Sodium chloride NaCl and magnesium oxide MgO. Hg2O mercury I oxide 35.
Chemidplus 1314 84 7 Nqdyswqrwwtvju Uhfffaoysa N Zinc Phosphide Iso Similar Structures Search Synonyms Formulas Resource Links And Other Chemical Information
The first box is the intersection between the zinc cation and the chloride anion so.

Zinc phosphide formula. 2 provides a dispersion formula based on data from ref. IronII chloride or ferrous chloride The cation charge must be specified since iron can form more than one charge. This activity includes every compound formula and name that can be formed from the list 44 Ions provided in Chemistry A at Pickerington High School Central.
A monatomic meaning one-atom cation takes its name from the name of the element. µm Angle of incidence 090. Hg23P2 mercury I phosphide Section D Write the formula for the ionic.
Fe 3 ferric. Using this program will help you to learn how to write ionic compound names and formulas for Chemistry A. 12D1 Gallium arsenide GaAs and gallium phosphide GaP both have the zinc blende crystal structure and are soluble in one another at all concentrations.
Ionic Compound Formula Writing Worksheet Write chemical formulas for the compounds in each box. Zn2 Zinc Cd2 Cadmium 3 Al3 Aluminum 3- N3- Nitride P3- Phosphide Table 2 Metals That Form More Than One Monatomic Ion ELEMENT ION FORMULA SYSTEMATIC NAME COMMON NAME Chromium Cr2 Chromium II Chromous Cr3 Chromium III Chromic Cobalt Co2 Cobalt II Cobaltous Co3 Cobalt III Cobaltic Copper Cu Copper I Cuprous. For example Cu is called CopperI and Cu 2 is called CopperII in the names of compounds containing these ions.
Potassium phosphide K 3P zinc carbide Zn 2C manganese IV sulfide MnS 2 cobalt II. 18 ammonium oxide NH 4 2O. Reacts with calcium phosphide incandescently at about 300C.
Zn 3 P 2 is a II-V semiconductor with a direct band gap of 15 eV and may have applications in photovoltaic cells. Use the stock form for the transition metals. Many occur in families of names.
Circle the symbol for the metal in each of the compounds in Model 2. Chemical Formula Nomenclature Practice. In the compound zinc phosphide what is the.
SrCl2 strontium chloride 36. Zinc carbonate ZnCO 3 aluminum hypochlorite Aℓ C ℓO 3 calcium phosphate Ca 3PO 4 2 cadmium phosphate Cd 3PO 4 2 iron III sulfate Fe 2SO 4 3 mercury II chlorite HgC ℓO 2 2 potassium phosphite K 3PO 3 magnesium hydroxide MgOH. 13 manganese II phosphate Mn 3PO 4 2.
Sodium thiosulfate ____Na2S2O3_____ 3. If theres just a plus sign it means the charge is plus 1. Ag Silver.
Hg23N2 mercury I nitride 45. One of the earliest methods of distinguishing between these ions used the suffixes -ous and -ic added to the Latin name of the element to represent the lower and higher oxidation states respectively. The positive charge more protons versus electrons for a cation is shown by a number and plus sign after the formula.
Zn 2 zinc ion. You should complete this by Sunday. Na Mg 2 Non.
T LogX LogY eV Reflection calculator. A second compound exists in the zinc-phosphorus system zinc diphosphide ZnP 2. Ionic Compound Naming and Formula Writing List 1.
12 nickel III sulfide Ni 2S 3. LiBr lithium bromide 39. An ionic compound is composed of a metal and a non-metal.
Ionic Compounds Containing a Metal and a Polyatomic Ion. Name the cation first specifying the charge if necessary then. SrO strontium oxide 40.
16 magnesium sulfate heptahydrate MgSO 47H 2O. There are many polyatomic anions. Metals lose electrons to produce positve ions called cations.
The chemical formula of ionic compounds can be quickly calculated using the chemical formula calculator. Cs2S cesium sulfide 42. Write the chemical formula for the following ionic compounds.
H hydrogen. Aluminum Al 3 barium Ba 2 bismuth Bi 3 cadmium Cd 2. P-polarized R P S.
The names are found by finding the intersection between the cations and anions. Give the formula for the following. Ca 2 calcium.
