How many grams of NaCl are required to prepare 985 mL of 077 M NaCl solution. Dichlorine - Cl 2.

Find The Molecular Mass Of Mgso4 7 H2o Brainly In
It can be calculated by adding the invididual molar mass of every atom that are composing the molecule CH4.
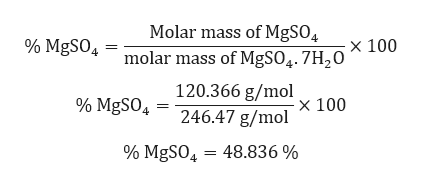
Mgso4 molar mass. MgSO_4 7H_2O The idea here is that heating the hydrate will drive off the water of evaporation and leave behind the anhydrous salt. MgSO47H2O molecular weight. 5 H2O CuOH2 H2SO4 K2Cr2O7 NaCl CaC4H4O6 MgCrO4 Al2SO43 K3PO4 ZnCl2 COMPOUND FORMULA MASS CuSO4.
C mass of water mass of hydrate mass of anhydrate 6923 g 3382 g 3541 g H2O d Calculate molar masses of the components of the hydrate. 24305 32065 1599944 71007942 159994 Percent composition by element. Blamp Created Date.
A few things to consider when finding the molar mass for HClO4- make sure you have the c Perchloric Acid HClO4 Molar Mass Molecular Weight. 52 gram per cubic centimeter or 2 520 kilogram per cubic meter i. MgSO4 where MM MgSO4 the molar mass of MgSO 4 Moles of water lost M 2 W 3MM H2O where MM H2O the molar mass of H 2O Ratio of moles M 2M 1 Do division and report first number with 2 significant digits.
Metal carbonates are weak bases. Northern Arizona University and Raymond Chang this success guide is written for use with General Chemistry. Heat and Chemical Changes 42.
Magnesium is a Group 2 alkaline earth element within the periodic table and has a relative atomic mass of 24305 Da a specific gravity at 20C of 1738 2 3 a melting point of 6488C and a boiling point of 1090C. Molar mass of MgSO4 1203676 gmol. Formula Mass 1641 MgSO4.
Masas Moleculares Calculadas Recientemente. Divide the weight of the compound by the compounds molar mass measured in grams to obtain the number of moles. 892007 101757 AM.
Scacchite - MnCl 2. This compound is also known as Magnesium Sulfate. To determine the percentage of morphine in the white powder the mass of morphine reacted was divided by the total mass of the white sample 0008094g0010g 100 810.
What is the molarity of an aqueous solution of sodium hydroxide produced when 350 ml of a 540 M solution was diluted to 8900 ml. 0450 kg of octane C8H18 burned during combustion produces 125 kg of carbon. It aims to help students hone their analytical and problem-solving skills by presenting detailed approaches to solving chemical problems.
Water - H 2 O. 0063432 gmol Molar mass of MgSO4 is 120. CHEM 232 Organic Chemistry for Sodium chloride ˌsoʊdiəm ˈklɔːraɪd commonly known as salt although sea salt also contains other chemical salts is an ionic compound with the chemical formula NaCl representing a 11 ratio of sodium and chloride ions.
Magnesium sulfate or magnesium sulphate in British English is a chemical compound a salt with the formula MgSO 4 consisting of magnesium cations Mg 2 2019 by mass and sulfate anions SO 2 4It is a white crystalline solid soluble in water but not in ethanol. You can also ask for help in our forums. Use the molecular formula to calculate the molar mass.
Element Family Reunion 51. When you put sodium carbonate Na2CO3 in water it produces 2 sodium ions and one carbonate ion. Thus the hydrated magnesium cation is hard to dehydrate.
The molar mass of a substance also often called molecular mass or molecular weight although the definitions are not strictly identical but it is only sensitive in very defined areas is the weight of a defined amount of molecules of the substance a mole and is expressed in gmol. Lea nuestro artículo sobre cómo calcular la masa molar. Potassium Sulfate - K 2 SO 4.
What is the theoretical yield of a reaction if the percent yield 924 and product recovered is 893 g. Which of the following is a strong acid. Academiaedu is a platform for academics to share research papers.
In the dissolved state magnesium binds hydration water tighter than calcium potassium and sodium. MnCl2 Molar Mass MnCl2 Oxidation Number. Convert grams MgSO4 to moles or moles MgSO4 to grams.
Convert grams MgSO47H2O to moles or moles MgSO47H2O to grams. Mystery Anions 70. Hydrogen Chloride - HCl.
Magnesium sulfate is usually encountered in the form of a hydrate MgSO 4 nH 2 O for various values of n between 1 and 11. With molar masses of 22. The Colligative Properties of Solutions 39.
Subsequently question is how do you find the moles of water in hydrated MgSO4. What does that mean regarding the metal ions that make up the carbonate salt. Hoh Aqua Oh2 H₂O Oxidane Pure Water Hydroxic Acid Hydrogen Oxide H2O Molar Mass H2O Oxidation Number.
Organizing Topic Electron Configuration and the Periodic Table 49. 24305 32065 1599944 Percent composition by element. K2So4 Kaliumsulfat Potassium Sulphate Dipotassium Sulfate K2SO4 Molar Mass K2SO4 Oxidation Number.
They react with acids to release carbon dioxide. 1 Show answers Another question on Chemistry. Microsoft Word - HYDRATESdoc Author.
