles of H 2504 2. The total is 98.
What Is The Mechanism For The Acid Catalyzed Esterification Of Formic Acid And 2 Butanol Socratic
ROH Na OH RO Na HOH.

Is 2-butanol an acid or base. Most alcohols are slightly weaker acids than water so the left side is favored. D All of the above. 2-methylbutan-2-ol is a tertiary alcohol that is propan-1-ol in which both of the hydrogens at position 1 have been replaced by methyl groups.
That means the hydroxyl group is attached to the second carbon atom. As a result it transfers a proton to water. Any correct structure of.
02 moles x 32. Alkalosis is the opposite condition with blood pH being excessively high. Or the reaction in which the 2-butanol.
The elimination of water from an alcohol is called dehydration. Sodium salt of Jun 19 2020 The product formed when ferrous sulphate is heated is A Ferric oxide B Sulphur dioxide C Sulphur trioxide D All of the above Answer. The companys unparalleled raw material expertise is focused on the supply of essential oils and aromatic chemicals.
The Solubility Test Flowchart 2. Several assessments are included with the guidelines models databases state-based RSL Tables local contacts and framework documents used to perform these assessments. 0299 moles HCN.
We are a leading supplier to the global Life Science industry with solutions and services for research biotechnology development and production and pharmaceutical drug therapy development and production. The index into Erowids collection of more than 20000 personal reports describing first-hand experiences with psychoactive plants and drugs. It has a role as a protic solvent.
Please use this book to increase your knowledge for the laboratory pratictioner. An acid dissociation constant K a is a quantitative measure of the strength of an acid in solutionIt is the equilibrium constant for a chemical reaction known as dissociation of acidbase reactions. 2-butanol is a clear organic secondary alcohol.
2-Butanol 2 4-DNP Test for Aldehydes and Ketones 1 acetates 1 Acetone 1 Acetone Test 1 acidimetry and alkalimetry 1 activation energy 1 Alternatives for Pesticides 2 Analysis of an antacid tablet 1 Andrews condition 1 antacid When the reaction involves in a titration does not satisfies the conditions for a direct titration to be performed 1 aryl 1 Baeyer Test 1. The product of this reaction is a stronger acid than water. Chemistry 1 decade ago.
Malolactic conversion also known as malolactic fermentation or MLF is a process in winemaking in which tart-tasting malic acid naturally present in grape must is converted to softer-tasting lactic acidMalolactic fermentation is most often performed as a secondary fermentation shortly after the end of the primary fermentation but can sometimes run concurrently with it. In the group shown below which of the following alcohols is are likely to yield a product where skeletal rearrangement has occurred when treated with sulfuric acid. 204 g CaCO3 4a.
In aqueous solution the equilibrium of acid dissociation can be written symbolically as. Finally base catalyzed hydrolysis of the phthalimide moiety and the esters followed by acidification and thermal decarboxylation produces an amino acid and phthalic acid not shown. The sodium salt of the acid as the acid is neutralized by the base.
2 2 Only functional group correct Slegs funksionele groep korrek Max. A Lewis acid-base reaction therefore rapidly occurs in which a pair of nonbonding electrons on a water molecule are donated to the carbocation to form a covalent C O bond. 3 An elegant procedure known as the Strecker synthesis assembles an alpha-amino acid from ammonia the amine precursor cyanide the carboxyl precursor and an aldehyde.
Marking criteriaNasienriglyne Whole structure correctHele struktuur korrek. Pore size 02 μm pore size 05 μm cartridge nominal length 5 in. How the EPA conducts risk assessment to protect human health and the environment.
The pH of blood is usually slightly basic with a value of pH 7365. HA H 2 O A-H 3 O. The primary aim of the cards is to promote the safe use of chemicals in the workplace.
This base was expanded upon more than two decades ago to include fragrance production. The cards are data sheets intended to provide essential safety and health information on chemicals in a clear and concise way. After placing five bonds between the atoms and adding the two.
03 g NH3. B Calculate the mole fraction of ascorbic acid in this solution. Plaque can create a local.
The ICSC project is a common undertaking between the World Health Organization WHO and. Amines that are insoluble in pure water will be soluble in acid due to the formation of an ammonium chloride salt. Silver Nitrate Test for Alkyl Halides and Carboxylic Acids The reaction of an alkyl.
A solution containing 815 g of ascorbic acid dissolved in 230 g of water has a density of 122 gmL at 55 C. A Calculate the mass percentage of ascorbic acid in this solution. Its very The concentration of an acid or base in solution can be determined by titration with a strong base or strong acid respectively.
