2 kg calcium hydrogen phosphate to moles. How many moles of silver are in the ring.
Notified of all changes on this drug.
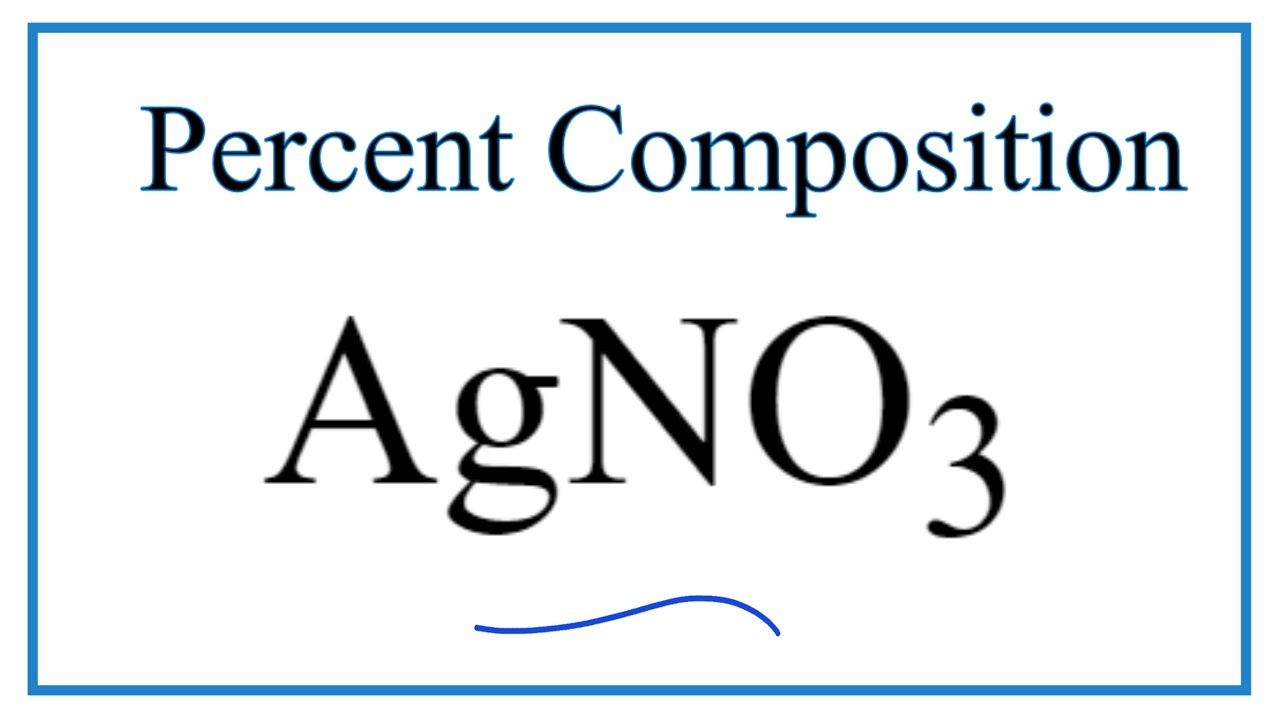
Silver nitrate mass. They are each made of a ca. The actual yield was 220g how do you calculate percent yield. A photon of light hitting the negative nitrate anion frees an electron which ultimately combines with the positive silver ions to make neutral silver metal darkening the surface of the material.
Calculate the molar mass for each of the following substances. 3 c magnesium nitrate MgNO. Targets 1 Enzymes 1 Transporters 1 Potassium nitrate.