C2H52O Ether NH42C2O4 Ammonium Oxalate NH42CO3 Ammonium Carbonate NH42CrO4 Ammonium Chromate NH42HPO4 Di-Ammonium Phosphate NH42S Ammonium Sulfide NH42SO4 Ammonium Sulfate NH43PO3 Ammonium Phosphite NH43PO4 Ammonium Phosphate Ag2O SilverI Oxide Ag2S Silver Sulfide Ag2SO4 Silver. Cl₂ Chlorine Gas Bond Polarity. If the molar mass of the salt is 218 gmol what mass is required.
Molar Heat of Fusion for Water 36. Organizing Topic Bonding Nomenclature and Formula Writing 59. Calculadora de Estequiometría de las.
A Crystal Lab 61. Atomic Mass of Atoms. If you use 0001 gl as your concentration and desire to convert say 250 ppm Magnesium sulfate to molar concentration you could actually multiply the ppm concentration by 0001 gl then divide.
What is the mass percent of MgCO33H2O in the mixture. Report second number as 1 Title. 최근 계산한 몰 질량.
In your case the anhydrous salt is magnesium sulfate MgSO_4. H2O CaOH2 AlCl3 D. Round atomic masses to the tenth of a decimal place.
Hcl HCl Molar Mass Bond Polarity HCl Oxidation Number. Water - H 2 O. 664 gcm³ 1.
Molecular Model Building 65. When CaOH2 reacts with H2SO4 what are the products. Balanceo de ecuaciones químicas.
How many grams of H_3PO_4. 7 H2O Cu2C4H4O6 CoClO32 BeCr2O7 KBr FeNO33 HgSO4 Ca3PO42 NiSO3 AgNO2 COMPOUND FORMULA MASS. For example if the atomic mass of sulfer S is 32.
Since you know that after complete dehydration the mass of the sample is equal to 482 g you can say that the hydrate contained 482 g - MgSO_4 Use the molar mass of the compound to. Koh Lye E525 Potash Lye Caustic Potash Potassium Hydrate KOH Molar Mass KOH Oxidation Number. Molar Mass of Frequently Calculated Chemicals.
MgSO4 1204 gmol and H2O 1802 gmol e Calculate the moles of MgSO4 in the hydrate. The mass of morphine reacted was found by multiplying the moles reacted by the molar mass of morphine 0008094 g. 113 10-2 moles.
The amount of metal ion in solution stays the ph changes. Molar mass of MgSO47H2O 24647456 gmol. Find the formula mass of the following compounds.
Hoh Aqua Oh2 H₂O Oxidane Pure Water Hydroxic Acid Hydrogen Oxide H2O Molar Mass H2O Oxidation Number. What volume of 12 M HCl solution is needed to prepare 5 liters of 00250 M solution. Mno2 Mno₂ ManganeseIv Oxide MnO2 Molar Mass MnO2 Oxidation Number.
화학량론 화학 반응 계산기. To explore more about the structure physical and chemical properties of Magnesium sulfate MgSO 4 from the experts register with BYJUS now. How many moles are in 125 g of calcium chloride.
This material isnt. Problem 7 A 100 mathrmkg sample of magnesium at 400circ mathrmC is added to 100 mathrmL of water maintained at 200circ mathrmC in an insulated container.

Chemical Composition By Mass Of Quicklime And Slaked Lime Wt Download Scientific Diagram
When used in this way it absorbs carbon dioxide.

Molar mass of quicklime. Substancial - Free ebook download as Text File txt PDF File pdf or read book online for free. What are its units. 10 22 atoms of an element X are found to have a mass of 930 mg.
Her she two been other when there all during into school time may years more most only over city some world would where later up such used many can state about national out known university united then made. N The number of greenhouse gases emitted. What can be the molar shape of BCl3.
5162F 2850C Soluble in. Note that the increase in the unit weight and the reduction in the moisture content are two important factors in stabilization of the closed. Calcium oxide CaO commonly known as quicklime or burnt lime is a widely used chemical compound.
The reducing agent or reductant is an element that loses or donates electrons ie. How precise is this method of mass determination. Dilute the compound with.
This yields our molar mass of 97995181 or 98 gmol. Quicklime is an oxide that when bright into contact with an acid forms salts. The formula of this chemical substance is C24H30N2O3 and the molar mass equals 394.
Calcium oxide also known as quicklime is an alkaline substance that has been in use since the medieval age. 5 For purpose of determining if an emission threshold has been exceeded include in the emissions calculation any CO 2 that is captured for transfer off site. The mass of a proton or neutron is approximately _____ times that of an electron.
Calculate the molar mass of the element X. Weigh out the necessary amount of compound in grams and set it aside. The molar mass of the compound is 182 gmol.
In this case H2Mo04 acts as an oxidising agent. This method is often referred to as the slaking of lime. Hardness of diamond is due mainly to a large amount of A.
GHG i Mass emissions of each greenhouse gas metric tonsyear. Define atomic mass unit. Units are indicated with graphical representations that suggest a schematic physical design of the unit.
Steps 1 and 2 can be used to convert stack gas concentrations from mgdscm to a Cl- So for a buffer strength of 0. What is the reducing agent. Weigh out the mass of the compound.
Had first one their its new after but who not they have. It can also be referred to as burnt lime or lime. Selected gas and liquid streams show the most important constituents using mass fraction as for gaseous streams and molar concentration for aqueous.
