Benedicts test is performed by heating the reducing sugar with Benedicts reagent. Muby Chemicals of Mubychem Group established in 1976 is the original manufacturers of Specialty Chemicals Pharmaceutical Excipient Fragrance Food Flavor chemicals Reagent Grade Chemicals Shale Gas Fracturing Chemicals in India.

What Is Copper Sulfate Pentahydrate And What Is It Used For Mistral How To
CID 1118 Sulfuric acid CID 23978 Copper CID 962 Water Dates.

What is cupric sulfate pentahydrate. This dark blue to purple solid is a salt of the metal complex CuNH 3 4 H 2 O 2It is closely related to Schweizers reagent which is used for the production of cellulose fibers in the production of rayon. The added word begins with a Greek prefix denoting the number of water molecules see Table 5 and ends with hydrate For example the anhydrous compound copperII sulfate also exists as a hydrate containing five water molecules and named copperII sulfate pentahydrate. No OSHA Vacated PELs are listed for this chemical.
Their Chemical Abstracts Service CAS registry numbers are 1344-73-6 1332-14-5 7758-99-8 and 7758- 98-7 respectively. 5H 2 O sodium citrate Na 3 C 6 H 5 O 7 and sodium carbonate Na 2 CO 3 in distilled water 4. The chemical formula of Benedicts solution is not present since the benedicts solution is a mixture and not a chemical compound.
The anhydrous form is a pale green or gray-white powder whereas the pentahydrate the most commonly encountered salt is bright blue. Melting point 110C with decomposition. GL8800000 anhydrous GL8900000 pentahydrate คณสมบต สตรเคม.
002 hydrate Analysis Pre Lab v1 Docx Name Date H. As silver salts have antiseptic properties they have been used as a treatment for gonorrhea prevention a cauterizing agent for the healing of oral ulcers among others. Ammonium Sulfate Ammonium ions and Sulfate ions NH 4 and SO 4 2 NH 4 2SO 4 Calcium Phosphate Calcium ions and Phosphate ions Ca2 and PO 4 3 Ca 3PO 2 Waters of hydration use table to left copperII sulfate pentahydrate CuSO 45H 2O 11 Undeca.
The main target users are workers and those responsible for occupational safety and health. TetraamminecopperII sulfate is the salt with the formula CuNH 3 4SO 4 H 2 O. None listed OSHA Vacated PELs.
When the conditions are carefully controlled the colouration developed and the amount of precipitate formed Cuprous oxide depends upon the amount of reducing sugars present. Cupric sulfate anhydrous none listed 1 mgm3 TWA as Cu except Copper fume listed under Copper compounds nos100 mgm3 IDLH dust and mist as Cu listed under Copper compounds nos. The ICSC project is a common undertaking between the World Health Organization WHO and.
Formulations include basic copper sulfate copper sulfate monohydrate copper sulfate pentahydrate and copper sulfate anhydrous. Silver nitrate is an inorganic compound discovered in the 13 th century by Albertus Magnus and has since been used in the medical industry for various issues including wound care. CopperII sulfate also known as copper sulphate are the inorganic compounds with the chemical formula Cu SO 4 H 2 O x where x can range from 0 to 5The pentahydrate x 5 is the most common form.
The liver is the critical organ for the homeostasis of copper. Cupric sulfate may also have a role in iron turnover ascorbic acid metabolism phospholipid metabolism myelin formation glucose homeostasis and cellular immune defense. Older names for this compound include blue vitriol bluestone vitriol of copper and Roman vitriol.
Copper II sulfate exothermically dissolves in water to give the aquo complex CuH2O62 which has octahedral molecular geometry and is paramagnetic. Other names for copperII sulfate are blue vitriol and. Magnesium sulfate is gaining popularity as an initial.
The pentahydrate CuSO 4 5H 2 O the most commonly encountered salt is bright blue. Magnesium is reacted with oxygen from the air in a crucible and the masses before and after the oxidation are measured. The systematic name is determined by guidelines set forth by the International Union of Pure and Applied Chemistry IUPAC.
61 gmol Q3 - 5 Q4 - CuSO 4 5H 2 O Q5 - copperII sulfate pentahydrate Q6 - Turned back to blue hydrateComposition of Hydrates. No OSHA Vacated PELs are listed for this chemical. The cards are data sheets intended to provide essential safety and health information on chemicals in a clear and concise way.