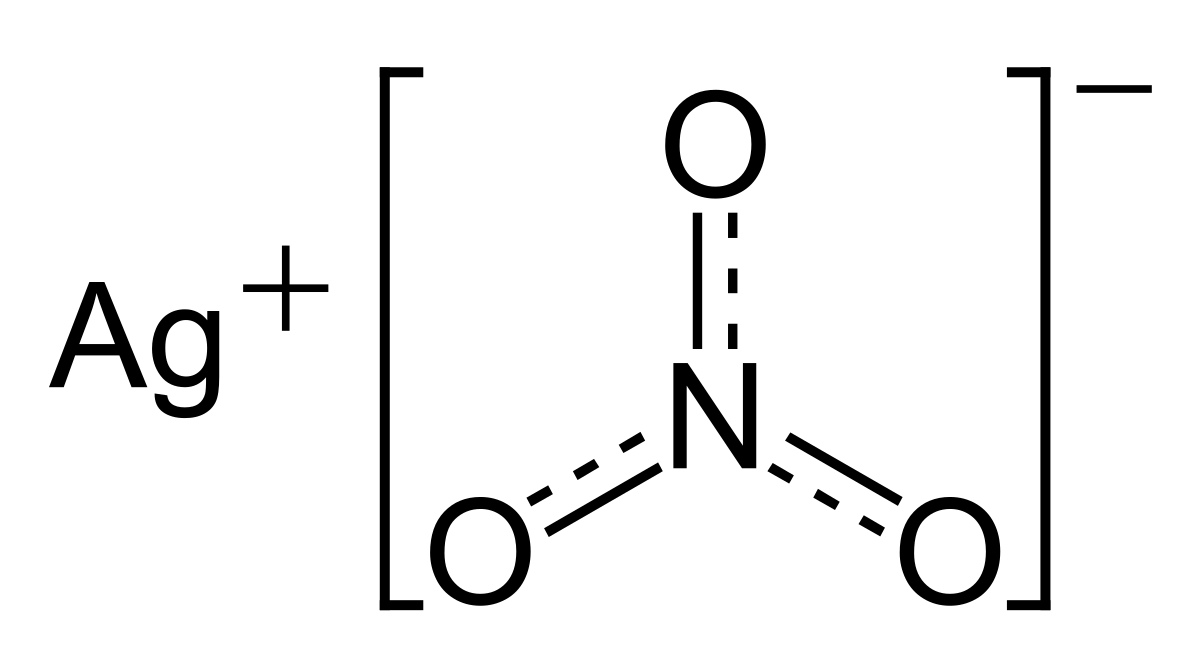
Convert quantities to volumes. Silver nitrate is an inorganic compound with chemical formula AgNO 3This salt is a versatile precursor to many other silver compounds such as those used in photographyIt is far less sensitive to light than the halidesIt was once called lunar caustic because silver was called luna by the ancient alchemists who associated silver with the moon. Find the number of moles from a given mass.
In this case both of the reactants are salts. A 50 reduction in the radioactivity content of spleen occurred after about 70 hr while that of brain took about 84 hr. Silver injected into rats as radioactive silver nitrate had a half-time of 22 days.
Find the equivalent mass of silver. 1030 atoms of helium to liters. Therefore silver sulfadiazines effectiveness as an antimicrobial agent for preventing burn wound infections is due to its tendency.
1 mol C 1201 g C or 12. To calculate chlorides solution concentration use. The mass of A g C l ceAgCl A g C l formed is 7175 g 7175text.
The molar mass of magnesium nitrate equals 14833 gmol. Silver nitrate is used in many ways in organic synthesis eg. Titrate with silver nitrate solution till the first color change.
It can be calculated by adding the invididual molar mass of every atom that are composing the molecule CH4. Calculate the mass of silver bromide which should be produced from 225g of silver nitrate. I will give the lowest level of explanation and then follow up with a high level explanation.
Silver occurs natively and. For instance if you had a 800 g sample of a compound that was 200 g element X and 600 g element y then the percent composition of each element would be. In solid silver nitrate the silver ions are.
Percent composition in chemistry typically refers to the percent each element is of the compounds total mass. For rats given a single ip injection of radioactive silver nitrate a 50 reduction in radioactivity of whole blood plasma kidney liver after approx 40 hr. A silver ring contains 11 x 1022 silver atoms.
The molar mass of silver nitrate is 169872 grams per mole. In this experiment the amount of chloride in an unknown sample was determined by. 245 mol Cu 6022 1023 atoms 1 mol 18 10 148 1024 atoms Cu 11 1022 atoms Ag 1 mol 6022 1023 atoms -2 mol Ag Molar Mass.