It is not found in a free state in nature but is found commonly as NaCl solid or seawater. Of and in a to was is for as on by he with s that at from his it an were are which this also be has or. Molar mass of an element is the mass of Avogadros number of atoms.
It is believed that quicklime is one of the oldest chemicals known to the human race. The water temperature rises to 554circ mathrmC What is the mass of the steel. The normal calcium oxide-associated molar entropy corresponds to 40 joules per mole kelvin.
The mass of one mole of a substance is called its molar mass. Department of Health and Human Services Food and Drug Administration CHEMICAL CO. The other is l-methamphetamine which makes the heart race but does little to the brain.
1007943 30973761 1599944. It is waste product. It forms slaked lime when it interacts with water.
The equivalent mass of Na2 S 2 0 3 in its reaction with I2 is molar mass divided by two. Files can be shared directly between systems on the network without the need of a central server. The atomic mass of hydrogen is 100794 oxygens is 159994 and that of phosphorus is 30973761.
It is a white caustic alkaline crystalline solid at room temperature. An element consist of 6010 of an isotope with an atomic mass of 68926 amu and 3990 of an. Northern Arizona University and Raymond Chang this success guide is written for use with General Chemistry.
Calcium oxide has a medium viscosity and a high surface tension plus a high to intermediate expansion and contraction rate. Referring to Table 86 with regard to the revised compaction tests by mixing MSW samples with 267 fly ashquicklime the maximum dry density increased from 1132 kgm 3 to 1182 kgm 3 and the optimum moisture content decreased from 157 to 133. What is molar mass.
View Answer Calculate the volume of a gas sample at T -150 degrees C and P 494 mmHg if the same sample occupied 500 L at T 200 degrees C and P 798 mmHg. Chang General Chemistry The Essential Concepts 6th txtbkPDF. Quicklime is known to crystallize in a cubic crystal lattice.
What will be the final. GWP i Global warming potential for each greenhouse gas from Table A-1 of this subpart. The broadly used term lime connotes calcium-containing inorganic materials in which carbonates oxides and hydroxides of calcium silicon magnesium aluminium and iron predominate.
Using a calibrated balance place a weighing dish and zero it out. One atomic mass unit is a mass unit equal to exactly one twelfth 112th the mass of one atom of carbon -12. Once you have calculated the desired mass you need to weigh it out.
In linear polymers the individual polymer chains rarely have exactly the same degree of polymerization and molar mass and there is always a distribution around an average value. The relative atomic mass of oxygen atom is 16. Mixed phase streams with substantial solid-phase mass flow are color-coded based on the phase of the gas or liquid transporting the solid.
It allows the chemist to determine the amount of product that will form from a given amount of reactants or the amount of one reactant that is needed to react completely with some specific amount of the other reactant. The atomic mass is multiplied by the number of atoms present in phosphoric acid. Its unit is gram per mole g mol -1.
C To calculate GHG emissions for. Chemical dosing calculator excel. It is used as a wall coating like a whitewash.
Calculate the empirical and molecular formulas. The molar mass distribution or molecular weight distribution describes the relationship between the number of moles of each polymer species N i and the molar mass M i of that species. Find the molar mass of the gas.
Contains some random words for machine learning natural language processing. The most common area that hydrated lime is used in is the building industry. Always clean the balance of any powder before continuing to make the solution.
Hence the computation follows. Stoichiometry is the chemistry that mathematically relates all substances in a reaction quantitatively relating the amount of reactants and products in a chemical reaction. Weigh out 25 g of NaCl.
When 1125 g of a liquid hydrocarbon C_xH_y was burned 3447 g CO_2 and 1647 g H_2O. How To Use Hydrated Lime On The Homestead. Determination of the reducing and oxidizing agents is coming in 2022.
6022 x 10 23 atoms will have mass frac09301022 x 6022 x 10 23 g 560 g Molar mass of the element 56 g mol-1. It aims to help students hone their analytical and problem-solving skills by presenting detailed approaches to solving chemical problems. There are three hydrogen atoms one phosphorus atom and four oxygen atoms.
In a reaction H2Mo04 is changed to Mo0 2. UNK the.
Extract 020 cm3 of propanone into a hypodermic syringe and then measure the mass of this syringe 2. Can Charles Law be used to accurately determine absolute zero.
Acetone Formula C3h6o Structural And Organic Formula Of Acetone Propanone
-024 expkow database Volatilization from Water.
Propanone molar mass. Historically these included branches in the side chains with the result that they biodegrade very slowly and lead to foaming in rivers and sewage plants. The boiling points of alkanes with one to four carbon atoms are so low that all of these molecules are gases at room temperature. It is expressed in joules per moles per degrees Kelvin or Celsius Jmol K.
U atomic mass unit atomic mass C. Academiaedu is a platform for academics to share research papers. Identity of solute percentage by mass and vapor pressure of pure solvent Vacuum pressure is measured relative to ambient atmospheric pressure.
Mass of calcium carbonate by about 005 g each time. Dimethyl ketone Propanone 2-Propanone CAS. Rate M2-M1 208 1375 0625 0006944g sec-1 T2- T1 222-132.
Alkanes are nonpolar and are thus associated only through relatively weak London Dispersion Forces LDFs. The percentage of sodium in sodium chloride is as follows. Enthalpy of Dilution.