Zinc phosphate is an inorganic compound with the formula Zn 3 PO 4 2This white powder is widely used as a corrosion resistant coating on metal surfaces either as part of an electroplating process or applied as a primer pigment see also red leadIt has largely displaced toxic materials based on lead or chromium and by 2006 it had become the most commonly used corrosion inhibitor. Which element comes first in the name and formula of the compounds in Model 2 or the nonmetal. Optical constants of CaCO 3 Calcium carbonate Calcite Ghosh 1999.
CuO copper II oxide 43. 17 potassium carbonate K 2CO 3. Li Lithium Zn2 Zinc Na Sodium Hg 2 2 MercuryI K Potassium NH 4 Ammonium Rb Rubidium Cs Cesium Be2 Beryllium Anions Name Mg2 Magnesium NO 2-Nitrite Ca2 Calcium NO 3-Nitrate Ba2 Barium SO 3 2-Sulfite Sr2 Strontium SO 4 2-Sulfate Al3 Aluminum HSO 4-Hydrogen sulfate bisulfate OH-Hydroxide Anions Name CN-Cyanide H-Hydride PO 4 3-Phosphate F-Fluoride HPO 4 2.
Complete these in lab and on your own time for practice. Mixtures with ammonium nitrate or with metal powders can be exploded by shock Kirk and Othmer 8644. K potassium.
The name or symbol of the metal comes first. Metals combine with polyatomic ions to give ionic compounds. Determine the concentration in weight percent of GaP that must be added to GaAs to yield a unit cell edge length of 05570 nm.
FeI2 iron II iodide 38. Fe 2 ferrous. N Zinc phosphide Al Aluminum oxide Strontium chloride Sr 4.
Zinc phosphide Zn 3 P 2 is an inorganic chemical compoundIt is a grey solid although commercial samples are often dark or even black. The transfer of electrons between metals and non-metals produces charged particles called ions. Use the table of ions in Model 1 to answer the following questions.
In a formula parentheses are used around a polyatomic ion only when there are 2 or more of that polyatomic ion in a formula unit ie when the subscript is. Start by learning the polyatomic ions ending with -atesuch as chlorate ClO 3- nitrate NO3 2- sulfate SO4 2 carbonate CO 3 and phosphate PO 4 3-The corresponding -ite ion name has one less oxygen and the same. Copy this to my account.
11 silicon dioxide SiO 2. 14 silver acetate AgC 2H 3O 2. PbCl4 lead IV chloride 41.
15 diboron tetrabromide B 2Br 4. 10 V 2S 3 vanadiumIII sulfide. Review some examples of cations or positive ions.
Binary Ionic Compounds Type II The cation of a transition metal is always named first like any cation and the anion second. Data Expressions for n CSV - comma separated TXT - tab separated Full database record Optical transmission calculator. Reacts violently with phosphorus trioxide Chem.
E-mail to a friend. Sulfur dioxide ____SO2_____ 2. Some metals form positive ions in more than one oxidation state.
Write the formulas for the following chemical compounds. Sn 2 stannous. This problem asks that we.
I Calcium phosphide ZnCO 3 I Zinc carbonate NaNO 3 I Sodium nitrate H 2 SO 3 A Sulfurous acid AgCl I Silver chloride CH 4 M Carbon tetrahydride Cu 2 C 2 O 4 I Copper I oxalate SnI 4 I Tin IV iodide PbS 2 I Lead IV sulfide CO 2 M Carbon dioxide Al 2 SO 4 3 I Aluminum sulfate. Combinations of finely divided sulfur with finely divided bromates chlorates or iodates of barium calcium magnesium potassium sodium or zinc can. Zinc phosphide 29 SrC 2 H 3 O 2 2 strontium acetate 30 Cu 2 O copper I oxide 31 Ag 3 PO 4 silver phosphate 32 YClO 3 yttrium I chlorate 33 SnS 2 tin IV sulfide 34 TiCN 4 titanium IV cyanide 35 KMnO 4 potassium permanganate 36 Pb 3 N 2 lead II nitride 37 CoCO 3 cobalt II carbonate 38 CdSO 3 cadmium sulfite 39 CuNO 2.
FeS iron II sulfide 44. Al2O3 aluminum oxide 37. The densities of GaAs and GaP are 5316 and 4130 gcm3 respectively.
Zn3P2 zinc phosphide 34. ZnNO32 zinc nitrate Fe2SO43 ironIII sulfate or ferric sulfate CuCO3 copperII carbonate or cupric carbonate Note. It is used as a rodenticide.