7 5 g of A g C l ceAgCl A g C l. Its density in the liquid state at a temperature of 210 o C corresponds to 397 gcm 3. Due to these toxic effects many countries have placed severe restrictions on the amount of their utilisation in processed food products.
The mass of 1 mol of atoms of an element An elements molar mass in gmol is numerically equal to the elements atomic mass in amu. Silver nitrate or lunar caustic the most important silver compound is used extensively in photography. What occurs to the theoretical yield if a sample is incompletely dried.
Los Alamos National Laboratory US. Silver NH 4 ammonium Ca2 calcium Fe2 ironII Cu2 copperII Mg2 magnesium Ni2 nickelII Sr2 strontium Zn2 zinc Sn2 tinII Pb2 leadII Ba2 barium Fe3 ironIII Al3 aluminium Cr3 ChromiumIII F-fluoride Cl-chloride Br-bromide I-iodide HO-hydroxide NO 2-nitrite NO 3-nitrate HCO 3-hydrogencarbonate O2-oxide CO 3 2-carbonate SO 4 2-sulfate HSO 4-hydrogensulfate S2-sulphide N3. The biological half-time of.
Answer 1 of 5. When in 1840 Henry Talbot discovered an additional chemical twist that is so called latent silver image that had been briefly exposed onto a layer of silver iodide could be revealed using gallic. When you get questions like this it helps a lot if you can see the general sorts of patterns.
Discussion The well known Mohrs method in which alkaline or alkaline earth chlorides react with silver nitrate in the presence of a few drops of potassium chromate solution as indicator is a simple direct and accurate method for chloride determination. It is solid white or sometimes white to dirty grey in colour. Calculate the molar mass for each of the following substances.
6 c strontium acetate SrC. Formation of nitrite and propagation of its downstream NO-signaling effects depends on the oral bacterial reduction of inorganic nitrate by a set of bacterial nitrate reductase enzymes NaRs that are largely absent from the human genome In a process referred to as the enterosalivary nitrate circulation or the nitrate-nitrite-NO pathway dietary nitrate is swallowed and rapidly. The melting and boiling points of silver nitrate are 4828 K and 713 K respectively.
Nitrate is also recognized as hazardous on the basis that it can be easily reduced to nitrite in-vivo. Isotopes of silver range in relative atomic mass from 92950 u 93 Ag to 129950 u. A silver metal Ag b ammonia gas NH.
Excessive amount of nitrite and nitrate in food has been a major concern of food safety thus a highly sensitive selective speedy. Notice in Figure 6 how silver sulfadiazine Ag sulfadiazine gradually releases the majority of its silver ions into solution over an extended period of time whereas silver nitrate immediately released all of its silver ions into solution Fox and Modak 1974. 12 lbs of 4-cyanoindole.
It is an alkali metal nitrate because it is an ionic salt of potassium ions K ions and nitrate ions NO 3. Later a displacement reaction takes place when N a C l ceNaCl N a C l is added to A g N O X 3 ceAgNO3 A g N O X 3 to form 7175 g 7175text g 7 1. For deprotection and oxidations.
Atomic mass of Cl- 3545 gmole mmoles of Cl- A M. A 5494 g. Potassium nitrate contains potassium a soft light and silver metal oxygen and nitrogen a colourless and odourless gas.
AgNO 3 as a colourless appearance in its solid state and is odourless. Potassium nitrate is a medication used to cauterize small wounds remove granulation tissue warts and verrucae and treat tooth sensitivity. According to the reaction equation.
What occurs to the reported yield. Compute properties of a chemical solution. Notified of marketing changes.
Silver for centuries has been used traditionally for coinage by many countries of the world. The resulting adduct can be decomposed with ammonia to release the free alkene. Chemical Solutions Explore properties of different solutions which depend on the solvent solute and concentration.
In recent times however consumption of silver has greatly exceeded the output. Ag binds alkenes reversibly and silver nitrate has been used to separate mixtures of alkenes by selective absorption. Ag Cl- AgCl.