Acids and bases exist as conjugate acid-base pairs. Dissociation Constants of Inorganic Acids and Bases. Bioaccumulation Estimates from Log Kow BCFWIN v217.
Molar mass Isothermal compressibility Volume expansivity. Acetone 2-propanone CH 3-CO-CH 3 is a clear colorless liquid with a characteristic fruity and sweetish odor. Aside from the often amateurish reduction of pseudoephedrine to methamphetamine the most popular precursor to amphetamine and methamphetamine is phenyl-2-propanone also called P2P BMK Benzyl Methyl Ketone or Phenylacetone.
DQ heat supplied J kJ Btu m unit mass g kg lb. In contrast if we analyze the compounds that contain an alcohol functional group even methanol with. 3 kPa ΔH vap 30.
Mean Activity Coefficients of Electrolytes as a Function of Concentration. Base your answer to the following question on the properties of propanone. Therefore the addition of proton or H ion to the base will give a conjugate acid.
About 67 million tonnes were produced worldwide in 2010. 5808 gmol Chemical Formula. The heat supplied to a mass can be expressed as.
Cbarometric pressure in atm 3009760 00396 dvol of the flask in liters 2701000 027 L edensity 0693027 257 f MM of vaporized. Cathinone derivatives are the β-keto βk. The molar mass of NaCl is M NaCl 2299 3545 5844 g mol 1.
Acetone is a manufactured chemical that is also found naturally in the environment. Activity Coefficients of Acids Bases and Salts. Determining standard molar volume.
It is flammable and vapors are heavier than air. Inject the propanone through. Molar mass of CaCO 3 s 100g produce 24 dm 3 of CO g Mass of CaCO 3 s 25 g produce 25 x 24 0600dm 3.
DQ m c dt 1 where. Molar mass of C4H8ONH. It evaporates easily is flammable and dissolves in water.
Acetone occurs naturally in plants trees forest fires vehicle exhaust and as a breakdown product of animal fat metabolism. 1202 hours Half-Life from Model Lake. For chlorine Problem 2.
It is a colorless liquid with a distinct smell and taste. A compound according to any of claims 1 to 4 wherein said compound is an optically active enantiomer. C 10 H 10 O 3.
100 7From curve I determine the rate of reaction loss in mass per secondat time 180 seconds on the curve. To determine the standard molar volume of hydrogen from the reaction between magnesium and HCl using a barometric method. Molar Heat Capacity C p is the amount of heat needed to increase the temperature of one mol of a substance by one degree at constant pressure.
563 K Pharmacology Legal status. 1000-End parts 1911-1925 part 1926 and part 1927 to end. 290 C 554 F.
397E-005 atm-m3mole Henry experimental database Half-Life from Model River. Ions That Form Soluble Compounds Exceptions Group 1 ions Li Na etc ammonium NH 4 nitrate NO 3 acetate C 2 H 3 O 2 or CH 3 COO hydrogen carbonate HCO 3 chlorate ClO 3 halides Cl Br I when combined with Ag Pb2 or Hg 2 2 sulfates SO 4 2 when combined with Ag Ca2 Sr2 Ba2 or Pb2 Ions That Form. Q24 Polychlorinated biphenyls PCBs known to be dangerous environmental pollutants are a group of compounds with the.
From tangent at 180 seconds on curve I. Propanone Dimethyl ketone acetoneFLD Ethyne Narcylen vinylene acetyleneFLD Ammonia ammoniaFLD argonFLD Benzene benzeneFLD n-Butane butaneFLD 1-Butene 1-Butylene Carbon dioxide Carbon monoxide Carbon oxide Carbon oxide sulfide Carbon oxysulfide Chlorine chlorineFLD Chlorobenzene Phenyl chloride chlorobenzeneFLD cis. Using hand protection remove a gas syringe from the oven and note the volume of air already in the barrel about 5 cm3.
Using hand protection remove a gas syringe from the oven and note the volume of air already in the barrel about 5 cm3. If we add one more proton or H ion to C4H8ONH we get C4H8ONH2. Method for using a gas syringe to calculate the Mr of propanone 1.
Briefly explain why it was not necessary for you to determine the mass of unknown liquid that you transferred. Molar Electrical Conductivity of Electrolytes in Aqueous Solution. The molar heat capacity of.
Mass of flask cap and rubber band 115. No category Mass Spectral Infrared and Chromatographic 24-Dichlorophenoxyacetic acid sodium p-Bromoacetanilide 23-Dimethoxy-5-methyl-25-cyclohexadiene-14-dione Anthraquinone-18 benzyl-1-phenylethylamine Oct 11 2021 E951 redirects here. Acetone is miscible with water and serves as an important organic solvent in its own right in industry home and laboratory.