Formula Type Chemical Name HCl A Hydrochloric Acid Hg 2 SO 4 I Mercury I sulfate N 2 O 3 M Dinitrogen trioxide CdS I. Sr 2 strontium.
Ionic compounds are compounds made up of ions. Copper II bromide CuBr2 8.

How To Write The Formula For Iron Iii Phosphide Youtube
Sign up and fill out a short form to start offering your service.
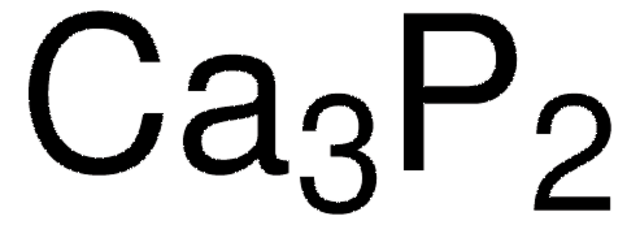
Formula for phosphide. PO 4 3-Æ HPO 4 2-Æ H 2PO 4-phosphate hydrogen phosphate dihydrogen phosphate. Many different phosphides are known with widely differing structures. Binary Ionic Compounds Type II The cation of a transition metal is always named first like any cation and the anion second.
Copy this to my account. Use Lewis dot structures to show the covalent bonding in the following pairs of. Which element comes first in the name and formula of the compounds in Model 2 or the nonmetal.
In chemistry a phosphide is a compound containing the P 3 ion or its equivalent. OptixeXpert is our new website that aims at connecting optical professionals with employers looking to outsource optics related work. 11 tetraphosphorus triselenide P4Se3.
Ba3N2 __barium nitride__ Na3P ___sodium phosphide Exercise. IronII chloride or ferrous chloride The cation charge must be specified since iron can form more than one charge. E-mail to a friend.
10 N2O3 dinitrogen trioxide. Those materials consisting only of phosphorus and a less electronegative element. Numerous are polyphosphides which are solids consisting of anionic chains or clusters of phosphorus.
P 10 S 4. Ionic Compounds Naming and Formula Writing. Write the correct chemical formula for the ionic compound that forms.
3 aluminum oxide Al and O 6 magnesium phosphide Mg and P Formula. Lead II chloride PbCl2 5. Metals combine with polyatomic ions to give ionic compounds.
Cesium phosphide __Cs3P__ calcium iodide _CaI2_ barium fluoride __BaF2__ magnesium nitride __Mg3N2__ aluminum bromide __AlBr3__ sodium selenide _Na2Se. Aluminum Al 3 barium Ba 2 bismuth Bi 3 cadmium Cd 2. Write the formulas of the following chemical compounds.
13 iron II phosphide Fe3P2. In the compound zinc phosphide what is the. Most commonly encountered on the binary phosphides ie.
The positive charge more protons versus electrons for a cation is shown by a number and plus sign after the formula. 1 barium oxide Ba and O 4 sodium oxide Na and O Formula. Zinc iodide ZnI2 3.
I Calcium phosphide ZnCO 3 I Zinc carbonate NaNO 3 I Sodium nitrate H 2 SO 3 A Sulfurous acid AgCl I Silver chloride CH 4 M Carbon tetrahydride Cu 2 C 2 O 4 I Copper I oxalate SnI 4 I Tin IV iodide PbS 2 I Lead IV sulfide CO 2 M Carbon dioxide Al 2 SO 4 3 I Aluminum sulfate. Hg23P2 mercury I phosphide Section D Write the formula for the ionic compounds containing transition metals BE CAREFUL TRANSITION METALS MAY HAVE ROMAN NUMERALS and NICKNAMES 1. An ionic compound is formed by the complete transfer of electrons from a metal to a nonmetal and the resulting ions have achieved an octet.
Sodium chloride NaCl and magnesium oxide MgO. Metals lose electrons to produce positve ions called cations. Formula Type Chemical Name HCl A Hydrochloric Acid Hg 2 SO 4 I Mercury I sulfate N 2 O 3 M Dinitrogen trioxide CdS I.
Use the table of ions in Model 1 to answer the following questions. Type II Metal Non-Metal In general it is NOT possible to use the. Optical constants of CaCO 3 Calcium carbonate Calcite Ghosh 1999.