500mg of silver nitrate. In a double replacement reaction between silver nitrate and magnesium bromide. In its solid state it has a density of 435 grams per cubic centimetre.
A manganese metal Mn b sulfur hexafluoride SF. That makes calculation especially easy - when we calculate number of moles of AgNO 3 used it will be already number of moles of Cl-titrated. Silver nitrate reacts with chloride anion on the 11 basis.
The molar mass of a substance also often called molecular mass or molecular weight although the definitions are not strictly identical but it is only sensitive in very defined areas is the weight of a defined amount of molecules of the substance a mole and is expressed in gmol. Potassium nitrate is soluble in hot water. 54 g 54text g 5 4 g of silver reacts with H N O X 3 ceHNO3 H N O X 3 to form silver nitrate.
The basic equation mass of element mass of compound X 100.
The nutritional requirement for iodine under review by WHO is currently considered to be in the range of 010 to 014 mg per person per day for adults see potassium iodateThe Committee set a provisional maximumtolerable daily intake of 1 mg iodineday 0017 mgkg bw from all sources. There are 46 species of flowering plant of the genus Urtica Table 1The most prominent members of the genus are the stinging nettle Urtica dioica L.
Cholesterol is the main zoosterol but sterols in plants commonly occur as mixtures with β-sitosterol campesterol and stigmasterol representing three of the major phytosterolsThese sterols are all Δ 5-sterols Figure 6 but Δ 7-sterols may also be.
Ethanolic silver nitrate weight. 5 grams of glucose is about equivalent to a small sugar packet or a teaspoon full of sugarTo be considered a non-diabetic the American Diabetes Association recommends a post-meal glucose level less than 180 mgdl 10 mmoll and a pre-meal blood glucose level of 90-130 mgdl 5 to. Total body water includes more than merely blood and will be usually about 60 of the total body weight in men. Of course this method is useful for calculating the corrosion inhibition efficiency.
The volumes of. Average weight of each capsule is 550 mg with 222 WW weight variation. Wako和光纯药代理 166-07055 n2 potassium hydroxide solution ethanolic 1310-58-3.
40 45 kcal from fat Saturated fatty acids SFA 60 of total fatty acids Milkfatbutterfat. Wako和光纯药代理 166-03035 polyphospholic acid 8017-16-1. Pakistan journal of biological sciences.
Masters thesis Universiti Sains Malaysia. The method of using weight loss coupon for determining corrosion damage is very useful for monitoring corrosion and also for investigating the environmental condition which cannot be simulated in laboratory. Metal-based nanoparticles can be prepared by utilizing different biological physical and chemical methods.
The variation of weight among the capsules was least which showed a good ratio of excipients in the formulation. Ying-Chih Liao Langmuir 2021 37 46 13689-13695 Article Publication Date Web. Sucrose 34 by weight Cholesterol 02 total References.
Sterols occur in the membranes of plants animals and microorganisms and are termed phytosterols zoosterols and mycosterols respectively. Wako和光纯药代理 166-04032 potassium nitrate 7757-79-1. J Clin Invest 2000.
Organic Chemistry vol 2 - IL. The biological method is. The variation of weight among the capsules was least which showed a good ratio of excipients in the formulation.
Response surface methodology was utilized to evaluate the effects of the two independent variables on particle size and concentration of the. Febbraio M et al Targeted disruption of the class B scavenger receptor CD36 protects against atherosclerotic lesion development in mice. Capsule contents of twenty capsules from each batch were collected and stored in amber coloured container at room temperaturePhytochemical standards phyllanthin berberine chloride dihydate gallic acid.
Average weight of each capsule is 550 mg with 222 WW weight variation. Self-Healing Silicone Elastomer with Stable and High Adhesion in Harsh Environments. Microwave-Assisted Synthesis for Silver Nanoplates with a High Aspect Ratio.