The most common disorder in acidbase homeostasis is acidosis which means an acid overload in the body generally defined by pH falling below 735. When an alcohol is treated with sodium hydroxide the following acid-base equilibrium occurs. This base was expanded upon more than two decades ago to include fragrance production.
Solubility in 6M HCl is a positive identification test for bases. Because the cyclohexene has lower boiling point than the cyclohexanol the cyclohexene can be distilled as it forms. Ascorbic acid vitamin C C6H8O6 is a water-soluble vitamin.
It is usually An Arrhenius acidbase reaction occurs when a dissolved aqueous acid and a dissolved aqueous base are mixed together. This is supported by our long-standing relationships with a worldwide fabric of producers ensuring the greatest prospects for uninterrupted supply in markets that are often volatile. Because the slowest step of this reaction only involves t-butyl bromide.
This value is often referred to as physiological pH in biology and medicine. The companys unparalleled raw material expertise is focused on the supply of essential oils and aromatic chemicals. The difference in the R-2-butanol and the S-2-butanol has to do with the.
963 1 3 428 107 0. 567 X 1022 Molecules Of Glucose C6H1206. Recalling that water is a much better leaving group than hydroxide ion it is sensible to use acid.
1 10 -6. In step three the base HSO4 -removes a proton from the carbon adjacent to the positively charged carbon forming an alkene and regenerating the acid catalyst H2SO4. The main target users are workers and those responsible for occupational safety and health.
This is supported by our long-standing relationships with a worldwide fabric of producers ensuring the greatest prospects for uninterrupted supply in markets that are often volatile. Urine was analyzed immediately 1 2 8 and 9 hr after drinking during 2 hr 375 mlkg of beverages containing orange juice 15 or 40 ethanol and 1 gl of 1-propanol 2-propanol 1-butanol 2-butanol isobutyl alcohol or a mixture of 1-propanol isobutyl alcohol. Where HA is an acid that dissociates into A known as the conjugate.
3-methyl-3-pentanol 33-dimethyl-2-butanol 22-dimethylcyclohexanol A 3-methyl-3-pentanol only B 33-dimethyl-2-butanol only C 22-dimethylcyclohexanol only. Moles atomic weightgram weight. Dehydration of 2-methyl-2-butanol produces primarily 2-methyl-2-butene a tri-substituted alkene.
125 cm Code 7 EPDM rubber seal.
Have different connectivities not limited to alkanes C4H10 C5H12 C2H6O OH bu tanol O di ehylr straight-chain or normal hydrocarbons branched hydrocarbons n-butane n-pentane Systematic Nomenclature IUPAC System Prefix-Parent-Suffix Parent- number of carbons Prefix- substituents. Moreover the thresholds may be different even between position isomerism more than 100 times Figure 5.
Heat Energy Produced By Fuel Butanol And Its Isomers Owlcation
It is an essential element for life and is found in two amino acids Cysteine and Methionine.
Isomers of butanol. Consider the molecular formula C. Heptanediol and its isomers. Both compounds have zero degrees of unsaturation no multiple bonds or rings.
Imagen de la estructura. The other isomers of. Acetic anhydride vanillin methylene acetate cresol.
25167-67-3 250 ppm. N-2-benzyloxyphenyl isobutyrylacetamide Ac-met. 71-36-3 1 Número RTECS.
Hexanediol and its isomers. Butanol C 4 H 9 OH is commonly represented by the molecule butan-1-ol a primary alcohol which has the structural formula Other isomers of butanol can be drawn by changing the way that the OH group is joined to the molecule or by changing the way that the carbon atoms are joined to each other see the isomers of butane. Pick out the pairs of isomers for each pair state whether they are structural geometric or conformational isomers.
In mice after a single ip injection of 81 mmol tert-butanolkg body weight initial blood levels of 8 mmol took 8-9 hr for elimination blood- tert-butanol half-life was approximately 5 hr. Specify a facility by using any combination of facility name and geographic location. Isomerie von den altgr.
2 METHOXY 3 ISOPROPYL PYRAZINE. Butanol 1-Butanol Butyl alcohol Butyl hydrate Butylic alcohol Butyralcohol Butyric alcohol Butyryl alcohol n-Butyl alcohol 1-Hydroxybutane. Click on the image to view our PDF worksheet.
Thus a carboncarbon multiple bond or in some. 87 MB worksheet - naming hydrocarbons DOCX 58 KB worksheet - naming alkanes DOCX 164 KB worksheet - naming organic chemistry DOCX 47 KB. 1-Butanol occurs naturally as a minor product of the ethanol fermentation of sugars and other saccharides and is present in many foods and drinks.