Copy this to my account. Name Formula Systematic Name Common Name Formula Name Formula Methane CH 4 Methanoic acid Formic acid HCO 2H 12-Dichloroethane C 2H 4Cl 2 Ethane C 2H 6 Ethanoic acid Acetic acid CH 3CO 2H Methylamine CH 3NH 2 Propane C 3H 8 Propanoic acid Propionic acid C 2H 5CO 2H Methylammonium ion CH 3NH 3 Butane C 4H 10 Butanoic acid Butyric acid C 3H 7CO. 2-then to get the formula for hydrogen sulfate ion you add a hydrogen ion to the front of the formula.
P 4 S 10. Cadmium sulfide CdS 2. 12 potassium acetate KC2H3O2.
7 VO2 vanadium IV oxide. Since a hydrogen ion has a 1 charge the net charge on the new ion is less negative by one. The chemical formula of ionic compounds can be quickly calculated using the chemical formula calculator.
14 disilicon hexabromide Si2Br6. 2 calcium chloride Ca and Cl 5 sodium nitride Na and N Formula. This activity includes every compound formula and name that can be formed from the list 44 Ions provided in Chemistry A at Pickerington High School Central.
Binary Ionic Formula Practice Name_____ 1 sodium chloride Na1 Cl-1 NaCl 2 lithium bromide Li1 Br-1 LiBr 3 magnesium flouride Mg2 F-1 MgF2 4 potassium oxide K1 O-2 K2O 5 calcium sulfide Ca2 S-2 CaS 6 aluminum iodide Al3 I-1 AlI3 7 barium bromide Ba2 Br-1 BaBr2 8 aluminum sulfide Al3 S-2 Al2S 3 9 calcium phosphide P-3 Ca2 P-3. What is the. E-mail to a friend.
Iron III oxide Fe2O3 4. Zinc carbonate ZnCO 3 aluminum hypochlorite Aℓ C ℓO 3 calcium phosphate Ca 3PO 4 2 cadmium phosphate Cd 3PO 4 2 iron III sulfate Fe 2SO 4 3 mercury II chlorite HgC ℓO 2 2 potassium phosphite K 3PO 3 magnesium. Potassium phosphide K 3P zinc carbide Zn 2C manganese IV sulfide MnS 2 cobalt II bromide CoBr 2.
What is the correct molecular formula for the compound tetraphosphorus decasulfide. K 4 S 10. Circle the symbol for the metal in each of the compounds in Model 2.
N Zinc phosphide Al Aluminum oxide Strontium chloride Sr 4. Name the cation first specifying the charge if necessary then the polyatomic ion as listed in the table. It was designed by Alfred Stock 1876-1946 a German chemist and first published in 1919.
The transfer of electrons between metals and non-metals produces charged particles called ions. If theres just a plus sign it means the charge is plus 1. Gallium phosphide The template Gallium is being considered for deletion Ga The template Phosphorus is being considered for deletion P a phosphide of gallium is a compound semiconductor material with an indirect band gap of 224 eV at room temperature.
Ionic Compounds Containing a Metal and a Polyatomic Ion. Using this program will help you to learn how to write ionic compound names and formulas for Chemistry A. Na Mg 2 Non.
Se 4 F. Formula 21 sodium phosphide Na 3 P 22 magnesium nitrate MgNO 3 2 23 lead II sulfite PbSO 3 24 calcium phosphate Ca 3 PO 4 3 25 ammonium sulfate NH 4 2 SO 4 26 silver cyanide AgCN 27 aluminum sulfide Al 2 S 3 28 beryllium chloride BeCl 2 29 copper I arsenide Cu 3. For example Cu is called CopperI and Cu 2 is called CopperII in the names of compounds containing these ions.
Iron II fluoride FeF2 7. History- The type of naming you will learn about is called the Stock system or Stocks system. Below is a chemistry quiz on ionic compounds names and formulas give it a shot and see if you understood all we covered in this topic on Ions.
Undoped single crystals are orange but. P 2 S 5. Ionic Compound Naming and Formula Writing List 1.
What is the correct molecular formula for the compound selenium tetrafluoride. 6 IO2 iodine dioxide. Give formulas for the following compounds refer to periodic table only.