A Absolute molecular weight b Average molecular weight c Low molecular weight d Absolute melting point 15. All of the PLA films were equilibrated at 50 RH in a chamber containing magnesium nitrate saturated solution at 20 C. Effect of some environmental indicators on physiological disturbances and endocrine disorders in Tilapiazillii Gervais 1848 and Cyprinuscarpio l 1758.
Shengping Dai Meng Li Hao Yan Hao Zhu. Based on randomly central composite design 13 mixture solutions containing different amounts of the prepared extract 010090 mL and 1 mM silver nitrate solution 1525 mL were prepared and exposed to microwave irradiation for 180 s. Synthetic human hair wigs are made from a copolymer of vinyl chloride and.
In this low cost method small samples in a specific duration of time are immersed in corrosive media. The least MIC was 16mgml against Salmonella typhi while Bacillus subtilis and Staphylococcus aureus showed. In Selected Forest Reserve In Northern Region Of Peninsular Malaysia.
Body Weight Gain Nutrients Degradability and Fermentation Rumen Characteristics of Boerka Goat Supplemented Green Concentrate Pellets GCP Based on Indigofera zollingeriana. Huszar D et al. TS MPa was calculated by dividing the maximum load on the PLA film before failure by the cross.
Capsule contents of twenty capsules from each batch were collected and stored in amber coloured container at room temperaturePhytochemical standards phyllanthin berberine chloride dihydate. Rusdi Nur Diana Mohd 2019 Population Dynamics Of Calamus Castaneus Griff. A Tarigan SP Ginting A Ii DA Astuti L Abdullah.
This level may cause adverse effects for some individuals eg. Chemical name of melamine is. AL-Saeed MH AL-Saeed AH Alfaris AA Jori MM.
The variation of weight among the capsules was least which showed a good ratio of excipients in the formulation. And the small nettle U. Urens L which are native to Europe Africa Asia and North America.
Iodine is an essential micronutrient. Academiaedu is a platform for academics to share research papers. Yaccob Nur Syahirah 2019 Characterisation Of Nitrate Stable Isotopes To Identify Nitrate Sources In Bukit Merah Reservoir Perak.
Primary standards are defined via other quantities like length mass and timePrimary standards are used to calibrate other standards referred to as working standards. The ethanolic extract of the peels had MIC values ranging from 16mgml to 5125mgml. If the loss in weight is 280 per cent find the amount of lead nitrate and sodium nitrate in the mixture.
Average weight of each capsule is 550 mg with 222 WW weight variation. Wako和光纯药代理 166-04836 2-propanoliso propyl alcohol 67-63-0. Gervais 1848 CyprinuscarpioL 1758 Citation.
A primary standard in metrology is a standard that is sufficiently accurate such that it is not calibrated by or subordinate to other standards. PJBS 21 2 87-94 vol. PLA granules with a molecular weight of 197000 gmol and a density of 13.
High Fat Diet 20-23 by weight. An equal volume of a reducing agent is titrated separately with 1M KMnO4 in acid neutral and alkaline media. The primary grip separation and crosshead speed were set at 50 mm and 50 mmmin respectively.
1990 - 4 Marks 54. Nanotechnology emerged as a scientific innovation in the 21st century. The genus Urtica belongs to the family Urticaceae in the major group Angiosperms flowering plants.
Habitats of Urtica spp. A solid mixture 50 g consisting of lead nitrate and sodium nitrate was heated below 600ºC until the weight of the residue was constant. Yi-Chin Lai Yu-Chi Wang Yu-Chieh Chiu and.
Evaluation of mating behaviour and mating compatibility methods for the Old. A 2 4-diamino1 3 5-triazine b 2-amino1 3 5-triazine c 2 4 6-triamino1 3 5-triazine d 1 3 5-triamino-2 4 6-triazine 16. Metallic nanoparticles metal or metal oxide nanoparticles have attained remarkable popularity due to their interesting biological physical chemical magnetic and optical properties.
With our money back guarantee our customers have the right to request and get a refund at any stage of their order in case something goes wrong. Capsule contents of twenty capsules from each batch were collected and stored in amber coloured container at room temperaturePhytochemical standards phyllanthin berberine chloride dihydate.