The systematic name also called the IUPAC name is the preferred way to name a chemical because each systematic name identifies exactly one chemical. They can reduce cupric ions Cu 2 to cuprous form Cu which is responsible for the change in color of the reaction mixture. Copper sulfate pentahydrate appears as blue crystalline granules or powder.
The mass of the hydrated salt obtained was 7. Pesticides containing copper sulfate monohydrate andor copper sulfate anhydrous have been cancelled by the United States Environmental. The primary aim of the cards is to promote the safe use of chemicals in the workplace.
IP BP EP Ph Eur USP NF JP Analytical Reagent FCC Food Grade Chemicals Excipients Shale Gas Fracturing Chemicals Manufacturers. Washing soda is the common name for a hydrate of sodium carbonate. 5H 2 O sodium carbonate Na 2 CO 3 sodium citrate Na 3 C 6 H 5 O 7 and distilled water.
This is the basis of Benedicts test. Magnesium sulfate is a small colorless crystal used as an anticonvulsant a cathartic and an electrolyte replenisher in the treatment of pre-eclampsia and eclampsia. Benedicts solution is a deep-blue alkaline chemical reagent used to test for the presence of the aldehyde functional group -CHO which consists of copper sulfate pentahydrate CuSO 4.
Zinc Sulfate Monohydrate Our Associate Companies A progressive and dynamic company Buradon is one of India s leading manufacture and exporter of fine chemicals to the rigid standard of GR AR EL ACS and LR grades due to its industrial exposure commitment and market fixing capabilities from India. The presence of the alkaline sodium carbonate converts the sugar into a strong reducing. After the metal passes through the basolateral membrane it is transported to the liver attached to serum albumin.
The copper is then prepared for excretion. Hydrated ionic compounds ie hydrates have a specfic number of water molecules in their chemical formulas. Composition and Preparation of Benedicts.
Systematic or IUPAC Name. It causes direct inhibition of action potentials in myometrial muscle cells. Benedicts solution is made by dissolving 173 g monosodium citrate 173 g cupric sulphate pentahydrate and 100 g anhydrous sodium carbonate in distilled water.
Cupric sulphate Blue vitriol pentahydrate Bluestone pentahydrate Bonattite trihydrate mineral Boothite heptahydrate mineral Chalcanthite pentahydrate mineral Chalcocyanite mineral Copper Sulphate pentahydrate. Sodium carbonate renders alkaline conditions which are required for the redox reaction while sodium citrate is a. Cupric sulfate Blue vitriol Bluestone Chalcanthite.
7758-98-7 7758-99-8 pentahydrate 16448-28-5 trihydrate 19086-18-1 heptahydrate 3D 모델. It is a bright blue solution prepared by mixing copper sulfate pentahydrate CuSO 4. เลขทะเบยน เลขทะเบยน CAS 7758-98-7 7758-99-8 pentahydrate PubChem.
Copper sulfate is a term that can refer to either of the following chemical compounds cuprous sulfate Cu 2 SO 4 or cupric sulfate CuSO 4. The systematic name for CuSO 4 is copperII sulfate but it is also referred to as blue vitriol Roman vitriol the vitriol of copper and bluestone. Excitation and contraction are uncoupled which decreases the frequency and force of contractions.
The final product can be employed to check for the presence of reducing sugars. 식별자 CAS 번호. Principle of Benedicts Test.
However the latter is the preferred compound described by the term copper sulfate. Benedicts solution or Benedicts reagent can be prepared by complexing cupric ions Cu2 cations from the copper sulfate pentahydrate with citric acid molecules in a basic environment provided by sodium carbonate. In the solid these water molecules also called waters of hydration are part of the structure of the compound.
Benedicts solution is made by dissolving 173 g monosodium citrate 173 g cupric sulphate pentahydrate and 100 g anhydrous sodium carbonate in distilled water. Pesticides containing copper sulfate monohydrate andor copper sulfate anhydrous have been cancelled by the United States Environmental.

Copper Ii Sulfate Pentahydrate 99 Ph Eur Grade Cas 7758 99 8 Glentham Life Sciences
The pentahydrate CuSO 4 5H 2 O the most commonly encountered salt is bright blue.