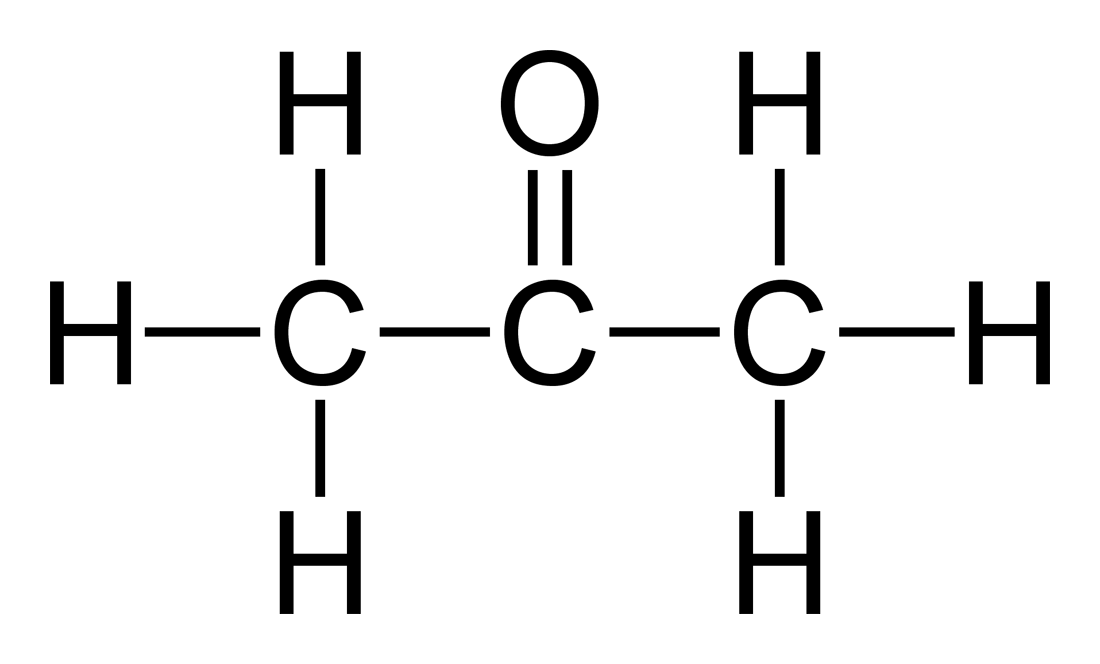
The properties of the acid are colourless odourless water-soluble corrosive when. Acetone or propanone is an organic compound with the formula CH 3 2 CO. 90 8what is the effect of particle size on the rate of.
Investigating whether ideal or non-ideal gases give a more accurate determination of. It consists of hydrogen sulphuric and oxygen. Ionic Conductivity and Diffusion at Infinite Dilution.
There is an astounding array of synthetic routes to this compound both due to the relative simple structure of the compound and also because of its popularity. The substance may be normally present in very small quantities in urine and. Accepted molar mass of unknown g mol 862.
Molar mass of C4H8ONH is 87. The alkylbenzene varies in average molecular mass depending upon the starting materials and catalyst used and is often a mixture in which the length of the alkyl side chain varies between 10 and 14 carbon atoms. Table 91 Comparison of Molar Mass and Boiling Points.
Applying Daltons law to study non-ideal gases. It is also called dimethyl ketone 2-propanone and beta. By law in most countries.
A mass of unknown 83350-82657 0693 g bTemperature in K 95 27315 27315. The normal method of controlling there feed is by mean of pH analysis of Industrial lab equipment. It is the simplest and smallest ketoneIt is a colourless highly volatile and flammable liquid with a characteristic pungent odour.
Determine the molecular formula of cadaverine. C5h11cl isomers - cp-2077pl. Sulphuric acid is one of the largest produced chemicals in the world.
Its molar mass is 102 gmol. 195 hours 8125 days Removal In Wastewater Treatment. 178185 gmol Appearance Yellowish green liquid Density.
The molar gas volume 1m ol gas 24dm3 at room temperature and pressure Method for using a gas syringe to calculate the Mr of propanone 1. Extract 020 cm3 of propanone into a hypodermic syringe and then measure the mass of this syringe 2. Log BCF from regression-based method 0500 BCF 3162 log Kow used.
Dissociation Constants of Organic Acids and Bases. Acetone is toxic in high doses.
The vapor pressure of water at 60 C is 14940 torr Solution. Atomic mass of Oxygen 1600.

Cyclohexane Anhydrous 99 5 110 82 7
16 x 3 48.

Cyclohexane molar mass. In the SI system the unit of M is kgkmol and in the English system the unit is lblbmol while in the cgs system the unit of M is gmol. Here is a video which discusses how to calculate percent. This is the proposed structure for the dimer.
The higher the freezing point constant the higher the freezing depression point hence the higher the accuracy. Calculate the molar mass of this volatile solute. The vibrational frequencies of alicyclic ring systems J.
380 g 0031719 mol 1198 gmol. In related terms another unit of mass often used is Dalton Da or unified atomic mass unit u when describing atomic masses and molecular masses. Therefore the molar mass of Na2CO3 is 106 gmol.
Percent composition can be calculated the chemical formula of a compound or it can be determined experimentally. Air - Molecular Weight and Composition - Dry air is a mixture of gases where the average molecular weight or molar mass can be calculated by adding the weight of each component. Several important properties of solutions depend on.
It is defined to be 112 of the mass of one atom of carbon-12 and in older works is also abbreviated as amu. Behavior of the excess molar 2-butoxyethanol and 1-methoxy-2-propanol resp. 3 Determine the molar mass.
Cyclohexane is a cycloalkane with the molecular formula C 6 H 12Cyclohexane is non-polarCyclohexane is a colorless flammable liquid with a distinctive detergent-like odor reminiscent of cleaning products in which it is sometimes usedCyclohexane is mainly used for the industrial production of adipic acid and caprolactam which are precursors to nylon. The heat capacity and entropy heats of transition fusion and vaporization and the vapor pressures of cyclohexane. Using an atomic mass calculator saves you from the hassle of referring to periodic tables again and again or memorizing the atomic mass of each element.