A monatomic meaning one-atom cation takes its name from the name of the element. Zinc phosphide 29 SrC 2 H 3 O 2 2 strontium acetate 30 Cu 2 O copper I oxide 31 Ag 3 PO 4 silver phosphate 32 YClO 3 yttrium I chlorate 33 SnS 2 tin IV sulfide 34 TiCN 4 titanium IV cyanide 35 KMnO 4 potassium permanganate 36 Pb 3 N 2 lead II nitride 37 CoCO 3 cobalt II carbonate 38 CdSO 3 cadmium sulfite 39 CuNO 2. Ag Silver.
Impure polycrystalline material has the appearance of pale orange or grayish pieces. 8 PbS lead II sulfide. The name or symbol of the metal comes first.
Write the chemical formula for the following ionic compounds. An ionic compound is composed of a metal and a non-metal. Magnesium nitride Mg3N2 6.
5 Ag3P silver phosphide. Review some examples of cations or positive ions.
𝐶𝑎𝐶2 2𝐻2𝑂 𝐶2𝐻2 𝐶𝑎𝑂𝐻2 Calculate the number of hours of service that can be derived from 10 lb of carbide in an acetylene lamp burning 2 cu ft. Examples are shown in Table PageIndex2.

How To Write The Formula For Potassium Hydrogen Phosphate Youtube
IronII chloride or ferrous chloride The cation charge must be specified since iron can form more than one charge.
Hydrogen phosphide chemical formula. I Calcium phosphide ZnCO 3 I Zinc carbonate NaNO 3 I Sodium nitrate H 2 SO 3 A Sulfurous acid AgCl I Silver chloride CH 4 M Carbon tetrahydride Cu 2 C 2 O 4 I Copper I oxalate SnI 4 I Tin IV iodide PbS 2 I Lead IV sulfide CO 2 M Carbon dioxide Al 2 SO 4 3 I Aluminum sulfate. Fumes irritate the eyes and mucous membranes. The nomenclature naming systems for IONIC and MOLECULAR compounds are different.
The total positive charge must balance the total negative charge. Chemical compounds are formed when elements are joined by chemical bonds. Circle the symbol for the metal in each of the compounds in Model 2.
HYDROCHLORIC ACID is an aqueous solution of hydrogen chloride an acidic gas. Formula Type Chemical Name HCl A Hydrochloric Acid Hg 2 SO 4 I Mercury I sulfate N 2 O 3 M Dinitrogen trioxide CdS I. It is much easier to refer to ethanol as ethanol than to refer to it as the organic compound with two.
A compound is a type of molecule with more than one element. Valence e. Hydrogen Nonmetal covalent compound usually Types of Ions.
Chemical Formula - Indicates the number and type of atoms in the base unit of a compound. Hydrochloric acid solution is a colorless watery liquid with a sharp irritating odor. He invited people to sign a petition that demanded either strict control of or a total ban on dihydrogen monoxide.
Na Mg 2 Non. Al 4 C 3 aluminum carbide. For the S- P-blocks.
Metal oxides acetic anhydride propiolactone vinyl acetate mercuric sulphate calcium phosphide formaldehyde alkalies c arbonates strong bases sulphuric acid chlorosulphonic acid. Sodium chloride NaCl and magnesium oxide MgO. N Zinc phosphide Al Aluminum oxide Strontium chloride Sr 4.
Fair experiment that tested anti-technology sentiments and associated chemical phobias in 1997. Chemical formulas for ionic compounds are called ionic formulas The chemical formula for an ionic compound. Main-Group Metals Groups IA IIA and.
Hydrocyanic acid hydrogen cyanide. Two principal formulations of lead arsenate were marketed. It is also used as a fumigant a polymerization initiator and as an intermediate for the preparation of several flame retardants.
The name or symbol of the metal comes first. Sinks and mixes with water. CSUS Chemistry 1A Nomenclature Worksheet Dr.
Compounds Containing Polyatomic Ions. Ca 3 P 2 calcium phosphide. The chemical formula of ionic compounds can be quickly calculated using the chemical formula calculator.
The key to writing proper ionic formulas is simple. These bonds are so strong that the compound behaves like a single substance. An ionic compound is never formed between two cations only or two anions only.
It eventually dissolves nearly anything it comes into. Long before chemists knew the formulas for chemical compounds they developed a system of nomenclature that gave each compound a unique name. Consists of a solution of hydrogen iodide in water.
Wang X Wang X Huang J. The transfer of electrons between metals and non-metals produces charged particles called ions. Consists of hydrogen chloride a gas dissolved in water.