You can also get position isomers on benzene rings. A Predict the products of the following reactions b Name the starting materials. Consider 2-butanol drawn in two dimensions below.
-4-3-2-1 0 1 2 log Threshold secButanol ppm Isopropanol n-Propanol tertButanol n-Butanol. Sec-Butanol 78-92-2 100 ppm. Butyl acetates all isomers 105-46-4.
If the oxygen atom were inserted in a C-C bond of n-butane ethers would be formed both of which diethyl etherand methyl n-propyl ether have zero degrees of unsaturation. Therefore we are providing the current palette of ingredients we use as well as a list of ingredients we do not use below. Numerando los carbonos de la cadena del butano y realizando esta sustitución en el carbono.
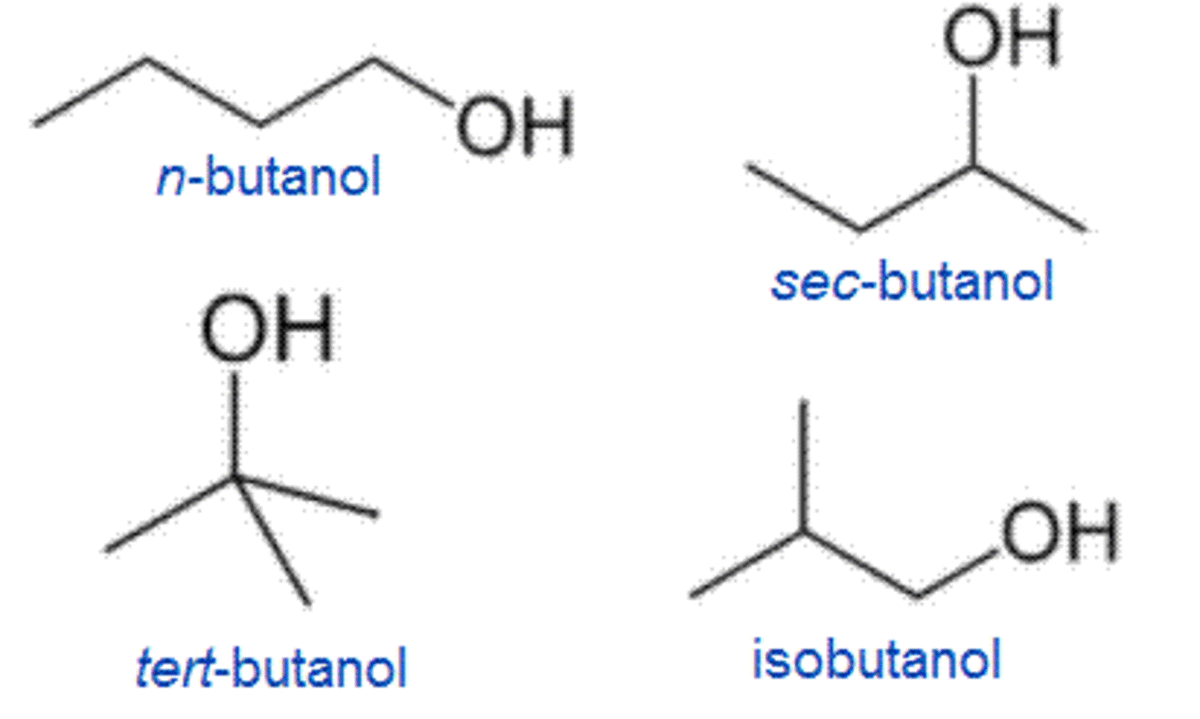
2-Butoxyethanol EGBE 111-76-2 20 ppm. In one of them the functional group -OH is on the end of the chain whereas in the other it is attached to the second carbon of the main carbon chain. The four isomers of butanol are shown below.
All are a butyl or isobutyl group linked to a hydroxyl group sometimes represented as BuOH n-BuOH i-BuOH and t-BuOHThese are n-butanol 2 stereoisomers of. Benzophenone ether hexane xylene acrylate alkyl. Butanol and isomers.
Simple rotation about a CC σ bond in an alkene for example cannot occur because of the presence of the π bond. Compounds with the same chemical formula but different arrangement of atoms Constitutional isomer. The SEMS Search allows you to retrieve Superfund data from the Superfund Enterprise Management System SEMS database in Envirofacts.
Green bell pepper tomato. 2-butanol butan-2-ol o sec-butanol Este tipo de isomería resulta de la posibilidad de colocar grupos funcionales cadenas laterales en posiciones estructuralmente no equivalentes. Tert-Butanol 75-65-0 100 ppm.