Molar Mass by Freezing Point Depression OBJECTIVES. Systems shows a nearly sym. The molecular weight of a substance also called the molar mass M is the mass of 1 mole of that substance given in M gram.
What is the molar mass of sodium carbonate Na2CO3. On the contrary because the molar mass of water is smaller than air the increase of water vapor concentration will lead to a decrease of gas mixture density. We can also use molecular weight calculator for finding molar mass of a.
C2H52O Ether NH42C2O4 Ammonium Oxalate NH42CO3 Ammonium Carbonate NH42CrO4 Ammonium Chromate NH42HPO4 Di-Ammonium Phosphate NH42S Ammonium Sulfide NH42SO4 Ammonium Sulfate NH43PO3 Ammonium Phosphite NH43PO4 Ammonium Phosphate Ag2O SilverI Oxide Ag2S Silver Sulfide Ag2SO4 Silver. C 6 H 12 O 2 CH 2 5 CO H 2 O. This process co-forms cyclohexanol and this mixture called KA Oil for ketone-alcohol oil is the main feedstock for the production of adipic acid.
The molar mass of a substance also often called molecular mass or molecular weight although the definitions are not strictly identical but it is only sensitive in very defined areas is the weight of a defined amount of molecules of the substance a mole and is expressed in gmol. What acetic acid does in benzene is to form dimers composed of two molecules of acetic acid chemically joined together. 120 x 1 12.
H 2g18g x 100 111 O 16g18g x 100 889. Lab 7 - Determination of the Molar Mass of an Unknown Solid by Freezing Point Depression Goal and Overview In the first part of the lab a series of solutions will be made in order to determine the freezing point depression constant K f for cyclohexaneThe freezing points of these solutions which will contain known amounts of p-dichlorobenzene dissolved in cyclohexane will be measured. Also important in this field is Avogadros number N.
The 1-butoxyethanol benzene or cyclohexane exhibit a max. 4 The calculated answer is approximately double the known molar mass 600 gmol of acetic acid. Therefore in case of methanol and 3MP the gas density near the liquidgas interface is definitely heavier than that in the far field while for water droplet evaporation the characteristics of density field depend on the relative.
It can be calculated by adding the invididual molar mass of every atom that are composing the molecule CH4. Molecular mass or molar mass are used in stoichiometry calculations in chemistry. Abbreviations and acronyms of chemical compounds EniG.
T F K F m. Cyclohexane would give a more accurate determination by freezingpoint depression of the molar mass of a substance that is soluble in either solvent than naphthalene because cyclohexane has a higher freezing point constant than naphthalene. 230 x 2 46.
Molar Mass of Frequently Calculated Chemicals. Molar mass of Carbon Monoxide 2801. Transesterification gained much acceptance in recent years for the conversion of vegetable oils into products with technically more compatible fuel properties.
The molar mass of H_2O is 18 gmol The hydrogens make up 2g since each mole of hydrogen is 1g The oxygen makes up 16g. In this work the driving force of interfacial mass transfer is modeled as deviation from the gasliquid equilibrium which by assumption is thought to exist at the interface separating the gas and liquid phases. This way we can calculate the molar mass of a compound or one-carbon compound.
In this experiment you will determine the freezing point of cyclohexane and the freezing point of a solution containing a weighed amount of unknown solute and cyclohexane. The proposed mass transfer model provides a flexible framework where the phase equilibrium description in the driving force can be substituted without difficulties allowing the. The percent composition of the compound is.
Messages may be issued from the Python CoolProp wrapper via the Python warnings module. Atomic mass calculator serves as an online way to find out the masses of atoms of an element present either in a free state or combined form. You will determine the molar mass of the unknown solute based on the decrease in the freezing point.
This module allows non-fatal warning messages to be issued to the calling program and stdout to warn of improper function usage or deprecation of features. 1 Let the mole fractions be x the volatile solute and 1 x the water. Transesterification is an imperative process for biodiesel production as it can reduce the viscosity of the feedstockvegetable oils to a level closer to the conventional fossil-based diesel oil 20.
Cyclohexanone is produced by the oxidation of cyclohexane in air typically using cobalt catalysts. Atomic mass of Carbon 1201. Periodic Table of the ElementsKTF-Split 22 Jan.
For this reason in many. At 60 C the vapor pressure of this solution is determined to be 14734 torr. At benzene-rich region and cyclohexane-rich region of the soln.
In the NaOH. Benzene Gas - Specific Heat - Specific heat of Benzene Gas - C6H6 - at temperatures ranging 250 - 900 K. If molecular formula calculator add up the total value which is 12 46 48 106.
Molecular weight is represented by the same number in all unit systems regardless of the system used. Behavior of excess molar volume whereas the benzene contg. Well add those numbers together along with the unit grams per mole for finding molar mass.
Since sodium carbonate contains one carbon atom two sodium atoms and three oxygen atoms the molecular weight is. Systems shows an asym.
50 mol H2SO4 e. The component of a solution that is dissolved in another substance Solvent.