Corrosive to metals and to tissue. Citation needed Lead arsenate was the most extensively used arsenical insecticide. 1327-31-7 and acid lead arsenate.
One oxygen and six hydrogen atoms but one of these molecules can be used as a freeze spray to remove warts and one is a nervous system depressant that makes people drunk. Barium oxide BaO lithium sulfide Li 2S magnesium bromide MgBr 2 strontium iodide SrI 2 calcium sulfide CaS hydrogen bromide HBr lithium oxide Li 2O potassium chloride KC ℓ cadmium fluoride CdF 2 silver sulfide Ag 2S potassium phosphide K 3P zinc carbide Zn 2C. You can go here to learn more about molecules and compounds.
The hydrogen evolution reaction HER is a crucial step in electrochemical water splitting and demands an efficient durable and cheap catalyst if it is to succeed in real applications 123For. For example O. Metals combine with polyatomic ions to give ionic compounds.
Ionic Compounds Containing a Metal and a Polyatomic Ion. Reacts exothermically with carbonates including limestone and building materials containing limestone and hydrogen carbonates to generate carbon dioxide. In a chemical formula subcripts are used to specify the numbers of a type of atom in the formula.
If H is the positive ion the compound is an acid as we will see later on page 6 The positive ion cation may be a monatomic metal ion such as. Basic lead arsenate Pb 5 OHAsO 4 3 CASN. Because the charges on the ions.
An ionic compound is composed of a metal and a non-metal. Type of compound Base unit Ionic Formula unit fu Molecular Molecule Valence Electrons - Electrons in the outermost shell of an atom The only e s involved in bonding and chemical reactions. It is a major component of acid rain.
Today we often use chemical formulas such as NaCl C 12 H 22 O 11 and CoNH 3 6 ClO 4 3 to describe chemical compoundsBut we still need unique names that unambiguously identify each compound. Phosphine has an odor of garlic or decaying. Chemical formula Synonyms CAS number HArF.
Interfacial chemical bond and internal electric field modulated Z-scheme S v-ZnIn 2 S 4 MoSe 2 photocatalyst for efficient hydrogen evolution. Every compound has its own CHEMICAL FORMULA and its own NAME. Gas per hour at a temperature of 75oF and a pressure of 743 mmHg.
Phosphine is used in the semiconductor industry to introduce phosphorus into silicon crystals. Which element comes first in the name and formula of the compounds in Model 2 or the nonmetal. These consist of any positive ion except H combined with any negative ion.
Write the chemical formulas for the following binary ionic compounds. Use the table of ions in Model 1 to answer the following questions. The gas acetylene is produced according to the following reaction by treating calcium carbide with water.
OU Chemical Safety Data No longer updated. Hydriodic acid appears as a colorless to yellow liquid with a pungent odor. Compounds containing polyatomic ions are named similarly to those containing only monatomic ions except there is no need to change to an ide ending since the suffix is already present in the name of the anion.
Hydrogen bromide hydrobromic acid. Lead hydrogen arsenate also called lead arsenate acid lead arsenate or LA chemical formula PbHAsO 4 is an inorganic insecticide used primarily against the potato beetle. Reacts violently with acetic anhydride 2-aminoethanol ammonium hydroxide calcium phosphide chlorosulfonic acid 11-difluoroethylene ethylenediamine ethyleneimine oleum perchloric acid b-propiolactone propylene oxide silver perchloratecarbon tetrachloride mixture sodium hydroxide uraniumIV phosphide vinyl acetate calcium carbide rubidium carbide cesium acetylide rubidium.
Metals lose electrons to produce positve ions called cations. Compounds have their own properties that are unique from the elements they are made of. Reacts exothermically with organic bases amines amides and inorganic bases oxides and hydroxides of metals.
Mack Page 3 of 9 S S2 sulfide ion IA H H hydride ion Polyatomic Ions Polyatomic ions are ions that are composed of two or more atoms that are linked by covalent bonds but that still have a net deficiency or surplus of electrons resulting in. A proper ionic formula has a cation and an anion in it. He listed some of the odious properties of this colorless and odorless substance.
Incompatible with alkalies most metalsAvoid contact with water. OU Chemical Safety Data No longer updated More details. OU Chemical Safety Data No.
Reacts with sulfides carbides borides and.