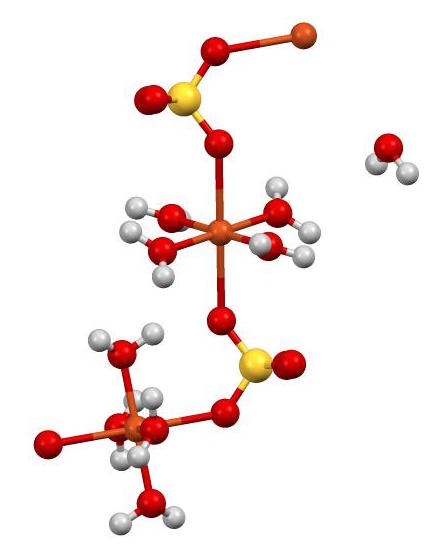
Cupric sulfate pentahydrate. Cupric sulfate anhydrous none listed 1 mgm3 TWA as Cu except Copper fume listed under Copper compounds nos100 mgm3 IDLH dust and mist as Cu listed under Copper compounds nos. Cupric sulphate Blue vitriol pentahydrate Bluestone pentahydrate Bonattite trihydrate mineral Boothite heptahydrate mineral Chalcanthite pentahydrate mineral Chalcocyanite mineral Copper Sulphate pentahydrate. Aluminum nitrite AlNO 23 64.
Cupric sulfate may also have a role in iron turnover ascorbic acid metabolism phospholipid metabolism myelin formation glucose homeostasis and cellular immune defense. No OSHA Vacated PELs are listed for this chemical. Agriculture soil additive pesticides Bordeaux mixture water treating applications animal feed additive germicides textile mordant leather industry pigments electric batteries electroplated coatings copper salts reagent in analytical chemistry medicine wood preservative preservation of pulp wood and ground pulp.
Formulations include basic copper sulfate copper sulfate monohydrate copper sulfate pentahydrate and copper sulfate anhydrous. Cupric bromate CuBrO 22 67. 002 hydrate Analysis Pre Lab v1 Docx Name Date H.
Copper sulfate pentahydrate appears as blue crystalline granules or powder. After the metal passes through the basolateral membrane it is transported to the liver attached to serum albumin. Ammonium Sulfate Ammonium ions and Sulfate ions NH 4 and SO 4 2 NH 4 2SO 4 Calcium Phosphate Calcium ions and Phosphate ions Ca2 and PO 4 3 Ca 3PO 2 Waters of hydration use table to left copperII sulfate pentahydrate CuSO 45H 2O 11 Undeca.
The systematic name also called the IUPAC name is the preferred way to name a chemical because each systematic name identifies exactly one chemical. No OSHA Vacated PELs are listed for this chemical. Sodium carbonate is used to maintain an alkaline medium required for the redox reaction during the test.
Sodium carbonate renders alkaline conditions which are required for the redox reaction while sodium citrate is a. Beryllium chloride BeCl2 91. However the latter is the preferred compound described by the term copper sulfate.
The final product can be employed to check for the presence of reducing sugars. This dark blue to purple solid is a salt of the metal complex CuNH 3 4 H 2 O 2It is closely related to Schweizers reagent which is used for the production of cellulose fibers in the production of rayon. Silver nitrate is an inorganic compound discovered in the 13 th century by Albertus Magnus and has since been used in the medical industry for various issues including wound care.
Composition and Preparation of Benedicts. Silver oxide Ag 2O 65. The final volume of the solution is one litre.
When the conditions are carefully controlled the colouration developed and the amount of precipitate formed Cuprous oxide depends upon the amount of reducing sugars present. CID 1118 Sulfuric acid CID 23978 Copper CID 962 Water Dates. Copper II sulfate exothermically dissolves in water to give the aquo complex CuH2O62 which has octahedral molecular geometry and is paramagnetic.
Benedicts solution or Benedicts reagent can be prepared by complexing cupric ions Cu2 cations from the copper sulfate pentahydrate with citric acid molecules in a basic environment provided by sodium carbonate. Older names for this compound include blue vitriol bluestone vitriol of copper and Roman vitriol. Magnesium sulfate is gaining popularity as an initial.
CopperII sulfate also known as copper sulphate are the inorganic compounds with the chemical formula Cu SO 4 H 2 O x where x can range from 0 to 5The pentahydrate x 5 is the most common form. Hydrated ionic compounds ie hydrates have a specfic number of water molecules in their chemical formulas. Calcium bicarbonate CaHCO 3 92.