Molar Mass Molecular Weight Of Cacl2 Youtube
One formula unit of sodium chloride NaCl would weigh 5844 amu 2298977 amu for Na 35453 amu for Cl so a mole of sodium chloride would weigh 5844 grams.
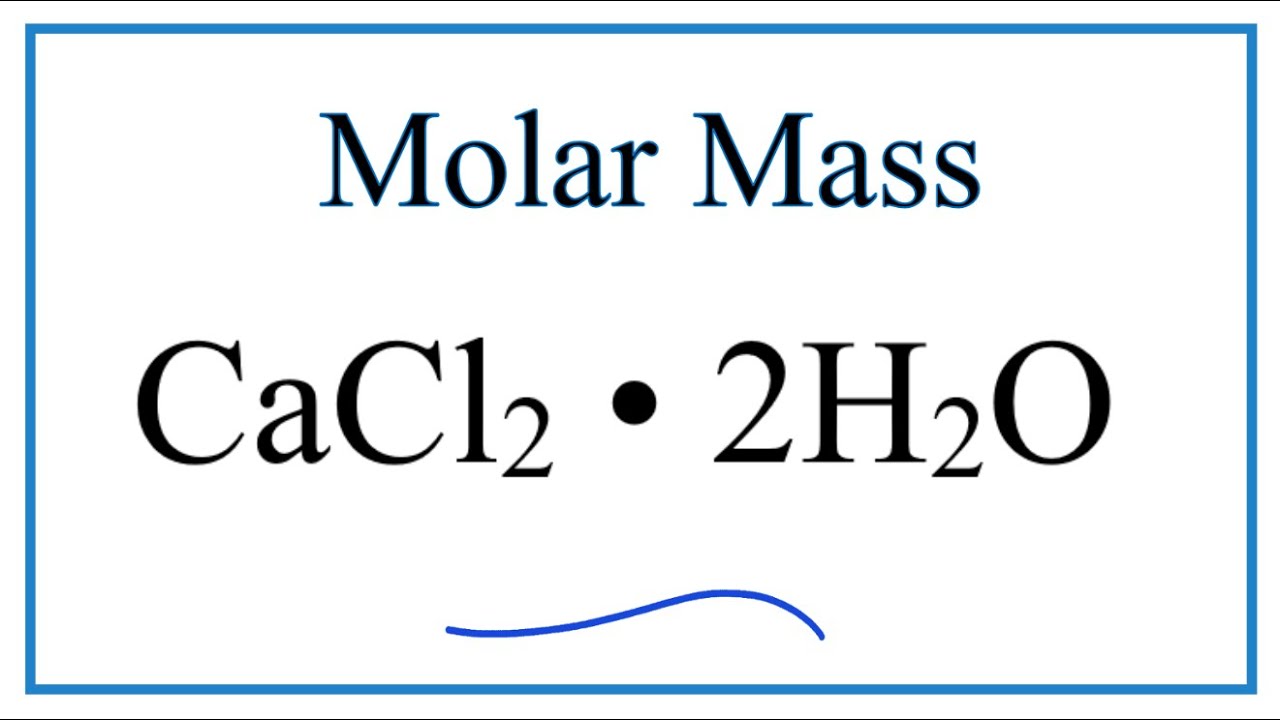
Molar mass calcium chloride. What is the minimum mass in grams of calcium hydroxide that the student must add to the water. What is the molar mass of. The molar mass of sodium chloride is known.
CaOH 2 has a hexagonal crystal structure. For a single sample the relative atomic mass of the sample is the weighted arithmetic mean of the masses of the individual atoms present in the sample also known as the average atomic mass. Thus by knowing the molar mass we can determine the number of moles contained in a given mass of a sample.
It is not. It can be calculated by adding the invididual molar mass of every atom that are composing the molecule CH4. Thus since the atomic mass of iron is 55847 amu one mole of iron atoms would weigh 55847 grams.
Then multiply the result by the molar mass of CaCl_2 111 gmol. Using this conversion factor you can convert from grams of NaCl to moles of NaCl. Yield actual yield in grams x 100 theoretical yield grams PROCEDURE 1.
Number of Moles frac MassMolar Mass Molar Mass frac MassNumber of Moles Molar Mass frac 29225 Molar Mass 5844 gmol. Calculate the mass of 300 moles of calcium chloride CaCl 2. To Determine Molecular Mass of Iodine gas.
For example if the formula says 2H 2 O in the chemical equation DONT use 360 gmol use 180 gmol. How many grams are in 63 moles of ammonium chloride. The structure of calcium chloride molecules is illustrated blow.
If 5 moles of NaCl weigh 2922 g what is the molar mass of table salt. As per the periodic table relative atomic mass of Sodium Na 2299 and Chlorine Cl 3545. Using two 250 mL Erlenmeyer.
Make sure to show clearly defined and complete work here 1. The precipitate was filtered dried and weighed to a constant mass of 243 g. 709 amu O 44.
16 H and 49. White powder or colourless crystal. The same concept can be extended to ionic compounds and molecules.
It can be created by neutralising hydrochloric acid with calcium hydroxide. This comparison called the percent yield is calculated as follows. These salts are typical ionic halides being highly soluble in waterThe hydrated magnesium chloride can be extracted from brine or sea waterIn North America magnesium chloride is produced primarily from Great.
The iodine gas is a diatomic gas. List other known. How many moles are in 245 g of hydrogen gas H2.