1-Butanol Butyrate RR meso mixed isomers Valine leucine Isoleucine R S mixed isomers Benzoate p-hydroxyl 2-amino dihydroxy salicylate Farnesene Mevalonate FAEE FABE Citramalate Long chain alcohols Acetone 1 Selectivity 100. So two other isomers of butanol are. If we mentally insert an oxygen atom into a C-H bond we will have an alcohol either 1-butanol or 2-butanol.
Biological Monitoring Methods for Industrial Chemicals. PSG Publishing Co Inc. However after 3 days inhalation at a vapor concentration to give levels of 8 mmolL blood tert-butanol disappeared within 3 hr of removal of mice from the inhalation chamber half-life of tert.
Alkanediols with 7 or fewer carbon atoms. Supongamos que sustituimos uno de los átomos de hidrógeno del butano CH 3-CH 2-CH 2-CH 3 por un grupo hidroxilo. Functional group isomerism is a type of structural isomerism where isomers have.
2 METHYL 2 PENTENOIC ACID. Methyl Propanol ethylene chlorine butyl acetic. Wortbildungselementen ἴσος isos gleich μέρος meros Anteil Teil Stück ist das Auftreten von zwei oder mehreren chemischen Verbindungen mit gleicher Summenformel und Molekülmasse die sich jedoch in der Verknüpfung oder der räumlichen Anordnung der Atome unterscheidenDie entsprechenden Verbindungen werden Isomere genannt und lassen sich.
From Mill to Wing-An ATJ Pathway Ethanol Lanzanol Chemical Conversion Diesel Jet Fuel 4000 gallons of on-spec jet and 600 gallons of diesel. It is also a permitted artificial flavorant in the United States used in butter cream fruit rum whiskey. When the functional group is different such as aldehyde and ketone fatty acid and ester it is not rare that the thresholds are different about 10000 times between isomers.
Isomers of butan-1-ol are isobutanol butan-2-ol and tert-butanol. Alcohols Butanol Isomers Iso-pentanol Alkanes 2-Methyl and n-Alkanes n-Dodecane. Geometric isomers differ in the relative positions of substituents in a rigid molecule.
Pentanediol and its isomers. Exposure to an air concn of 50 ppm for 2 hr resulted in blood levels less than 008 mgl. Ethyl methanol benzaldehyde dimethyl hydrofluoric hydroxy.
The substituents are therefore rigidly locked into a particular spatial arrangement. 2 METHOXY-3-ISO BUTYL PYRAZINE. Isobutene 115-11-7 250 ppm.
While all our product fragrance ingredients are safe to use we understand some people want to learn more. The answer is B by definition 4. Alkoxyalkanols with 6 or fewer carbon atoms.
Benzyl ester sulfonic cyclohexane acetone chloride. A enantiomersB diastereomersC structural isomers D identicalE none of the above The key here is to know the definition of all terms and then determine R or S configuration for each chiral carbon. 2 METHYL 3 FURANTHIOL 5 ETOH.
Volunteers exposed to n-butanol for 2 hr at air concn of 100 and 200 ppm developed blood concn that never exceeded 10 mgL whether at rest or during excercise. Chemistry of Natural Substances Organic Chemistry Worksheets 13 Worksheets 6 AlkanolsAlkyl halidesAlkanalsAlkanones and functional group tests Question 1. Butanediol and its isomers.
Butane All isomers 75-28-5106-97-8 1000 ppm EX n-Butanol 71-36-3 20 ppm. Acid nitric butyl phenyl benzene toluene. Butanol also called butyl alcohol is a four-carbon alcohol with a formula of C 4 H 9 O H which occurs in five isomeric structures four structural isomers from a straight-chain primary alcohol to a branched-chain tertiary alcohol.
Its essential if you are asked to draw isomers in an exam not to restrict yourself to chain isomers or position isomers. For example there are two structural isomers occurs in n-butanol with the molecular formula C 4 H 9 OH. 2 METHOXY-356 METHYL PYRAZINE.
Isomers are compounds with different physical and chemical properties but the same molecular formula. C 4 H 10 O. The unmodified term butanol usually refers to the straight chain isomer.
The specific rotation of pure R-2-butanol is. You will see that we do not use many common food allergens in our fragrances including Animal Derived Materials Egg Derivatives Milk Derivatives Gluten Barley. You must be aware of all the possibilities.
H-CH 2 4 OH. Butenes All isomers 106-98-9. 2-Butoxyethyl acetate EGBEA 112-07-2 20 ppm.