As silver salts have antiseptic properties they have been used as a treatment for gonorrhea prevention a cauterizing agent for the healing of oral ulcers among others. When hydrogen sulphide gas is passed through a blue solution of copper sulphate a black precipitate of copper sulphide is obtained and the sulphuric acid so formed remains in the solution. In the solid these water molecules also called waters of hydration are part of the structure of the compound.
식별자 CAS 번호. In this reaction cupric ions from copper sulphate combine with sulphide ions from hydrogen sulfide to. Hydrobromic acid HBr 89.
Magnesium chloride pentahydrate 86. TetraamminecopperII sulfate is the salt with the formula CuNH 3 4SO 4 H 2 O. Magnesium is reacted with oxygen from the air in a crucible and the masses before and after the oxidation are measured.
Systematic or IUPAC Name. 61 gmol Q3 - 5 Q4 - CuSO 4 5H 2 O Q5 - copperII sulfate pentahydrate Q6 - Turned back to blue hydrateComposition of Hydrates. It causes direct inhibition of action potentials in myometrial muscle cells.
So sodium citrate is used to stabilize the copper ions by complexing with them. Benedicts solution is a deep-blue alkaline chemical reagent used to test for the presence of the aldehyde functional group -CHO which consists of copper sulfate pentahydrate CuSO 4. 5H 2 O sodium carbonate Na 2 CO 3 sodium citrate Na 3 C 6 H 5 O 7 and distilled water.
Sulfur trioxide SO 3 63. The chemical formula of Benedicts solution is not present since the benedicts solution is a mixture and not a chemical compound. Melting point 110C with decomposition.
Hydrogen fluoride HF 88. The liver is the critical organ for the homeostasis of copper. TinII sulfide SnS 90.
The CuSO 45H 2 Ocopper sulfate pentahydrate salt is the source for the blue color due to the presence of copperII ions. Copper sulfate is a term that can refer to either of the following chemical compounds cuprous sulfate Cu 2 SO 4 or cupric sulfate CuSO 4. Ammonium sulfate NH 42SO 4 87.
Excitation and contraction are uncoupled which decreases the frequency and force of contractions. The copper is then prepared for excretion. They can reduce cupric ions Cu 2 to cuprous form Cu which is responsible for the change in color of the reaction mixture.
Calcium nitride Ca 3N2 MgCl25H 20 62. Zinc Sulfate Monohydrate Our Associate Companies A progressive and dynamic company Buradon is one of India s leading manufacture and exporter of fine chemicals to the rigid standard of GR AR EL ACS and LR grades due to its industrial exposure commitment and market fixing capabilities from India. Copper II can sometimes degrade to copperI.
Water is just added as a solvent for. IP BP EP Ph Eur USP NF JP Analytical Reagent FCC Food Grade Chemicals Excipients Shale Gas Fracturing Chemicals Manufacturers. None listed OSHA Vacated PELs.
The systematic name is determined by guidelines set forth by the International Union of Pure and Applied Chemistry IUPAC. Magnesium sulfate is a small colorless crystal used as an anticonvulsant a cathartic and an electrolyte replenisher in the treatment of pre-eclampsia and eclampsia. The anhydrous form is a pale green or gray-white powder whereas the pentahydrate the most commonly encountered salt is bright blue.
Ammonium phosphate NH 43PO 4 66. The mass of the hydrated salt obtained was 7. Other names for copperII sulfate are blue vitriol and.
7758-98-7 7758-99-8 pentahydrate 16448-28-5 trihydrate 19086-18-1 heptahydrate 3D 모델. This is the basis of Benedicts test. The systematic name for CuSO 4 is copperII sulfate but it is also referred to as blue vitriol Roman vitriol the vitriol of copper and bluestone.
Product Applications for Copper Cupric Sulfate Pentahydrate. Muby Chemicals of Mubychem Group established in 1976 is the original manufacturers of Specialty Chemicals Pharmaceutical Excipient Fragrance Food Flavor chemicals Reagent Grade Chemicals Shale Gas Fracturing Chemicals in India. Their Chemical Abstracts Service CAS registry numbers are 1344-73-6 1332-14-5 7758-99-8 and 7758- 98-7 respectively.