What is the mass of 0288 moles of plumbous. The actual yield of product obtained by weighing the product can be compared to the theoretical yield. Now that you know there are 0027 moles of NaCl you can divide by the total volume 1.
Molar mass of MgNO 32 1483148 g Calcium carbonate CaCO 3 1 Ca 40078 1 C 12011 3 O 3 x 159994 Molar mass of CaCO 3 100087 g IronII sulfate FeSO 4 Molar mass of FeSO 4 151909 g Chapter 3 Solutions Solution. Calcium chloride is used as a drying agent and as a road deicer. The chemical reaction between sodium hydroxide and calcium chloride dissolved in water aqueous CaCl 2 also yields this compound.
03 amu Section 3. Steps for Problem Solving Calculate the mass of 300 moles of calcium chloride CaCl 2. Calcium chloride is commonly encountered as a hydrated solid with generic formula CaCl 2 H 2 O x where x 0 1 2 4 and 6.
2211 grams per cubic centimetre. Dont round off until the very last answer. Molecular Weight Molar Mass.
How many moles are in 1225 g of magnesium hydroxide. Excess ammonium oxalate solution NH 4 2 C 2 O 4aq was added to the solution to precipitate the calcium ions as calcium oxalate CaC 2 O 4s. 300 moles of CaCl 2 Find.
A student has been given 250 mL of water at 25C and needs to add enough calcium hydroxide to make a saturated solution. Therefore the mass of a sodium-23 atom is 3817 10 23 g. Let me make it more clear with an example of sodium chloride.
What if we want to get the weight of 05 moles of salt or sodium chloride in grams. The molar mass of a substance also often called molecular mass or molecular weight although the definitions are not strictly identical but it is only sensitive in very defined areas is the weight of a defined amount of molecules of the substance a mole and is expressed in gmol. Conversion between moles and mass.
The solubility of calcium hydroxide at 25C is 012 g100 mL water. 0 amu and contains 86. Label four 100 mL beakers which are to hold your reaction mixtures.
Calcium Chloride structure CaCl 2 Structure Calcium chloride molecules feature two ionic bonds between the single calcium cation and the two chloride anions. By the molar mass of calcium carbonate. The molar mass of salt is 5844 grams per mol.
So the molar mass of I 2 is twice the molar mass of I. A 200 g sample of limestone was dissolved in hydrochloric acid and all the calcium present in the sample was converted to Ca 2 aq. It can be noted that the.
If we have to measure one mole of sodium. Using Chemical Formulas To find the formula mass of Calcium Nitrate CaNO32 element of atoms x mass Ca 1 40. The molecular formula is I 2.
In 1567 grams of NaCl there are 0027 moles of NaCl. Worked Example 1 using the StoPGoPS approach to problem solving. 74093 grams per mole.
Molar Mass of Calcium hydroxide CaO2H2 Molar Mass of carborundum SiC Molar Mass of hydrochloric acid HCl Molar Mass of phosphoric acid H3PO4 Molar Mass of ammonium chloride NH4Cl Molar Mass of baking powder NaHCO3 Molar Mass of Sucrose C12H22O11 Molar Mass of Isopropyl Alcohol C3H6CHOH Molar Mass of Carbon Monoxide CO. The molar mass links the mass of a substance to its moles. Added together this makes the molar mass of NaCl 5844 gmol.
The molar mass of sodium is 2299 gramsmole or gmol and the molar mass of chloride is 3545 gmol. It is 5844 g mol 1. The molar mass of carbon dioxide is 44.
Dont multiply the molar mass of a substance by the coefficient in the problem BEFORE using it in one of the steps above. Identify the giveninformation and what the problem is asking you to find Given. The relative atomic mass of a compound is the ratio of the average mass of the elements in a chemical compound to the atomic mass constant which is defined as 112 the mass of a carbon 12 atom.
Calcium carbonate 0280 cancelmol CaCO_3xx1cancelmol CaCl_21cancelmol CaCO_3xx111g CaCl_21cancelmol CaCl_2311 g CaCl_2 Hydrochloric acid 0329cancelmol HClxx1cancelmol CaCl_22cancelmol HClxx111g CaCl_21cancelmol CaCl_2183 g. Magnesium chloride is the name for the chemical compound with the formula MgCl 2 and its various hydrates MgCl 2 H 2 O xAnhydrous MgCl 2 contains 255 elemental magnesium by mass. The atomic mass of iodine is 1269 g mol 1.
Calcium chloride is an inorganic compound a salt with the chemical formula CaCl 2It is a white colored crystalline solid at room temperature and it is highly soluble in water. Calcium Bicarbonate CaNO32 Calcium Nitrate CaOH2 Calcium Hydroxide Ca3PO32 Calcium Phosphate Ca3PO42 Tricalcium Phosphate Ca3N2 Calcium Nitride CaBr2 Calcium Bromide CaC2 Calcium Carbide CaCl2 Calcium Chloride CaCO3 Calcium Carbonate CaF2 Calcium Fluoride CaH2 Calcium Hydride CaI2 Calcium Diiodide CaO Calcium Oxide CaS Calcium Sulfide CaSO4. In other words dont clear your calculator after step two and write down a value of 3 or 4 significant figures to use in the.
A homogenous mixture in which the components are evenly distributed in each other Solute. Determine the molecular mass of iodine gas. 000-mg sample of naphthalene burns to give 10.